Abstract
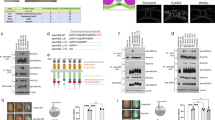
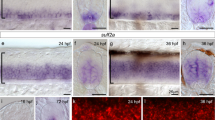
Sonic Hedgehog (Shh) is a secreted protein that controls cell fate and mitogenesis in the developing nervous system. Here we show that a constitutively active form of Smoothened (Smo-M2) mimics concentration-dependent actions of Shh in the developing neural tube, including activation of ventral marker genes (HNF3β, patched, Nkx2.2, netrin-1), suppression of dorsal markers (Pax-3, Gli-3, Ephrin A5) and induction of ventral neurons (dopaminergic, serotonergic) and ventrolateral motor neurons (Islet-1+, Islet-2+, HB9+) and interneurons (Engrailed-1+, CHX10+). Furthermore, Smo-M2's patterning activities were cell autonomous, occurring exclusively in cells expressing Smo-M2. These findings suggest that Smo is a key signaling component in the Hh receptor and that Shh patterns the vertebrate nervous system as a morphogen, rather than through secondary relay signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Belloni, E. et al. Identification of Sonic hedgehog as a candidate gene responsible for holoprosencephaly. Nat. Genet. 14, 353–356 (1996).
Roessler, E. et al. Mutations in the human Sonic Hedgehog gene cause holoprosencephaly. Nat. Genet. 14, 357–360 (1996).
Chiang, C. et al. Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature 382, 407–413 (1996).
Tanabe, Y. & Jessell, T. M. Diversity and pattern in the developing spinal cord. Science 274, 1115 –1123 (1996).
Hynes, M. et al. Control of cell pattern in the neural tube by the zinc finger transcription factor and oncogene Gli-1. Neuron 19, 15–26 (1997).
Ericson, J. et al. Sonic hedgehog induces the differentiation of ventral forebrain neurons: a common signal for ventral patterning within the neural tube. Cell 81, 747–756 ( 1995).
Ericson, J. et al. Pax6 controls progenitor cell identity and neuronal fate in response to graded Shh signaling. Cell 90, 169–180 (1997).
Goodrich, L. V., Johnson, R. L., Milenkovic, L., McMahon, J. A. & Scott, M. P. Conservation of the hedgehog/patched signaling pathway from flies to mice: induction of a mouse patched gene by Hedgehog. Genes Dev. 10, 301– 312 (1996).
Serafini, T. et al. The netrins define a family of axon outgrowth-promoting proteins homologous to C. elegans UNC-6. Cell 78, 409–424 (1994).
Kennedy, T. E., Serafini, T., de la Torre, J. R. & Tessier-Lavigne, M. Netrins are diffusible chemotropic factors for commissural axons in the embryonic spinal cord. Cell 78, 425– 435 (1994).
Yamada, T., Placzek, M., Tanaka, H., Dodd, J. & Jessell, T. M. Control of cell pattern in the developing nervous system: polarizing activity of the floor plate and notochord. Cell 64, 635–647 (1991).
Hynes, M. et al. Induction of midbrain dopaminergic neurons by Sonic Hedgehog. Neuron 80, 95–101 (1995).
Wang, M. Z. et al. Induction of dopaminergic neuron phenotype in the midbrain by Sonic hedgehog protein. Nat. Med. 1, 1184–1188 (1995).
Marigo, V., Johnson, R. L., Vortkamp, A. & Tabin, C. J. Sonic hedgehog differentially regulates expression of Gli and Gli3 during limb development. Dev. Biol. 180, 273– 283 (1996).
Stone, D. M. et al. The tumour-suppressor gene patched encodes a candidate receptor for Sonic hedgehog. Nature 384, 129– 134 (1996).
