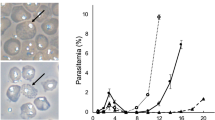
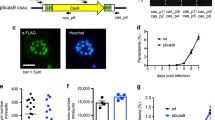
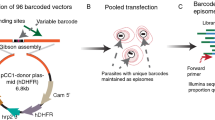
Abstract
Here we report an efficient, site-specific system of genetic integration into Plasmodium falciparum malaria parasite chromosomes. This is mediated by mycobacteriophage Bxb1 integrase, which catalyzes recombination between an incoming attP and a chromosomal attB site. We developed P. falciparum lines with the attB site integrated into the glutaredoxin-like cg6 gene. Transfection of these attB+ lines with a dual-plasmid system, expressing a transgene on an attP-containing plasmid together with a drug resistance gene and the integrase on a separate plasmid, produced recombinant parasites within 2 to 4 weeks that were genetically uniform for single-copy plasmid integration. Integrase-mediated recombination resulted in proper targeting of parasite proteins to intra-erythrocytic compartments, including the apicoplast, a plastid-like organelle. Recombinant attB × attP parasites were genetically stable in the absence of drug and were phenotypically homogeneous. This system can be exploited for rapid genetic integration and complementation analyses at any stage of the P. falciparum life cycle, and it illustrates the utility of Bxb1-based integrative recombination for genetic studies of intracellular eukaryotic organisms.
*Note: In the version of this article originally published figure 3 e-f appears in black and white due to a production error. The color version of this figure now has been substituted in the version of the paper available online. In addition, in the original version of figure 3 a, the pLN-ENR-GFP plasmid was incorrectly labeled with attB when the black box should indicate attP. The errors have been corrected in the HTML and PDF versions of the article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Change history
31 July 2006
In the version of this article originally published figure 3 e-f appears in black and white due to a production error. The color version of this figure now has been substituted in the version of the paper available online. In addition, in the original version of figure 3 a, the pLN-ENR-GFP plasmid was incorrectly labeled with attB when the black box should indicate attP. The errors have been corrected in the HTML and PDF versions of the article.
References
Crabb, B.S. et al. Transfection of the human malaria parasite Plasmodium falciparum. Methods Mol. Biol. 270, 263–276 (2004).
Groth, A.C. & Calos, M.P. Phage integrases: biology and applications. J. Mol. Biol. 335, 667–678 (2004).
Groth, A.C., Fish, M., Nusse, R. & Calos, M.P. Construction of transgenic Drosophila by using the site-specific integrase from phage phiC31. Genetics 166, 1775–1782 (2004).
Kim, A.I. et al. Mycobacteriophage Bxb1 integrates into the Mycobacterium smegmatis groEL1 gene. Mol. Microbiol. 50, 463–473 (2003).
Ghosh, P., Kim, A.I. & Hatfull, G.F. The orientation of mycobacteriophage Bxb1 integration is solely dependent on the central dinucleotide of attP and attB. Mol. Cell 12, 1101–1111 (2003).
Ghosh, P., Pannunzio, N.R. & Hatfull, G.F. Synapsis in phage Bxb1 integration: selection mechanism for the correct pair of recombination sites. J. Mol. Biol. 349, 331–348 (2005).
Su, X.-Z., Kirkman, L.S. & Wellems, T.E. Complex polymorphisms in a ∼330 kDa protein are linked to chloroquine-resistant P. falciparum in Southeast Asia and Africa. Cell 91, 593–603 (1997).
Mamoun, C.B., Gluzman, I.Y., Goyard, S., Beverley, S.M. & Goldberg, D.E. A set of independent selectable markers for transfection of the human malaria parasite Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 96, 8716–8720 (1999).
Wang, P., Wang, Q., Sims, P.F. & Hyde, J.E. Rapid positive selection of stable integrants following transfection of Plasmodium falciparum. Mol. Biochem. Parasitol. 123, 1–10 (2002).
