Abstract
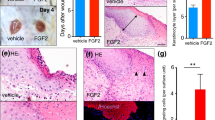
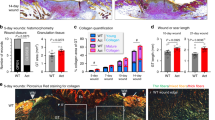
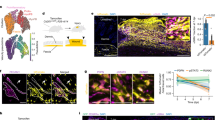
The upregulation of TGF-β1 and integrin expression during wound healing has implicated these molecules in this process, but their precise regulation and roles remain unclear. Here we report that, notably, mice lacking β3-integrins show enhanced wound healing with re-epithelialization complete several days earlier than in wild-type mice. We show that this effect is the result of an increase in TGF-β1 and enhanced dermal fibroblast infiltration into wounds of β3-null mice. Specifically, β3-integrin deficiency is associated with elevated TGF-β receptor I and receptor II expression, reduced Smad3 levels, sustained Smad2 and Smad4 nuclear localization and enhanced TGF-β1-mediated dermal fibroblast migration. These data indicate that αvβ3–integrin can suppress TGF-β1-mediated signaling, thereby controlling the rate of wound healing, and highlight a new mechanism for TGF-β1 regulation by β3-integrins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Clark, R.A.F. Wound repair (overview and general conditions). in The Molecular and Cellular Biology of Wound Repair 2nd edn (ed. Clark, R.A.F.) (Plenum, New York, 1995).
Blobe, G.C., Schiemann, W.P. & Lodish, H.F. Mechanisms of disease: role of transforming growth factor β in human disease. N. Engl. J. Med. 342, 1350–1358 (2000).
Massague, J. How cells read TGF-β signals. Nat. Rev. Mol. Cell Biol. 1, 169–178 (2000).
Martin, P. Wound healing—aiming for perfect skin regeneration. Science 276, 75–81 (1997).
Hebda, P.A. Stimulatory effects of transforming growth factor-beta and epidermal growth-factor on epidermal-cell outgrowth from porcine skin explant cultures. J. Invest. Dermatol. 91, 440–445 (1988).
Wakefield, L. TGF-β signaling: positive and negative effects on tumorigenesis. Curr. Opin. Genet. Dev. 12, 22–29 (2002).
Shah, M., Foreman, D.M. & Ferguson, M.W. Neutralising antibody to TGF-β 1,2 reduces cutaneous scarring in adult rodents. J. Cell Sci. 107, 1137–1157 (1994).
Cordeiro, M.F. Transforming growth factor-β function blocking already effective as therapeutic strategy. Circulation 107, E37–E37 (2003).
Shah, M., Foreman, D.M. & Ferguson, M.W.J. Neutralization of Tgf-β 1 and Tgf-β 2 or exogenous addition of Tgf-β 3 to cutaneous rat wounds reduces scarring. J. Cell Sci. 108, 985–1002 (1995).
Amendt, C., Mann, A., Schirmacher, P. & Blessing, M. Resistance of keratinocytes to TGF β-mediated growth restriction and apoptosis induction accelerates re- epithelialization in skin wounds. J. Cell Sci. 115, 2189–2198 (2002).
Leibovich, S.J. & Ross, R. The role of the macrophage in wound repair. A study with hydrocortisone and antimacrophage serum. Am. J. Pathol. 78, 71–100 (1975).
Gailit, J., Welch, M.P. & Clark, R.A.F. Tgf-β1 stimulates expression of keratinocyte integrins during reepithelialization of cutaneous wounds. J. Invest. Dermatol. 103, 221–227 (1994).
Zambruno, G. et al. Transforming growth factor-β1 modulates β1 and β5 integrin receptors and induces the de novo expression of the αvβ6 heterodimer in normal human keratinocytes—implications for wound-healing. J. Cell Biol. 129, 853–865 (1995).
Sellheyer, K. et al. Inhibition of skin development by overexpression of transforming growth factor β1 in the epidermis of transgenic mice. Proc. Natl. Acad. Sci. USA 90, 5237–5241 (1993).
Yang, L. et al. Healing of burn wounds in transgenic mice overexpressing transforming growth factor-β1 in the epidermis. Am. J. Pathol. 159, 2147–2157 (2001).
Garlick, J.A. & Taichman, L.B. Fate of human keratinocytes during reepithelialization in an organotypic culture model. Lab. Invest. 70, 916–924 (1994).
Garlick, J.A. & Taichman, L.B. Effect of Tgf-β1 on reepithilialization of human keratinocytes in vitro–an organotypic model. J. Invest. Dermatol. 103, 554–559 (1994).