Alcedo, J., Ayzenzon, M., Von Ohlen, T., Noll, M. & Hooper, J. E. The Drosophila smoothened gene encodes a seven-pass membrane protein, a putative receptor for the hedgehog signal. Cell 86, 221–232 (1996).
van den Heuvel, M. & Ingham, P. W. smoothened encodes a receptor-like serpentine protein required for hedgehog signalling. Nature 382, 547–551 (1996).
Ingham, P. W., Taylor, A. M. & Nakano, Y. Role of the Drosophila patched gene in positional signalling. Nature 353, 184– 187 (1991).
Goodrich, L. V., Milenkovic, K. M., Higgins, L. & Scott, M. P. Altered cell fates and medulloblastoma in mouse patched mutants. Science 277, 1109–1113 ( 1997).
Chen, Y. & Struhl, G. In vivo evidence that Patched and Smoothened constitute distinct binding and transducing components of a Hedgehog receptor complex. Development 125, 4943–4948 (1998).
Johnson, R. L. et al. Human homolog of patched, a candidate gene for the basal cell nevus syndrome. Science 272, 1668–1671 (1996).
Xie, J. et al. Activating Smoothened mutations in sporadic basal-cell carcinoma. Nature 391, 90– 92 (1998).
Chen, Y. & Struhl, G. Dual roles for patched in sequestering and transducing Hedgehog. Cell 87, 553– 563 (1996).
Johnson, R. L., Grenier, J. K. & Scott, M. P. Patched overexpression alters wing disc size and pattern: transcriptional and post-transcriptional effects on hedgehog targets. Development 121, 4161– 4170 (1995).
Yang, Y. et al. Relationship between dose, distance and time in Sonic Hedgehog-mediated regulation of anteroposterior polarity in the chick limb. Development 124, 4393–4404 ( 1997).
Perrimon, N. Hedgehog and beyond. Cell 80, 517– 520 (1995).
Struhl, G., Barbash, D. A. & Lawrence, P. A. Hedgehog acts by distinct gradient and signal relay mechanisms to organise cell type and cell polarity in the Drosophila. Development 124, 2155–2165 (1997).
Heemskerk, J. & DiNardo, S. Drosophila hedgehog acts as a morphogen in cellular patterning. Cell 76, 449– 460 (1994).
Roelink, H. et al. Floor plate and motor neuron induction by different concentrations of the amino-terminal cleavage product of Sonic hedgehog autoproteolysis. Cell 81, 445–455 (1995).
Sasaki, H. & Hogan, B. L. M. HNF-3β as a regulator of floor plate development. Cell 76, 103 –115 (1994).
Murone, M., Rosenthal, A. & de Sauvage, F. J. Sonic hedgehog signaling by the patched-smoothened receptor complex. Curr. Biol. 9, 76– 84 (1999).
Echelard, Y. et al. Sonic hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell 75, 1417–1430 ( 1993).
Davis, C. A. & Joyner, A. L. Expression patterns of the homeobox containing genes En-1 and En-2 and the proto-oncogene int-1 diverge during mouse development. Genes Dev. 2, 1736–1744 (1988).
Davis, C. A., Noble-Topham, S. E., Rossant, J. & Joyner, A. L. Expression patterns of the homeobox containing gene en-2 delineates a specific region in the developing mouse brain. Genes Dev. 2, 361–371 (1988).
Tremblay, P., Pituello, F. & Gruss, P. Inhibition of floor plate differentiation by pax3: evidence from ectopic expression in transgenic mice. Development 122, 2555–2567 (1996).
Winslow, J. W. et al. Cloning of AL-1, a ligand for an Eph-related tyrosine kinase receptor involved in axon bundle formation. Neuron 14, 973–981 (1995).
Ruiz i Altaba, A. Combinatorial Gli gene function in floor plate and neuronal inductions by Sonic hedgehog. Development 125, 2203– 2212 (1998).
Marigo, V., Davey, R. A., Zuo, Y., Cunningham, J. M. & Tabin, C. J. Biochemical evidence that Patched is the Hedgehog receptor. Nature 384, 176– 179 (1996).
