Abstract
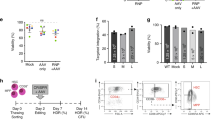
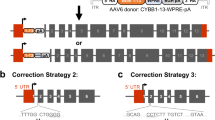
X-linked immunodeficiency with hyper-IgM (HIGM1), characterized by failure of immunoglobulin isotype switching, is caused by mutations of the CD40 ligand (CD40L), which is normally expressed on activated CD4+ T cells. As constitutive expression of CD40L induces lymphomas, we corrected the mutation while preserving the natural regulation of CD40L using pre-mRNA trans-splicing. Bone marrow from mice lacking CD40L was modified with a lentivirus trans-splicer encoding the normal CD40L exons 2–5 and was administered to syngenic CD40L-knockout mice. Recipient mice had corrected CD40L mRNA, antigen-specific IgG1 responses to keyhole limpet hemocyanin immunization, regulated CD4+ T-cell CD40L expression after CD3 stimulation in primary and secondary transplanted mice, attenuation of Pneumocystis carinii pneumonia, and no evidence of lymphoproliferative disease over 1 year. Thus, HIGM1 can be corrected by CD40L trans-splicing, leading to functional correction of the genetic defect without the adverse consequences of unregulated expression of the CD40L gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Callard, R.E., Armitage, R.J., Fanslow, W.C. & Spriggs, M.K. CD40 ligand and its role in X-linked hyper-IgM syndrome. Immunol. Today 14, 559–564 (1993).
Kroczek, R.A. et al. Defective expression of CD40 ligand on T cells causes “X-linked immunodeficiency with hyper-IgM (HIGM1)”. Immunol. Rev. 138, 39–59 (1994).
Di Santo, J.P., Bonnefoy, J.Y., Gauchat, J.F., Fischer, A. & de Saint, B.G. CD40 ligand mutations in X-linked immunodeficiency with hyper-IgM. Nature 361, 541–543 (1993).
Korthauer, U. et al. Defective expression of T-cell CD40 ligand causes X-linked immunodeficiency with hyper-IgM. Nature 361, 539–541 (1993).
Roy, M., Waldschmidt, T., Aruffo, A., Ledbetter, J.A. & Noelle, R.J. The regulation of the expression of gp39, the CD40 ligand, on normal and cloned CD4+ T cells. J. Immunol. 151, 2497–2510 (1993).
Stout, R.D., Suttles, J., Xu, J., Grewal, I.S. & Flavell, R.A. Impaired T cell-mediated macrophage activation in CD40 ligand-deficient mice. J. Immunol. 156, 8–11 (1996).
Grewal, I.S. & Flavell, R.A. CD40 and CD154 in cell-mediated immunity. Annu. Rev. Immunol. 16, 111–135 (1998).
Straw, A.D., MacDonald, A.S., Denkers, E.Y. & Pearce, E.J. CD154 plays a central role in regulating dendritic cell activation during infections that induce Th1 or Th2 responses. J. Immunol. 170, 727–734 (2003).
Gennery, A.R. et al. Treatment of CD40 ligand deficiency by hematopoietic stem cell transplantation: a survey of the European experience, 1993-2002. Blood 103, 1152–1157 (2004).
Levy, J. et al. Clinical spectrum of X-linked hyper-IgM syndrome. J. Pediatr. 131, 47–54 (1997).
Hadzic, N. et al. Correction of the hyper-IgM syndrome after liver and bone marrow transplantation. N. Engl. J. Med. 342, 320–324 (2000).
Thomas, C. et al. Brief report: correction of X-linked hyper-IgM syndrome by allogeneic bone marrow transplantation. N. Engl. J. Med. 333, 426–429 (1995).
Brown, M.P. et al. Thymic lymphoproliferative disease after successful correction of CD40 ligand deficiency by gene transfer in mice. Nat. Med. 4, 1253–1260 (1998).
Sacco, M.G. et al. Lymphoid abnormalities in CD40 ligand transgenic mice suggest the need for tight regulation in gene therapy approaches to hyper immunoglobulin M (IgM) syndrome. Cancer Gene Ther. 7, 1299–1306 (2000).
Sullenger, B.A. Targeted genetic repair: an emerging approach to genetic therapy. J. Clin. Invest. 112, 310–311 (2003).
Puttaraju, M., Jamison, S.F., Mansfield, S.G., Garcia-Blanco, M.A. & Mitchell, L.G. Spliceosome-mediated RNA trans-splicing as a tool for gene therapy. Nat. Biotechnol. 17, 246–252 (1999).
Garcia-Blanco, M.A., Puttaraju, M., Mansfield, S.G. & Mitchell, L.G. Spliceosome-mediated RNA trans-splicing in gene therapy and genomics. Gene Ther. Regul. 1, 141–163 (2000).
Liu, X. et al. Partial correction of endogenous DeltaF508 CFTR in human cystic fibrosis airway epithelia by spliceosome-mediated RNA trans-splicing. Nat. Biotechnol. 20, 47–52 (2002).
Mansfield, S.G. et al. Repair of CFTR mRNA by spliceosome-mediated RNA trans-splicing. Gene Ther. 7, 1885–1895 (2000).