Crowe, M.J., Doetschman, T. & Greenhalgh, D.G. Delayed wound healing in immunodeficient TGF-β1 knockout mice. J. Invest. Dermatol. 115, 3–11 (2000).
Shah, M. et al. Role of elevated plasma transforming growth factor-β1 levels in wound healing. Am. J. Pathol. 154, 1115–1124 (1999).
Larjava, H., Salo, T., Haapasalmi, K., Kramer, R.H. & Heino, J. Expression of integrins and basement-membrane components by wound keratinocytes. J. Clin. Invest. 92, 1425–1435 (1993).
Greiling, D. & Clark, R.A.F. Fibronectin provides a conduit for fibroblast transmigration from collagenous stroma into fibrin clot provisional matrix. J. Cell Sci. 110, 861–870 (1997).
Shattil, S.J. Function and regulation of the β3 integrins in hemostasis and vascular biology. Thromb. Haemost. 74, 149–155 (1995).
Leavesley, D.I., Schwartz, M.A., Rosenfeld, M. & Cheresh, D.A. Integrin β 1-mediated and β3-mediated endothelial-cell migration is triggered through distinct signaling mechanisms. J. Cell Biol. 121, 163–170 (1993).
Stefansson, S. & Lawrence, D.A. The serpin PAI-1 inhibits cell migration by blocking integrin αvβ3 binding to vitronectin. Nature 383, 441–443 (1996).
Clark, R. Tonnesen, M.G., Gailit, J. & Cheresh, D.A. Transient functional expression of αvβ3 on vascular cells during wound repair. Am. J. Pathol. 148, 1407–1421 (1996).
Huang, X.Z. et al. Inactivation of the integrin β6 subunit gene reveals a role of epithelial integrins in regulating inflammation in the lung and skin. J. Cell Biol. 133, 921–928 (1996).
Huang, X., Griffiths, M., Wu, J., Farese, R.V.J. & Sheppard, D. Normal development, wound healing, and adenovirus susceptibility in β5-deficient mice. Mol. Cell. Biol. 20, 755–759 (2000).
Reynolds, L. et al. Enhanced pathological angiogenesis in mice lacking β3 integrin or β3 and β5 integrins. Nat. Med. 8, 27–34 (2002).
Hodivala-Dilke, K. et al. β3-integrin-deficient mice are a model for Glanzmann thrombasthenia showing placental defects and reduced survival. J. Clin. Invest. 103, 229–238 (1999).
Basson, C.T., Kocher, O., Basson, M.D., Asis, A. & Madri, J.A. Differential modulation of vascular cell integrin and extracellular-matrix expression in vitro by Tgf-β1 correlates with reciprocal effects on cell-migration. J. Cell. Physiol. 153, 118–128 (1992).
Ignotz, R.A. & Massague, J. Transforming growth factor-β stimulates the expression of fibronectin and collagen and their incorporation into the extracellular matrix. J. Biol. Chem. 261, 4337–4345 (1986).
Inman, G.J., Nicolas, F.J. & Hill, C.S. Nucleocytoplasmic shuttling of Smads 2, 3, and 4 permits sensing of TGF-β receptor activity. Mol. Cell 10, 283–294 (2002).
Grose, R. et al. A crucial role of β1 integrins for keratinocyte migration in vitro and during cutaneous wound repair. Development 129, 2303–2315 (2002).
Liaw, L. et al. Altered wound healing in mice lacking a functional osteopontin gene. J. Clin. Invest. 101, 1468–1478 (1998).
Quaglino, D., Jr, Nanney, L.B., Ditesheim, J.A. & Davidson, J.M. Transforming growth factor-β stimulates wound healing and modulates extracellular matrix gene expression in pig skin: incisional wound model. J. Invest. Dermatol. 97, 34–42 (1991).
Ashcroft, G. et al. Mice lacking Smad3 show accelerated wound healing and an impaired local inflammatory response. Nat. Cell Biol. 1, 260–266 (1999).
Seoane, J., Le, H.V., Shen, L., Anderson, S.A. & Massague, J. Integration of Smad and forkhead pathways in the control of neuroepithelial and glioblastoma cell proliferation. Cell 117, 211–223 (2004).
Reynolds, A.R. et al. Elevated Flk1 (Vascular endothelial growth factor receptor 2) signaling mediates enhanced angiogenesis in β3-integrin-deficient mice. Cancer Res. 64, 8643–8650 (2004).
