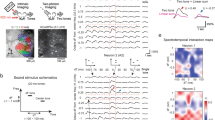
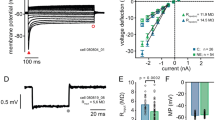
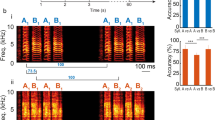
Abstract
Sound localization by auditory brainstem nuclei relies on the detection of microsecond interaural differences in action potentials that encode sound volume and timing. Neurons in these nuclei express high amounts of the Kv3.1 potassium channel, which allows them to fire at high frequencies with short-duration action potentials. Using computational modeling, we show that high amounts of Kv3.1 current decrease the timing accuracy of action potentials but enable neurons to follow high-frequency stimuli. The Kv3.1b channel is regulated by protein kinase C (PKC), which decreases current amplitude. Here we show that in a quiet environment, Kv3.1b is basally phosphorylated in rat brainstem neurons but is rapidly dephosphorylated in response to high-frequency auditory or synaptic stimulation. Dephosphorylation of the channel produced an increase in Kv3.1 current, facilitating high-frequency spiking. Our results indicate that the intrinsic electrical properties of auditory neurons are rapidly modified to adjust to the ambient acoustic environment. *Note: In the version of this article initially published online, some of the authors’ affiliations were incorrectly reported. These affiliations have been corrected for the HTML and print versions of the article. The correct affiliations are Ping Song 1 , Yue Yang 2 , Margaret Barnes-Davies 3,5 , Arin Bhattacharjee 1,5 , Martine Hamann 3 , Ian D Forsythe 4 , Douglas L Oliver 2 & Leonard K Kaczmarek 1
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Perney, T.M., Marshall, J., Martin, K.A., Hockfield, S. & Kaczmarek, L.K. Expression of the mRNAs for the Kv3.1 potassium channel gene in the adult and developing brain. J. Neurophysiol. 68, 756–766 (1992).
Lenz, S., Perney, T.M., Qin, Y., Robbins, E. & Chesselet, M.F. GABAergic interneurons of the striatum express the Shaw-like potassium channel Kv3.1. Synapse 18, 55–66 (1994).
Weiser, M. et al. The potassium channel subunit Kv3.1b is localized to somatic and axonal membranes of specific populations of CNS neurons. J. Neurosci. 15, 4298–4314 (1995).
Brew, H.M. & Forsythe, I.D. Two voltage-dependent K+ conductances with complementary functions in postsynaptic integration at a central auditory synapse. J. Neurosci. 15, 8011–8022 (1995).
Sekirnjak, C. et al. Subcellular localization of the K+ channel subunit Kv3.1b in selected rat CNS neurons. Brain Res. 766, 173–187 (1997).
Rudy, B. & McBain, C.J. Kv3 channels: voltage-gated K+ channels designed for high-frequency repetitive firing. Trends Neurosci. 24, 517–526 (2001).
Matsukawa, H., Wolf, A.M., Matsushita, S., Joho, R.H. & Knopfel, T. Motor dysfunction and altered synaptic transmission at the parallel fiber-Purkinje cell synapse in mice lacking potassium channels Kv3.1 and Kv3.3. J. Neurosci. 23, 7677–7684 (2003).
Smith, P.H., Joris, P.X., Carney, L.H. & Yin, T.C. Projections of physiologically characterized globular bushy cell axons from the cochlear nucleus of the cat. J. Comp. Neurol. 304, 387–407 (1991).
Wu, S.H. & Kelly, J.B. Response of neurons in the lateral superior olive and medial nucleus of the trapezoid body to repetitive stimulation: intracellular and extracellular recordings from mouse brain slice. Hear. Res. 68, 189–201 (1993).
Satzler, K. et al. Three-dimensional reconstruction of a calyx of Held and its postsynaptic principal neuron in the medial nucleus of the trapezoid body. J. Neurosci. 22, 10567–10579 (2002).
Oertel, D. Encoding of timing in the brain stem auditory nuclei of vertebrates. Neuron 19, 959–962 (1997).
Trussell, L.O. Synaptic mechanisms for coding timing in auditory neurons. Annu. Rev. Physiol. 61, 477–496 (1999).
Kopp-Scheinpflug, C., Lippe, W.R., Dorrscheidt, G.J. & Rubsamen, R. The medial nucleus of the trapezoid body in the gerbil is more than a relay: comparison of pre- and postsynaptic activity. J. Assoc. Res. Otolaryngol. 4, 1–23 (2003).
