Abstract
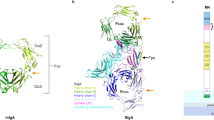
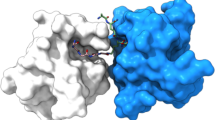
Streptococcal superantigen (SSA) is a 28,000 Mr toxin originally isolated from a pathogenic strain of Streptococcus pyogenes that has 60% sequence identity with staphylococcal enterotoxin B (SEB). SSA and SEB, however, do not compete for binding on the surfaces of cells expressing MHC class II molecules. This behavior had been ascribed to SSA and SEB binding to distinct sites on, or different subsets of, HLA–DR molecules. Here we demonstrate that SSA binds predominantly to HLA–DQ, rather than to HLA–DR molecules, and present the crystal structure of SSA at 1.85 Å resolution. These data provide a structural basis for interpreting the interaction of SSA with HLA–DQ molecules as well as a foundation for understanding bacterial superantigen affinities for distinct MHC isotypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Scherer, M.T. et al. Ann. Rev. Cell Biol. 9, 101–128 (1993).
Kotzin, B.L., Leung, D.Y., Kappler, J. & Marrack, P. Adv. Immunol. 54, 99–165 (1993).
Conrad, B. et al. Cell 90, 303–313 (1997).
Renno, T. & Acha–Orbea, H. Immunol. Rev. 154, 175–191 (1996).
Jardetzky, T.S. et al. Nature 368, 711–718 (1994).
Kim, J., Urban, R.G., Strominger, J.L. & Wiley, D.C. Science 266, 1870–1874 (1994).
Kozono, H., Parker, D., White, J., Marrack, P. & Kappler, J. Immunity 3, 187–196 (1995).
Thibodeau, J. et al. Science 266, 1874–1878 (1994).
Lavoie, P.M. et al. Proc. Natl. Acad. Sci. USA 94, 6892–6897 (1997).
Stevens, K.R., Van, M., Lamphear, J.G. &nd Rich, R.R. J. Immunol. 157, 4970–4978 (1996).
Toyosaki, T. et al. Eur. J. Immunol. 26, 2693–2701 (1996).
Norrby–Teglund, A. et al. Infect. Immun. 62, 5227–5233 (1994).
Esaki, Y. et al. Infect. Immun. 62, 1228–1235 (1994).
Busch, R., Cloutier, I., Sékaly, R.–P. & Hämmerling, G.J. EMBO J. 15, 418–428 (1996).
Mollick, J.A. et al. J. Clin. Invest. 92, 710–719 (1993).
Deringer, J.R., Ely, R.J., Stauffacher, C.V. & Bohach, G.A. Mol. Microbiol. 22, 523–534 (1996).
Abrahmsén, L. et al. EMBO J. 14, 2978–2986 (1995).
Hudson, K.R. et al. J. Exp. Med. 182, 711–720 (1995).
Clackson, T. & Wells, J.A. Science 267, 383–386 (1995).
Fremont, D.H. et al. Immunity 8, 305–317 (1998).
Scott, C.A., Peterson, P.A., Teyton, L. & Wilson, I.A. Immunity 8, 319–329 (1998).
Kabsch, W. J. Appl. Crystallogr. 21, 916 (1988).
Otwinowski, Z. Data collection and processing (Warrington, UK; 1993).
Navaza, J. Acta Crystallogr. A50, 157–163 (1994).
Collaborative Computational Project No. 4. Acta Crystallogr. D50, 760–763 (1994).
Read, R.J. Acta Crystallogr. A42, 140–149 (1986).
Brünger, A.T. XPLOR, V3.1 (Yale University Press, New Haven, Connecticut; 1992).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Acta Crystallogr. A 47, 110–119 (1991).
Evans, S.V. J. Mol. Graphics 11, 131–138 (1993).
Kraulis, P.J. J. Appl. Crystallogr. 24, 946–950 (1991).
Nicholls, A., Sharp, K. and Honig, B. Proteins 11, 281ff (1991).
Acknowledgements
We thank K.R. Stevens and R. Rich for the kind contribution of the SSA–expressing E. coli strain, L. Gu for the L243 antibody, D. Wiley for the CHAMP antibody, L. Bermann for assistance at the BNL–NSLS X–25A beamline, as well as P. Lavoie and R–P. Sékaly for provision of cell lines and for hosting E.S. in the Sékaly laboratory (Montreal, Quebec, Canada) in order to learn the experimental techniques of the cell binding assay. We also thank S. Garman and P. Lavoie for careful reading of, and editorial comments on, the manuscript. E.S. is supported by a grant from the NIH. This work has been funded by a grant from the NIH and an award from the Human Frontier Science Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sundberg, E., Jardetzky, T. Structural basis for HLA–DQ binding by the streptococcal superantigen SSA. Nat Struct Mol Biol 6, 123–129 (1999). https://doi.org/10.1038/5809
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/5809
This article is cited by
-
Elevated risk of invasive group A streptococcal disease and host genetic variation in the human leucocyte antigen locus
Genes & Immunity (2020)
-
Streptococcal toxic shock syndrome in the intensive care unit
Annals of Intensive Care (2018)
-
HLA-typing analysis following allogeneic bone grafting for sinus lifting
Cell and Tissue Banking (2017)
-
HLA-DQ6 and ingestion of contaminated water: possible gene–environment interaction in an outbreak of Leptospirosis
Genes & Immunity (2004)