Jang, Y.C., Arumugam, S., Gibran, N.S. & Isik, F.F. Role of αv integrins and angiogenesis during wound repair. Wound Repair Regen. 7, 375–380 (1999).
Wahl, S.M. et al. Transforming growth-factor type-β induces monocyte chemotaxis and growth-factor production. Proc. Natl. Acad. Sci. USA 84, 5788–5792 (1987).
Martin, P. et al. Wound healing in the PU.1 null mouse—tissue repair is not dependent on inflammatory cells. Curr. Biol. 13, 1122–1128 (2003).
Direkze, N.C. et al. Multiple organ engraftment by bone-marrow-derived myofibroblasts and fibroblasts in bone-marrow-tranplanted mice. Stem Cells 21, 514–520 (2003).
Stupack, D., Puente, X., Boutsaboualoy, S., Storgard, C.M. & Cheresh, D.A. Apoptosis of adherent cells by recruitment of caspase-8 to unligated integrins. J. Cell Biol. 155, 459–470 (2001).
Dennler, S., Huet, S. & Gauthier, J.M. A short amino-acid sequence in MH1 domain is responsible for functional differences between Smad2 and Smad3. Oncogene 18, 1643–1648 (1999).
Yanagisawa, K. et al. Induction of apoptosis by Smad3 and down-regulation of Smad3 expression in response to TGF-β in human normal lung epithelial cells. Oncogene 17, 1743–1747 (1998).
Lai, C.F. et al. Transforming growth factor-β up-regulates the β 5 integrin subunit expression via Sp1 and Smad signaling. J. Biol. Chem. 275, 36400–36406 (2000).
Munger, J.S. et al. The integrin αvβ6 binds and activates latent TGFβ1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96, 319–328 (1999).
Diaz-Gonzalez, F., Forsyth, J., Steiner, B. & Ginsberg, M.H. Trans-dominant inhibition of integrin function. Mol. Biol. Cell 7, 1939–1951 (1996).
Owens, D.M. Romero M.R., Gardner, C. & Watt FM. Suprabasal α6β4 integrin expression in epidermis results in enhanced tumourigenesis and disruption of TGFβ signalling. J. Cell Sci. 116, 3783–3791 (2003).
DiPersio, C.M., Shah, S. & Hynes, R.O. α3Aβ1 integrin localizes to focal contacts in response to diverse extracellular matrix proteins. J. Cell Sci. 108, 2321–2336 (1995).
Acknowledgements
We thank C. Hill, G. Inman F. Parkinson, I. Hart, F. Watt, D. Owens and A. Daley for their helpful advice and criticisms throughout the work; S. Watling and C. Wren for their technical assistance; G. Elias and colleagues for help with histology; F. Balkwill's laboratory for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Accelerated re-epithelialization is associated with enhanced epithelial migration. (PDF 60 kb)
Supplementary Fig. 2
Wound contraction is normal in β3-null wounds. (PDF 59 kb)
Supplementary Fig. 3
Sense control for in situ hybridization for Tgfb1 mRNA. (PDF 117 kb)
Supplementary Fig. 4
Negative control for TGF-β1 and P-smad2 immunostaining. (PDF 83 kb)
Supplementary Fig. 5
Keratinocyte proliferation is decreased, but dermal ECM deposition is increased in β3-integrin-deficient wounds. (PDF 663 kb)
Supplementary Fig. 6
Neutrophil numbers are not changed in β3-null wounds. (PDF 106 kb)
Rights and permissions
About this article
Cite this article
Reynolds, L., Conti, F., Lucas, M. et al. Accelerated re-epithelialization in β3-integrin-deficient- mice is associated with enhanced TGF-β1 signaling. Nat Med 11, 167–174 (2005). https://doi.org/10.1038/nm1165
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1165
This article is cited by
-
The role of antifreeze genes in the tolerance of cold stress in the Nile tilapia (Oreochromis niloticus)
BMC Genomics (2023)
-
The role of integrins in inflammation and angiogenesis
Pediatric Research (2021)
-
Sodium alginate/collagen hydrogel loaded with human umbilical cord mesenchymal stem cells promotes wound healing and skin remodeling
Cell and Tissue Research (2021)
-
Differential regulations of fibronectin and laminin in Smad2 activation in vascular endothelial cells in response to disturbed flow
Journal of Biomedical Science (2018)
-
The role of integrins in TGFβ activation in the tumour stroma
Cell and Tissue Research (2016)