Kanemasa, T., Perney, T.M., Gan, L., Wang, L.Y. & Kaczmarek, L.K. Electrophysiological and pharmacological characterization of a mammalian Shaw channel expressed in NIH 3T3 fibroblasts. J. Neurophysiol. 74, 207–217 (1995).
Wang, L.Y., Gan, L., Forsythe, I.D. & Kaczmarek, L.K. Contribution of the Kv3.1 potassium channel to high-frequency firing in mouse auditory neurons. J. Physiol. (Lond.) 509, 183–194 (1998).
Rudy, B. et al. Contributions of the Kv3 channels to neuronal excitability. Ann. NY Acad. Sci. 868, 304–343 (1999).
Macica, C.M. et al. Modulation of the kv3.1b potassium channel isoform adjusts the fidelity of the firing pattern of auditory neurons. J. Neurosci. 23, 1133–1141 (2003).
Parameshwaran-Iyer, S., Carr, C.E. & Perney, T.M. Localization of KCNC1 (Kv3.1) potassium channel subunits in the avian auditory nucleus magnocellularis and nucleus laminaris during development. J. Neurobiol. 55, 165–178 (2003).
von Hehn, C.A., Bhattacharjee, A. & Kaczmarek, L.K. Loss of Kv3.1 tonotopicity and alterations in cAMP response element-binding protein signaling in central auditory neurons of hearing impaired mice. J. Neurosci. 24, 1936–1940 (2004).
Luneau, C.J. et al. Alternative splicing contributes to K+ channel diversity in the mammalian central nervous system. Proc. Natl. Acad. Sci. USA 88, 3932–3936 (1991).
Ozaita, A., Martonem, M.E., Ellisman, M.H. & Rudy, B. Differential subcellular localization of the two alternatively spliced isoforms of the Kv3.1 potassium channel subunit in brain. J. Neurophysiol. 88, 394–408 (2002).
Critz, S.D., Wible, B.A., Lopez, H.S. & Brown, A.M. Stable expression and regulation of a rat brain K channel. J. Neurochem. 60, 1175–1178 (1993).
Paolini, A.G., FitzGerald, J.V., Burkitt, A.N. & Clark, G.M. Temporal processing from the auditory nerve to the medial nucleus of the trapezoid body in the rat. Hear. Res. 159, 101–116 (2001).
Parameshwaran, S., Carr, C.E. & Perney, T.M. Expression of the Kv3.1 potassium channel in the avian auditory brainstem. J. Neurosci. 21, 485–494 (2001).
Li, W., Kaczmarek, L.K. & Perney, T.M. Localization of two high-threshold potassium channel subunits in the rat central auditory system. J. Comp. Neurol. 437, 196–218 (2001).
Phillips, D.P. & Hall, S.E. Psychophysical evidence for adaptation of central auditory processors for interaural differences in time and level. Hear. Res. 202, 188–199 (2005).
Fujisaki, W., Shimojo, S., Kashino, M. & Nishida, S. Recalibration of audiovisual simultaneity. Nat. Neurosci. 7, 773–778 (2004).
Kopp-Scheinpflug, C., Dehmel, S., Dorrscheidt, G.J. & Rubsamen, R. Interaction of excitation and inhibition in anteroventral cochlear nucleus neurons that receive large endbulb synaptic endings. J. Neurosci. 22, 11004–11018 (2002).
Sanes, D.H. An in vitro analysis of sound localization mechanisms in the gerbil lateral superior olive. J. Neurosci. 10, 3494–3506 (1990).
Grothe, B. & Park, T.J. Structure and function of the bat superior olivary complex. Microsc. Res. Tech. 51, 382–402 (2000).
Brand, A., Behrend, O., Marquardt, T., McAlpine, D. & Grothe, B. Precise inhibition is essential for microsecond interaural time difference coding. Nature 417, 543–547 (2002).
Augustine, C.K. & Bezanilla, F. Phosphorylation modulates potassium conductance and gating current of perfused giant axon of squid. J. Gen. Physiol. 95, 245–271 (1990).
Perozo, E. & Bezanilla, F. Phosphorylation affects voltage gating of the delayed rectifier K+ channel by electrostatic interactions. Neuron 5, 685–690 (1990).
Levitan, I.B. Modulation of ion channels by protein phosphorylation and dephosphorylation. Annu. Rev. Physiol. 56, 193–212 (1994).
Jonas, E.A. & Kaczmarek, L.K. Regulation of potassium channels by protein kinases. Curr. Opin. Neurobiol. 6, 318–323 (1996).
Misonou, H. et al. Regulation of ion channel localization and phosphorylation by neuronal activity. Nat. Neurosci. 7, 711–718 (2004).
