Abstract
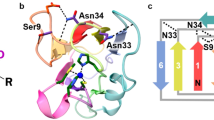
Elucidating how proteins control the reduction potentials (E0′) of [Fe–S] clusters is a longstanding fundamental problem in bioinorganic chemistry. Two site-directed variants of Azotobacter vinelandii ferredoxin I (FdI) that show large shifts in [Fe–S] cluster E0′ (100–200 mV versus standard hydrogen electrode (SHE)) have been characterized. High resolution X-ray structures of F2H and F25H variants in their oxidized forms, and circular dichroism (CD) and electron paramagnetic resonance (EPR) of the reduced forms indicate that the overall structure is not affected by the mutations and reveal that there is no increase in solvent accessibility nor any reorientation of backbone amide dipoles or NH–S bonds. The structures, combined with detailed investigation of the variation of E0′ with pH and temperature, show that the largest increases in E0′ result from the introduction of positive charge due to protonation of the introduced His residues. The smaller (50–100 mV) increases observed for the neutral form are proposed to occur by directing a Hδ+–Nδ− dipole toward the reduced form of the cluster.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Beinert, H. J. Biol. Inorg. Chem. 5, 2–15 (2000).
Bertini, I., Gori-Savellini, G. & Luchinat, C. J. Bio. Inorg. Chem. 2, 114–118 (1997).
Przysiecki, C.T., Meyer, T.E. & Cusanovich, M.A. Biochemistry 24, 2542–2549 (1985).
Stephens, P.J., Jollie, D.R. & Warshel, A. Chem. Rev. 96, 2491–2513 (1996).
Heering, H.A., Bulsink, Y.B.M., Hagen, W.R. & Meyer, T.E. Eur. J. Biochem. 232, 811–817 (1995).
Iwagami, S.G. et al. Protein Sci. 4, 2562–2572 (1995).
Soriano, A., Li, D., Bian, S., Agarwal, A. & Cowan, J.A. Biochemistry 35, 12479–12486 (1996).
Denke, E. et al. J. Biol. Chem. 271, 9085–9093 (1998).
Schroter, T. et al. Eur. J. Biochem. 255, 100–106 (1998).
Eidsness, M.K. et al. Biochemistry 38, 14803–14809 (1999).
Backes, G. et al. J. Am. Chem. Soc. 113, 2055–2064 (1991).
Shen, B. et al. J. Biol. Chem. 269, 8564–8575 (1994).
Mauk, A.G. & Moore, G.R. J. Biol. Inorg. Chem. 2, 119–125 (1997).
Náray-Szabó, G. J. Biol. Inorg. Chem. 2, 135–138 (1997).
Warshel, A., Papazyan, A. & Muegge, I. J. Biol. Inorg. Chem. 2, 143–152 (1997).
Moore, G.R. FEBS Lett. 161, 171–175 (1983).
Schejter, A. & Eaton, W.A. Biochemistry 23, 1081–1084 (1984).
Chen, K. et al. J. Biol. Chem. 274, 36479–36487 (1999).
Li, J., Nelson, M.R., Peng, C.Y., Bashford, D. & Noodleman, L. J. Phys. Chem. 102, 6311–6324 (1998).
Shen, B. et al. J. Biol. Chem. 268, 25928–25939 (1993).
Chen, K. et al. Nature 405, 814–817 (2000).
Ryle, M.J., Lanzilotta, W.N. & Seefeldt, L.C. Biochemistry 35, 9424–9434 (1996).
Iismaa, S.E. et al. J. Biol. Chem. 266, 21563–21571 (1991).
Armstrong, F.A., Butt, J.N. & Sucheta, A. Methods Enzymol. 227, 479–500 (1993).
Dutton, P.L. & Wilson, D.F. Biochim. Biophys. Acta 346, 165–212 (1974).
Stephens, P.J. et al. Biochemistry 30, 3200–3209 (1991).
Hunter, E.P. & Lias, S.G. J. Phys. Chem. Ref. Data 27, 413 (1998).
Delahay, P., Pourbaix, M. & Van Rysselberghe, P. J. Chem. Educ. 27, 683–688 (1950).
Armstrong, F.A., Heering, H.A. & Hirst, J. Chem. Soc. Rev. 26, 169–179 (1997).
Acknowledgements
The authors wish to acknowledge V. Roberts and L. Noodleman at The Scripps Research Institute, and M. Gunner, City College, New York, and J. Hirst, MRC, Cambridge for helpful discussions. G. Murcott (Oxford) assisted in some of the experiments. We also thank T.L. Poulos and H. Li at University of California-Irvine for use of laboratory facilities for X-ray crystallography. This work was funded by grants from UK EPSRC and BBSRC to F.A.A. and NIH grants to C.D.S. and B.K.B.
Barbara K. Burgess died unexpectedly on December 30, 2001. We would like to pay tribute to her many contributions to science; in particular her studies of the proteins and cofactors involved in biological nitrogen fixation, and her elucidation of many important properties of iron-sulfur clusters.
Author information
Authors and Affiliations
Additional information
Correspondence should be addressed to Susan Bryant. email: svbryant@uci.edu
Rights and permissions
About this article
Cite this article
Chen, K., Bonagura, C., Tilley, G. et al. Crystal structures of ferredoxin variants exhibiting large changes in [Fe–S] reduction potential. Nat Struct Mol Biol 9, 188–192 (2002). https://doi.org/10.1038/nsb751
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb751