Abstract
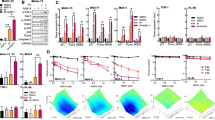
The induction of tumor cell death by anticancer therapy results from a genetic program of autonomous cell death termed apoptosis. Because the p53 tumor suppressor gene is a critical component for induction of apoptosis in response to DNA damage, its inactivation in cancers may be responsible for their resistance to genotoxic anticancer agents. The cellular response to DNA damage involves a cell–cycle arrest at both the G1/S and G2/M transitions; these checkpoints maintain viability by preventing the replication or segregation of damaged DNA. The arrest at the G1 checkpoint is mediated by p53–dependent induction of p21WAFI/ClP1, whereas the G2 arrest involves inactivation of p34cdc2 kinase. Following DNA damage, p53–deficient cells fail to arrest at G1 and accumulate at the G2/M transition. We demonstrate that abrogation of G2 arrest by caffeine–mediated activation of p34cdc2 kinase results in the selective sensitization of p53–deficient primary and tumor cells to irradiation–induced apoptosis. These data suggest that pharmacologic activation of p34cdc2 kinase may be a useful therapeutic strategy for circumventing the resistance of p53–deficient cancers to genotoxic anticancer agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Thompson, C.B. Apoptosis in the pathogenesis and treatment of disease. Science 267, 1456–1462 (1995).
Steller, H. Mechanisms and genes of cellular suicide. Science 267, 1445–1449 (1995).
Fisher, D.E. Apoptosis in cancer therapy: Crossing the threshold. Cell 78, 539–542 (1994).
Lowe, S.W., Ruley, H.E., Jacks, T. & Housman, D. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74, 957–967 (1993).
Clark, A.R. et al. Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 362, 849–852 (1993).
Lowe, S.W., Schmitt, E.M., Smith, S.W., Osborne, B. & Jacks, T. p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362, 847–849 (1993).
Symonds, H. et al. p53-dependent apoptosis suppresses tumor growth and progression in vivo. Cell 78, 703–711 (1994).
Lowe, S.W. et al. p53 status and the efficacy of cancer therapy in vivo. Science 266, 807–810 (1994).
Kinzler, K.W. & Vogelstein, B. Clinical implications of basic research: Cancer therapy meets p53. N. Engl. J. Med. 331, 49–50 (1994).
Dulic, V. et al. p53-dependent inhibition of cyclin-dependent kinase activities in human fibroblasts during radiation-induced G1 arrest. Cell 76, 1013–1023 (1994).
El-Deiry, W.S. et al. WAF1, a potential mediator of p53 tumor suppression. Cell 75, 817–825 (1993).
Kuerbitz, S.J., Plunkett, B.S., Walsh, W.V. & Kastan, M.B. Wild-type p53 is a cell cycle checkpoint determinant following irradiation. Proc. Natl. Acad. Sci. USA 89, 7491–7495 (1992).
Smith, M.L. et al. Interaction of the p53-regulated protein Gadd45 with proliferating cell nuclear antigen. Science 266, 1376–1380 (1994).
Nurse, P. Universal control mechanism regulating onset of M-phase. Nature 344, 503–507 (1990).
Nurse, P. Ordering S phase and M phase in the cell cycle. Cell 79, 547–550 (1994).
Hartwell, L.H. & Weinert, T.A. Controls that ensure the order of cell cycle events. Science 246, 629–634 (1989).
Murray, A.W. Creative blocks: Cell-cycle checkpoints and feedback controls. Nature 359, 599–604 (1992).
Lotem, J. & Sachs, L. Hematopoietic cells from mice deficient in wild-type p53 are more resistant to induction of apoptosis by some agents. Blood 82, 1092–1096 (1993).
Hunter, T. Protein kinases and phosphatases: The yin and yang of protein phosphorylation and signaling. Cell 80, 225–236 (1995).
Lock, R.B., Galperina, O.V., Feldhoff, R.C. & Rhodes, L.J. Concentration-dependent differences in the mechanisms by which caffeine potentiates etoposide cytotoxicity in HeLa cells. Cancer Res. 54, 4933–4939 (1994).
Michalovitz, D., Halevy, O. & Oren, M. Conditional inhibition of transformation and of cell proliferation by a temperature-sensitive mutant of p53. Cell 62, 671–680 (1990).
Harbour, J.W. et al. Abnormalities in structure and expression of the human retinoblastoma gene in SCLC. Science 241, 353–357 (1988).
Powell, S.N. et al. Differential sensitivity of p53(−/−) and p53(+/+) cells to caffeine-induced radiosensitization and override of G2 delay. Cancer Res. 55, 1643–1648 (1995).
Russell, K.J. et al. Abrogation of the G2 checkpoint results in differential radiosensitization of G1 checkpoint-deficient and G1 checkpoint-competent cells. Cancer Res. 55, 1639–1642 (1995).
Fan, S. et al. Disruption of p53 function sensitizes breast cancer MCF-7 cells to cisplatin and pentoxifylline. Cancer Res. 55, 1649–1654 (1995).
Donehower, L.A. et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356, 215–221 (1992).
Bedi, A. et al. BCR-ABL-mediated inhibition of apoptosis with delay of G2/M transition following DNA damage: A mechanism of resistance to multiple anticancer agents. Blood 86, 1148–1158 (1995).
Th'ng, J.P.H. et al. The FT210 cell line is a mouse G2 phase mutant with a temperature-sensitive CDC2 gene product. Cell 63, 313–324 (1990).
Kharbanda, S. et al. Ionizing radiation induces rapid tyrosine phosphorylation of p34cdc2. Cancer Res. 54, 1412–1414 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yao, SL., Akhtar, A., Mckenna, K. et al. Selective radiosensitization of p53–deficient cells by caffeine–mediated activation of p34cdc2 kinase. Nat Med 2, 1140–1143 (1996). https://doi.org/10.1038/nm1096-1140
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1096-1140
This article is cited by
-
The flavonoid apigenin improves glucose tolerance through inhibition of microRNA maturation in miRNA103 transgenic mice
Scientific Reports (2013)
-
Caffeine does not enhance radiosensitivity of normal liver tissue in vivo
Molecular Biology Reports (2011)
-
Discovery of gene expression-based pharmacodynamic biomarker for a p53 context-specific anti-tumor drug Wee1 inhibitor
Molecular Cancer (2009)
-
Many forks in the path: cycling with FoxO
Oncogene (2008)
-
Hsp90-inhibitor geldanamycin abrogates G2 arrest in p53-negative leukemia cell lines through the depletion of Chk1
Oncogene (2008)