Wang, L.Y., Gan, L., Perney, T.M., Schwartz, I. & Kaczmarek, L.K. Activation of Kv3.1 channels in neuronal spine-like structures may induce local potassium ion depletion. Proc. Natl. Acad. Sci. USA 95, 1882–1887 (1998).
Sommer, I., Lingenhohl, K. & Friauf, E. Principal cells of the rat medial nucleus of the trapezoid body: an intracellular in vivo study of their physiology and morphology. Exp. Brain Res. 95, 223–239 (1993).
Cohen, Y.E. & Saunders, J.C. The effect of acoustic overexposure on the tonotopic organization of the nucleus magnocellularis. Hear. Res. 81, 11–21 (1994).
Weiser, M. et al. Differential expression of Shaw-related K+ channels in the rat central nervous system. J. Neurosci. 14, 949–972 (1994).
Baranauskas, G., Tkatch, T., Nagata, K., Yeh, J.Z. & Surmeier, D.J. Kv3.4 subunits enhance the repolarizing efficiency of Kv3.1 channels in fast-spiking neurons. Nat. Neurosci. 6, 258–266 (2003).
Moreno, H. et al. Thalamocortical projections have a K+ channel that is phosphorylated and modulated by cAMP-dependent protein kinase. J. Neurosci. 15, 5486–5501 (1995).
Atzori, M. et al. H2 histamine receptor-phosphorylation of Kv3.2 modulates interneuron fast spiking. Nat. Neurosci. 3, 791–798 (2000).
Covarrubias, M., Wei, A., Salkoff, L. & Vyas, T.B. Elimination of rapid potassium channel inactivation by phosphorylation of the inactivation gate. Neuron 13, 1403–1412 (1994).
Antz, C. et al. Control of K+ channel gating by protein phosphorylation: structural switches of the inactivation gate. Nat. Struct. Biol. 6, 146–150 (1999).
Barnes-Davies, M., Barker, M.C., Osmani, F. & Forsythe, I.D. Kv1 currents mediate a gradient of principal neuron excitability across the tonotopic axis in the rat lateral superior olive. Eur. J. Neurosci. 19, 325–333 (2004).
Hamann, M., Billups, B. & Forsythe, I.D. Non-calyceal excitatory inputs mediate low fidelity synaptic transmission in rat auditory brainstem slices. Eur. J. Neurosci. 18, 2899–2902 (2003).
Acknowledgements
This work was supported by US National Institutes of Health grants DC01919 (L.K.K.) and DC00189 (D.L.O.) and the Wellcome Trust (I.D.F., M.H. & M.B.-D.). Part of this research was conducted in a facility constructed with support from Research Facilities Improvement grant C06 RR13551 from the National Center for Research Resources, US National Institutes of Health. We thank M. Browning of PhosphoSolutions for generation of the Kv3.1b phospho-specific antibody.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Phospho-Kv3.1b is present both pre- and post-synaptically in MNTB neurons. (PDF 174 kb)
Supplementary Fig. 2
Inhibition of AMPA and NMDA receptors attenuates stimulation-induced dephosphorylation of Kv3.1b channels. (PDF 250 kb)
Supplementary Fig. 3
Computer simulations of the effects of an increase in Kv3.1 conductance on the firing pattern of a representative model MNTB neuron. (PDF 35 kb)
Supplementary Fig. 4
Lack of significant change in the firing pattern of a real MNTB neuron that received no synaptic input. (PDF 40 kb)
Rights and permissions
About this article
Cite this article
Song, P., Yang, Y., Barnes-Davies, M. et al. Acoustic environment determines phosphorylation state of the Kv3.1 potassium channel in auditory neurons. Nat Neurosci 8, 1335–1342 (2005). https://doi.org/10.1038/nn1533
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1533
This article is cited by
-
Emergence of 5-HT5A signaling in parvalbumin neurons mediates delayed antidepressant action
Molecular Psychiatry (2020)
-
Precisely timed inhibition facilitates action potential firing for spatial coding in the auditory brainstem
Nature Communications (2018)
-
Evaluation of Possible Effects of a Potassium Channel Modulator on Temporal Processing by Cochlear Implant Listeners
Journal of the Association for Research in Otolaryngology (2018)
-
Pharmacological modulation of Kv3.1 mitigates auditory midbrain temporal processing deficits following auditory nerve damage
Scientific Reports (2017)
-
Redistribution of Kv1 and Kv7 enhances neuronal excitability during structural axon initial segment plasticity
Nature Communications (2015)