Puttaraju, M., Di Pasquale, J., Baker, C.C., Mitchell, L.G. & Garcia-Blanco, M.A. Messenger RNA repair and restoration of protein function by spliceosome-mediated RNA trans-splicing. Mol. Ther. 4, 105–114 (2001).
Chao, H. et al. Phenotype correction of hemophilia A mice by spliceosome-mediated RNA trans-splicing. Nat. Med. 9, 1015–1019 (2003).
Garcia-Blanco, M.A. Messenger RNA reprogramming by spliceosome-mediated RNA trans-splicing. J. Clin. Invest. 112, 474–480 (2003).
Bonen, L. Trans-splicing of pre-mRNA in plants, animals, and protists. FASEB J. 7, 40–46 (1993).
Caudevilla, C. et al. Natural trans-splicing in carnitine octanoyltransferase pre-mRNAs in rat liver. Proc. Natl. Acad. Sci. USA 95, 12185–12190 (1998).
Xu, J. et al. Mice deficient for the CD40 ligand. Immunity. 1, 423–431 (1994).
Testi, R., D'Ambrosio, D., De Maria, R. & Santoni, A. The CD69 receptor: a multipurpose cell-surface trigger for hematopoietic cells. Immunol. Today 15, 479–483 (1994).
Kolls, J.K., Beck, J.M., Nelson, S., Summer, W.R. & Shellito, J. Alveolar macrophage release of tumor necrosis factor during murine Pneumocystis carinii pneumonia. Am. J. Respir. Cell Mol. Biol. 8, 370–376 (1993).
Kolls, J.K. et al. IFN-γ and CD8+ T cells restore host defenses against Pneumocystis carinii in mice depleted of CD4+ T cells. J. Immunol. 162, 2890–2894 (1999).
Logan, A.C., Lutzko, C. & Kohn, D.B. Advances in lentiviral vector design for gene-modification of hematopoietic stem cells. Curr. Opin. Biotechnol. 13, 429–436 (2002).
Baumgartner, R. et al. Evidence for the requirement of T cell costimulation in the pathogenesis of natural Pneumocystis carinii pulmonary infection. Microb. Pathog. 33, 193–201 (2002).
Campbell, K.A. et al. CD40 ligand is required for protective cell-mediated immunity to Leishmania major. Immunity 4, 283–289 (1996).
MacDonald, A.S. et al. Impaired Th2 development and increased mortality during Schistosoma mansoni infection in the absence of CD40/CD154 interaction. J. Immunol. 168, 4643–4649 (2002).
Netea, M.G., Meer, J.W., Verschueren, I. & Kullberg, B.J. CD40/CD40 ligand interactions in the host defense against disseminated Candida albicans infection: the role of macrophage-derived nitric oxide. Eur. J. Immunol. 32, 1455–1463 (2002).
Soong, L. et al. Disruption of CD40-CD40 ligand interactions results in an enhanced susceptibility to Leishmania amazonensis infection. Immunity 4, 263–273 (1996).
Stephens, J., Cosyns, M., Jones, M. & Hayward, A. Liver and bile duct pathology following Cryptosporidium parvum infection of immunodeficient mice. Hepatology 30, 27–35 (1999).
Thomsen, A.R., Nansen, A., Christensen, J.P., Andreasen, S.O. & Marker, O. CD40 ligand is pivotal to efficient control of virus replication in mice infected with lymphocytic choriomeningitis virus. J. Immunol. 161, 4583–4590 (1998).
Merali, S., Chin, K., Grady, R.W., Weissberger, L. & Clarkson, A.B. Response of rat model of Pneumocystis carinii pneumonia to continuous infusion of deferoxamine. Antimicrob. Agents Chemother. 39, 1442–1444 (1995).
Zheng, M. et al. CD4+ T cell-independent vaccination against Pneumocystis carinii in mice. J. Clin. Invest. 108, 1469–1474 (2001).
Pergolizzi, R.G. et al. In vivo trans-splicing of 5′ and 3′ segments of pre-mRNA directed by corresponding DNA sequences delivered by gene transfer. Mol. Ther. 8, 999–1008 (2003).
Armitage, R.J. et al. Molecular and biological characterization of a murine ligand for CD40. Nature 357, 80–82 (1992).
Field, J. et al. Purification of a RAS-responsive adenylyl cyclase complex from Saccharomyces cerevisiae by use of an epitope addition method. Mol. Cell. Biol. 8, 2159–2165 (1988).
Xu, K., Ma, H., McCown, T.J., Verma, I.M. & Kafri, T. Generation of a stable cell line producing high-titer self-inactivating lentiviral vectors. Mol. Ther. 3, 97–104 (2001).
Naldini, L. et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 272, 263–267 (1996).
Dull, T. et al. A third-generation lentivirus vector with a conditional packaging system. J. Virol. 72, 8463–8471 (1998).
Szilvassy, S.J., Lansdorp, P.M., Humphries, R.K., Eaves, A.C. & Eaves, C.J. Isolation in a single step of a highly enriched murine hematopoietic stem cell population with competitive long-term repopulating ability. Blood 74, 930–939 (1989).
Vigna, E. et al. Robust and efficient regulation of transgene expression in vivo by improved tetracycline-dependent lentiviral vectors. Mol. Ther. 5, 252–261 (2002).
Follenzi, A., Ailles, L.E., