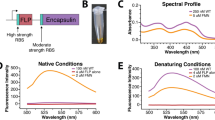
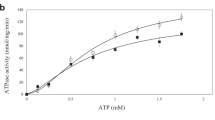
Abstract
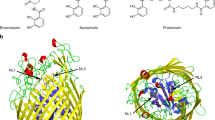
Ferritin is characterized by a highly conserved architecture that comprises 24 subunits assembled into a spherical cage with 432 symmetry. The only known exception is the dodecameric ferritin from Listeria innocua. The structure of Listeria ferritin has been determined to a resolution of 2.35 Å by molecular replacement, using as a search model the structure of Dps from Escherichia coli. The Listeria 12-mer is endowed with 23 symmetry and displays the functionally relevant structural features of the ferritin 24-mer, namely the negatively charged channels along the three-fold symmetry axes that serve for iron entry into the cavity and a negatively charged internal cavity for iron deposition. The electron density map shows 12 iron ions on the inner surface of the hollow core, at the interface between monomers related by two-fold axes. Analysis of the nature and stereochemistry of the iron-binding ligands reveals strong similarities with known ferroxidase sites. The L. innocua ferritin site, however, is the first described so far that has ligands belonging to two different subunits and is not contained within a four-helix bundle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Harrison, P. & Arosio, P. Biochim. Biophys. Acta 1275, 161–203 (1996).
Bozzi, M. et al. J. Biol. Chem. 272, 3259– 3265 (1997).
Ilari, A., Savino, C., Stefanini, S., Chiancone, E. & Tsernoglou, D. Acta Crystallogr. D 55, 552–553 (1999).
Ford, G.C. et al. Philos. Trans. R. Soc. Lond. B 304, 551 –565 (1984).
Boyd, D., Vecoli, C., Belcher, D.M., Jain, S.K. & Drysdale, J.W. J. Biol. Chem. 260 , 11755–11761 (1985).
Hempstead, D.P. et al. J. Mol. Biol. 268, 424– 448 (1997).
Stefanini, S., Cavallo, S., Montagnini, B. & Chiancone, E. Biochem. J. 338, 71–75 ( 1999).
Levi, S. et al. Biochemistry 28, 5179–5184 (1989).
Sun, S., Arosio, P. Levi, S. & Chasteen, N.D. Biochemistry 32, 9362–9369 ( 1993).
Le Brun, N.E. et al. Biochem. J. 312, 385– 392 (1995).
Grant, R.A., Filman, D.J., Finkel, S.E., Kolter, R. & Hogle, J.M. Nature Struct. Biol. 5, 294–303 (1998).
Levi, S. et al. Biochem. J. 264, 381– 388 (1989).
Stefanini, S., Desideri, A., Vecchini, P., Drakenberg, T. & Chiancone E. Biochemistry 28, 378–382 (1989).
Treffry, A. et al. Biochem. J. 296, 721– 728 (1993).
Douglas, T. & Ripoll, D.R. Protein Sci. 7, 1083–1091 (1998).
Nicholls, A., Bharadwaj, R. & Honig, B. Biophys. J. 64, A 166 (1993).
Lawson, D.M. et al. Nature 349, 541–544 (1997).
Frolow, F., Kalb, A.J. (Gilboa) & Yariv, J. Nature Struct. Biol. 1, 453– 460 (1994).
deMaré, F., Kurtz, D.M., Jr. & Nordlund, P. Nature Struct. Biol. 3, 539–546 (1996).
Wade, V.J. et al. J. Mol. Biol. 221, 1443– 1452 (1991).
Treffry, A., Zhao, Z., Quail, M.A., Guest, J.R. & Harrison, P.M. Biochemistry 36, 432– 441 (1997).
Hempstead, P.D. et al. FEBS Lett. 350, 258– 262 (1994).
Bonomi, F. Kurtz, D.M. Jr. & Cui, X. J. Biol. Inorg. Chem. 1, 67–72 (1996).
Ottwinowski, Z. & Minor, W. Methods Enzymol. 276, 307–326 ( 1997).
Navaza, J. Acta Crystallogr. A 50, 157–163 (1994).
Cowtan, K. Newslett. Protein Crystallogr. 31, 34– 38 (1994).
Jones, A.T., Zou, Y.-J. & Kjeldgaard, M. Acta Crystallogr. A 42, 140– 149 (1994).
Tronrud, D.E., Ten Eyck, L.F. & Matthews, B.W. Acta Crystallogr. A 43, 489 –501 (1987).
Laskowski, R.A., McArthur, M.W., Moss, D.S. & Thornton . J. Appl. Crystallogr. 26, 283–291 ( 1993).
Brünger, A.T. J. Mol. Biol. 203, 803–816 (1998).
Powell, M.J.D. Math. Program. 12, 241–254 (1977).
Kraulis, P.J. J. Appl. Crystallogr. 26, 283–291 (1993).
Acknowledgements
This work was supported in part by grants from the Istituto Pasteur-Fondazione Cenci Bolognetti and the Agenzia Spaziale Italiana (to D.T.) and from the Ministero per l'Università e Ricerca Scientifica e Tecnologica, Progetto Biologia Strutturale (to E.C. and D.T.). The authors are grateful to M. Bozzi for providing the purified protein, to P. Kanellopoulos for data measurement at DESY and to M. Rizzi for continuous help and fruitful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ilari, A., Stefanini, S., Chiancone, E. et al. The dodecameric ferritin from Listeria innocua contains a novel intersubunit iron-binding site. Nat Struct Mol Biol 7, 38–43 (2000). https://doi.org/10.1038/71236
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/71236
This article is cited by
-
Dps–DNA interaction in Marinobacter hydrocarbonoclasticus protein: effect of a single-charge alteration
European Biophysics Journal (2021)
-
Disclosing the Molecular Mechanism of Iron Incorporation in Listeria innocua Dps by EPR Spectroscopy
Applied Magnetic Resonance (2020)
-
Effect of the charge distribution along the “ferritin-like” pores of the proteins from the Dps family on the iron incorporation process
JBIC Journal of Biological Inorganic Chemistry (2011)
-
Dps-like proteins: structural and functional insights into a versatile protein family
Cellular and Molecular Life Sciences (2010)
-
Crystal structures of Streptococcus pyogenes Dpr reveal a dodecameric iron-binding protein with a ferroxidase site
JBIC Journal of Biological Inorganic Chemistry (2010)