Ralph, S.A. et al. Tropical infectious diseases: metabolic maps and functions of the Plasmodium falciparum apicoplast. Nat. Rev. Microbiol. 2, 203–216 (2004).
O'Donnell, R.A. et al. A genetic screen for improved plasmid segregation reveals a role for Rep20 in the interaction of Plasmodium falciparum chromosomes. EMBO J. 21, 1231–1239 (2002).
Franke-Fayard, B. et al. A Plasmodium berghei reference line that constitutively expresses GFP at a high level throughout the complete life cycle. Mol. Biochem. Parasitol. 137, 23–33 (2004).
Goodyer, I.D. & Taraschi, T.F. Plasmodium falciparum: a simple, rapid method for detecting parasite clones in microtiter plates. Exp. Parasitol. 86, 158–160 (1997).
Balu, B., Shoue, D.A., Fraser, M.J., Jr. & Adams, J.H. High-efficiency transformation of Plasmodium falciparum by the lepidopteran transposable element piggyBac. Proc. Natl. Acad. Sci. USA 102, 16391–16396 (2005).
Sakamoto, H. et al. Towards systematic identification of Plasmodium essential genes by transposon shuttle mutagenesis. Nucleic Acids Res. 33, e174 (2005).
Thorpe, H.M., Wilson, S.E. & Smith, M.C. Control of directionality in the site-specific recombination system of the Streptomyces phage phiC31. Mol. Microbiol. 38, 232–241 (2000).
Thomason, L.C., Calendar, R. & Ow, D.W. Gene insertion and replacement in Schizosaccharomyces pombe mediated by the Streptomyces bacteriophage phiC31 site-specific recombination system. Mol. Genet. Genomics 265, 1031–1038 (2001).
Ghosh, P., Wasil, L.R. & Hatfull, G.F. Control of phage Bxb1 excision by a novel recombination directionality factor. PLoS Biol. 4, e186 (2006).
Branda, C.S. & Dymecki, S.M. Talking about a revolution: The impact of site-specific recombinases on genetic analyses in mice. Dev. Cell 6, 7–28 (2004).
Carvalho, T.G., Thiberge, S., Sakamoto, H. & Menard, R. Conditional mutagenesis using site-specific recombination in Plasmodium berghei. Proc. Natl. Acad. Sci. USA 101, 14931–14936 (2004).
Fidock, D.A. & Wellems, T.E. Transformation with human dihydrofolate reductase renders malaria parasites insensitive to WR99210 but does not affect the intrinsic activity of proguanil. Proc. Natl. Acad. Sci. USA 94, 10931–10936 (1997).
Lobo, C.A., Fujioka, H., Aikawa, M. & Kumar, N. Disruption of the Pfg27 locus by homologous recombination leads to loss of the sexual phenotype in P. falciparum. Mol. Cell 3, 793–798 (1999).
Walliker, D. et al. Genetic analysis of the human malaria parasite Plasmodium falciparum. Science 236, 1661–1666 (1987).
Coppel, R.L. & Black, C.G. in Malaria: Parasite Biology, Pathogenesis, and Protection (ed. I.W. Sherman) 185–202 (American Society for Microbiology Press, Washington, D.C.; 1998).
Gardner, M.J. et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 419, 498–511 (2002).
Roberts, D.J. et al. Rapid switching to multiple antigenic and adhesive phenotypes in malaria. Nature 357, 689–692 (1992).
Meissner, M. et al. Tetracycline analogue-regulated transgene expression in Plasmodium falciparum blood stages using Toxoplasma gondii transactivators. Proc. Natl. Acad. Sci. USA 102, 2980–2985 (2005).
Fidock, D.A., Nomura, T. & Wellems, T.E. Cycloguanil and its parent compound proguanil demonstrate distinct activities against Plasmodium falciparum malaria parasites transformed with human dihydrofolate reductase. Mol. Pharmacol. 54, 1140–1147 (1998).
