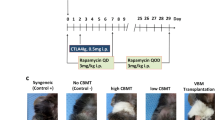
Abstract
Specific T–cell tolerance may be essential for successful xenotransplantation in humans. Grafting of thymectomized, T cell–depleted normal mice with xenogeneic fetal pig thymus and liver (FP THY/LIV) tissue results in the recovery of functional CD4 antigen–positive cells. We have tested T–cell tolerance by skin grafting. Donor–matched pig skin survived permanently (>200 days), whereas allogeneic mouse skin was rapidly rejected. Nontolerant control mice rejected pig skin within 26 days. Both porcine and murine histocompatibility class IIhigh cells were detected in long–term thymus grafts, and T–cell repertoire analyses suggested that tolerance to both donors and recipients developed, at least in part, by intragraft clonal deletion. This study demonstrates the principle that tolerance, measured by the stringent criterion of skin grafting, can be induced across a widely disparate species barrier.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sykes, M., Lee, L.A. & Sachs, D.H. Xenograft tolerance. Immunol. Rev. 141, 245–276 (1994).
Sachs, D.H., Sykes, M., Greenstein, J.L. & Cosimi, A.B. Tolerance and xenograft survival. Nature Med. 1, 969 (1995).
Lunney, J.K. Characterization of swine leukocyte differentiation antigens. Immunol. Today 14, 147–148 (1993).
Lee, L.A. et al. Specific tolerance across a discordant xenogeneic transplantation barrier. Proc. Natl. Acad. Sci. USA 91, 10864–10867 (1994).
Yang, Y.-G., Ohta, S., Yamada, S., Shimizu, M. & Takagaki, Y. Diversity of T cell receptor δ-chain cDNA in the thymus of a one-month-old pig. J. Immunol. 155, 1981–1993 (1995).
Sykes, M. Inducing specific tolerance across xenogeneic barrier. Xeno 2, 65–67 (1994).
Auchincloss, H.A. Why is cell-mediated xenograft rejection so strong? Xeno 3, 19–22 (1995).
Yamada, K., Sachs, D.H. & DerSimonian, H. Human anti-porcine xenogeneic T cell response: Evidence for allelic specificity of mixed leukocyte reaction and for both direct and indirect pathways of recognition. J. Immunol. 155, 5249–5256 (1995).
Moses, R.D., Winn, H.J. & Auchincloss, H. Jr., Evidence that multiple defects in cell-surface molecule interactions across species differences are responsible for diminished xenogeneic T cell responses. Transplantation 53, 203–209 (1992).
Ramsdell, F. & Fowlkes, B.J. Clonal deletion versus clonal anergy: The role of the thymus in inducing self tolerance. Science 248, 1342–1348 (1990).
Kappler, J.W., Roehm, N. & Marrack, P. T cell tolerance by clonal elimination in the thymus. Cell 49, 273–280 (1987).
Burkly, L.C., Lo, D., Kanagawa, O., Brinster, R.l. & Flavell, R.A. T-cell tolerance by clonal anergy in transgenic mice with nonlymphoid expression of MHC class II I–E. Nature 342, 564 (1989).
Webb, S., Morris, C. & Sprent, J. Extrathymic tolerance of mature T cells: Clonal elimination as a consequence of immunity. Cell 63, 1249–1256 (1990).
Yin, D. & Fathman, C.G. CD4-positive suppressor cells block allotransplant rejection. J. Immunol. 154, 6339–6345 (1995).
Davies, J.D., Leong, L.Y.W., Mellor, A., Cobbold, S.P. & Waldmann, H. T cell suppression in transplantation tolerance through linked recognition. J. Immunol. 156, 3602–3607 (1996).
Qin, S. et al. “Infectious” transplantation tolerance. Science 259, 974–977 (1993).
Sykes, M. Chimerism and central tolerance. Curr. Opin. Immunol. (in the press).
Tomonari, K., Fairchild, S. & Rosenwasser, O.A. Influence of viral superantigens on Vβ-and Vα-specific positive and negative selection. Immunol. Rev. 131, 131–168 (1993).
Tomonari, K. & Fairchild, S. The genetic basis of negative selection of TcrβV11+ T cells. Immunogenctics 33, 157–162 (1991).
Dyson, P.J., Knight, A.M., Fairchild, S., Simpson, E. & Tomonari, K. Genes encoding ligands for deletion of Vβ11 T cells cosegregate with mammary tumour virus genomes. Nature 349, 531–532 (1991).
Lenschow, D.J. et al. Long-term survival of xenogeneic pancreatic islet grafts induced by CTLA4Ig. Science 257, 789–792 (1992).
Lafferty, K.J. A contemporary view of transplantation tolerance: An immunologist's perspective. Clin. Transplant. 8, 181–187 (1994).
Weiens, P.W. et al. Tissue-specific differences in the establishment of tolerance. Transplantation 57, 1795–1798 (1994).
Tomita, Y., Khan, A. & Sykes, M. Role of intrathymic clonal deletion and peripheral anergy in transplantation tolerance induced by bone marrow transplantation in mice conditioned with a nonmyeloablative regimen. J. Immunol. 153, 1087–1098 (1994).
Tomita, Y., Lee, L.A. & Sykes, M. Engraftment of rat bone marrow and its role in negative selection of murine T cells in mice conditioned with a modified non-myeloablative regimen. Xenotransplant 1, 109–117 (1994).