Marigo, V., Scott, M. P., Johnson, R. L., Goodrich, L. V. & Tabin, C. J. Conservation in hedgehog signaling: induction of a chicken patched homolog by Sonic hedgehog in the developing limb. Development 122, 1225– 1233 (1996).
Roelink, H. et al. Floor plate and motor neuron induction by vhh-1, a vertebrate homolog of hedgehog expressed by the notochord. Cell 76, 761–775 ( 1994).
Krauss, S., Concordet, J.-P. & Ingham, P. W. A functionally conserved homolog of the drosophila segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 75, 1431– 1444 (1993).
Riddle, R. D., Johnson, R. L., Laufer, E. & Tabin, C. Sonic hedgehog mediates the polarizing activity of the limb. Cell 75, 1401–1416 ( 1993).
Hynes, M., Poulsen, K., Tessier-Lavigne, M. & Rosenthal, A. Control of neuronal diversity by the floor plate: contact-mediated induction of midbrain dopaminergic neurons. Cell 80, 95–101 (1995).
Ye, W., Shimamura, K., Rubenstein, J. L. R., Hynes, M. A. & Rosenthal, A. FGF8 and Shh signals control dopaminergic and serotonergic cell fate in the anterior neural plate. Cell 93, 755–766 (1998).
Specht, L. A., Pickel, V. M., Joh, T. H. & Reis, D. J. Light-microscopic immunocytochemical localization of tyrosine hydroxylase in prenatal rat brain. II. Late ontogeny. J. Comp. Neurol. 199, 255–276 (1981).
Parent, A. in Serotonin Neurotransmission and Behavior (eds. Jacobs, B. L. & Gelperin, A.) 3–34 (MIT Press, Cambridge, Massachusetts, 1981).
Pfaff, S. & Kintner, C. Neuronal diversification: development of motor neuron subtypes. Curr. Opin. Neurobiol. 8, 27–36 (1998).
Tsuchida, T. et al. Topographic organization of embryonic motor neurons defined by expression of LIM homeobox genes. Cell 79, 957–970 (1994).
Thaler, J. et al. Active suppression of interneuron programs within developing motor neurons revealed by analysis of homeodomain factor HB9. Neuron 23, 659–674 ( 1999).
Sasaki, H., Hui, C.-C., Nakafuku, M. & Kondoh, H. A binding site of Gli proteins is essential for HNF-3β floor plate enhancer activity in transgenics and can respond to Shh in vitro. Development 124, 1313–1322 ( 1997).
Acknowledgements
We thank Kenji Shimamura for help with the electroporation, Susan Brenner-Morton and T. Jessell for the Islet-1, En-1, Chx-10 and HB9 antibodies and A. Ruiz i Altaba for the HNF-3β antiserum. We also thank E. Berry and A. Bruce for help with preparation of the manuscript and A. Shih and K. Poulsen for technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hynes, M., Ye, W., Wang, K. et al. The seven-transmembrane receptor Smoothened cell-autonomously induces multiple ventral cell types. Nat Neurosci 3, 41–46 (2000). https://doi.org/10.1038/71114
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/71114
This article is cited by
-
Regulation of stem cell fate by HSPGs: implication in hair follicle cycling
npj Regenerative Medicine (2022)
-
Shh-mediated centrosomal recruitment of PKA promotes symmetric proliferative neuroepithelial cell division
Nature Cell Biology (2017)
-
Effects of altered ephrin-A5 and EphA4/EphA7 expression on tumor growth in a medulloblastoma mouse model
Journal of Hematology & Oncology (2015)
-
Ptch1 and Gli regulate Shh signalling dynamics via multiple mechanisms
Nature Communications (2015)
-
Balanced Shh signaling is required for proper formation and maintenance of dorsal telencephalic midline structures
BMC Developmental Biology (2010)