Sidhu, A.B.S., Valderramos, S.G. & Fidock, D.A. pfmdr1 mutations contribute to quinine resistance and enhance mefloquine and artemisinin sensitivity in Plasmodium falciparum. Mol. Microbiol. 57, 913–926 (2005).
Elmendorf, H.G. & Haldar, K. Plasmodium falciparum exports the Golgi marker sphingomyelin synthase into a tubovesicular network in the cytoplasm of mature erythrocytes. J. Cell Biol. 124, 449–462 (1994).
Tonkin, C.J. et al. Localization of organellar proteins in Plasmodium falciparum using a novel set of transfection vectors and a new immunofluorescence fixation method. Mol. Biochem. Parasitol. 137, 13–21 (2004).
Waller, R.F., Reed, M.B., Cowman, A.F. & McFadden, G.I. Protein trafficking to the plastid of Plasmodium falciparum is via the secretory pathway. EMBO J. 19, 1794–1802 (2000).
Acknowledgements
We thank P. Wang and J. Hyde (University of Manchester, UK) for their gift of the pPKDSneoII plasmid and G. McFadden (University of Melbourne, Australia) for his kind gift of the antibodies to ACP. We also gratefully acknowledge the Burroughs Wellcome Fund for providing a New Initiatives in Malaria Research Award (to W.R.J. and D.A.F.) that provided funding for this work, as well as US National Institutes of Health grants AI59114 to G.F.H. and AI060342 to D.A.F. and W.R.J. (P01; principal investigator, James C. Sacchettini).
Author information
Authors and Affiliations
Contributions
L.J.N., R.A.M., P.A.M., W.R.J. and D.A.F. designed the experiments, which were performed by L.J.N., R.A.M. and P.A.M. P.G. and G.F.H. provided integrase reagents and W.R.J., G.F.H. and D.A.F. conceived of the plan to test Bxb1 integrase in P. falciparum. All authors contributed to the manuscript, which was written primarily by L.J.N., R.A.M. and D.A.F. L.J.N. was co-mentored by D.A.F. and W.R.J.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Maps of P. falciparum plasmids used in the development of the Bxb1 integrase-mediated system of site-specific intergration. (PDF 490 kb)
Supplementary Fig. 2
Integration of the pCG6-attB plasmid into the hrp3 promoter by homologous recombination. (PDF 587 kb)
Supplementary Fig. 3
Stability of an attB x attP intergrated transgene over time in the absence of drug pressure, versus rapid loss of episomes from unselected parasites. (PDF 662 kb)
Supplementary Fig. 4
Plasmids greater than 10 kb in size stably intergrate into attB parasites. (PDF 950 kb)
Supplementary Table 1
Oligonucleotides. (PDF 20 kb)
Rights and permissions
About this article
Cite this article
Nkrumah, L., Muhle, R., Moura, P. et al. Efficient site-specific integration in Plasmodium falciparum chromosomes mediated by mycobacteriophage Bxb1 integrase. Nat Methods 3, 615–621 (2006). https://doi.org/10.1038/nmeth904
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth904
This article is cited by
-
Model-guided engineering of DNA sequences with predictable site-specific recombination rates
Nature Communications (2022)
-
Gametocyte-specific and all-blood-stage transmission-blocking chemotypes discovered from high throughput screening on Plasmodium falciparum gametocytes
Communications Biology (2022)
-
Maturation and substrate processing topography of the Plasmodium falciparum invasion/egress protease plasmepsin X
Nature Communications (2022)
-
Selective expression of variant surface antigens enables Plasmodium falciparum to evade immune clearance in vivo
Nature Communications (2022)
-
CRISPR/Cas9-engineered inducible gametocyte producer lines as a valuable tool for Plasmodium falciparum malaria transmission research
Nature Communications (2021)