Grusby, M.J. & Glimcher, L.H. Immune responses in MHC class II-deficient mice. Ann. Rev. Immunol. 13, 417–435 (1995).
Grusby, M.J. et al. Mice lacking major histocompatibility complex class I and class II molecules. Proc. Natl. Acad. Sci. USA 90, 3913–3917 (1993).
Modiglani, Y. et al. Lymphocytes selected in allogeneic thymic epithelium mediate dominant tolerance toward tissue grafts of the thymic epithelium haplotype. Proc. Natl. Acad. Sci. USA 92, 7555–7559 (1995).
Salaun, J. et al. Thymic epithelium tolerizes for histocompatibility antigens. Science 247, 1471–1474 (1990).
Ohki, H., Martin, C., Corbel, C. & Le Douarin, N.M. Tolerance induced by thymic epithelial grafts in birds. Science 237, 1032–1035 (1987).
Martin, C., Ohki-Hamazaki, H., Corbel, C., Coltey, M. & Le Douarin, N.M. Successful xenogeneic transplantation in embryos: induction of tolerance by extrathymic chick tissue grafted into quail. Dev. Immunol. 1, 265–277 (1991).
Salaun, J. et al. Transplantation tolerance is unrelated to superantigendependent deletion and anergy. Proc. Natl. Acad. Sci. USA 89, 10420–10424 (1992).
Houssaint, E. & Flajnik, M. The role of thymic epithelium in the acquisition of tolerance. Immunol. Today 11, 357–360 (1990).
Speiser, D.E. et al. Clonal deletion induced by either radioresistant thymic host cells or lymphohematopoietic donor cells at different stages of class I-restricted T cell ontogeny. J. Exp. Med. 175, 1277–1283 (1992).
Lorenz, R.G. & Allen, P.M. Thymic cortical epithelial cells lack full capacity for antigen presentation. Nature 340, 557–559 (1989).
Jenkinson, E.J., Anderson, G. & Owen, J.J.T. Studies on T cell maturation on defined thymic stromal cell populations in vitro. J. Exp. Med. 176, 845–853 (1992).
Guery, J-C. & Adorini, L. Dendritic cells are the most efficient in presenting endogenous naturally processed self-epitopes to class II-restricted T cells. J. Immunol. 154, 536–544 (1995).
Kyewski, B.A., Fathman, C.G. & Kaplan, H.S. Intrathymic presentation of circulating non-major histocompatibility complex antigens. Nature 308, 196–199 (1984).
Miller, J.F.A.P. Studies on mouse leukaemia: The role of the thymus in leukaemogenesis by cell-free leukemic infiltrates. Br. J. Cancer 14, 93 (1960).
Sykes, M. et al. Hematopoietic cells and radioresistant host elements influence natural killer cell differentiation. J. Exp. Med. 178, 223–229 (1993).
Sharabi, Y., Aksentijevich, I., Sundt, T.M. III, Sachs, D.H. & Sykes, M. Specific tolerance induction across a xenogeneic barrier: Production of mixed rat/mouse lymphohematopoietic chimeras using a nonlethal preparative regimen. J. Exp. Med. 172, 195–202 (1990).
Ildstad, S.T., Wren, S.M., Bluestone, J.A., Barbieri, S.A. & Sachs, D.H. Characterization of mixed allogeneic chimeras: Immunocompetence, in vitro reactivity, and genetic specificity of tolerance. J. Exp. Med. 162, 231–244 (1985).
Ozato, K., Mayer, N. & Sachs, D.H. Hybridoma cell lines secreting monoclonal antibodies to mouse H–2 and Ia antigens. J. Immunol. 124, 533–540 (1980).
Sachs, D.H., Mayer, N. & Ozato, K. Hybridoma antibodies directed toward murine H–2 and Ia antigens. in Monoclonal Antibodies and T Cell Hybridomas. (eds. Hammerling, G.J., Hammerling, U. & Kearney, J.F.) 95–101 (Elsevier/North-Holland Biomedical, New York, 1981).
Ozato, K. & Sachs, D.H. Monoclonal antibodies to mouse MHC antigens. III. Hybridoma antibodies reacting to antigens of the H–2b haplotype reveal genetic control of isotype expression. J. Immunol. 126, 317–321 (1981).
Watanabe, M., Suzuki, T., Taniguchi, M. & Shinohara, N. Monoclonal anti-Ia murine alloantibodies crossreactive with the Ia-homologues of other mammalian species including humans. Transplantation 36, 712–718 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhao, Y., Swenson, K., Sergio, J. et al. Skin graft tolerance across a discordant xenogeneic barrier. Nat Med 2, 1211–1216 (1996). https://doi.org/10.1038/nm1196-1211
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1196-1211
This article is cited by
-
Progress in xenotransplantation: overcoming immune barriers
Nature Reviews Nephrology (2022)
-
Extensive germline genome engineering in pigs
Nature Biomedical Engineering (2020)
-
Transplant Tolerance: Current Insights and Strategies for Long-Term Survival of Xenografts
Archivum Immunologiae et Therapiae Experimentalis (2018)
-
Xenotransplantation: Where Are We with Potential Kidney Recipients? Recent Progress and Potential Future Clinical Trials
Current Transplantation Reports (2017)
-
2-Gy whole-body irradiation significantly alters the balance of CD4+CD25−T effector cells and CD4+CD25+Foxp3+T regulatory cells in mice
Cellular & Molecular Immunology (2010)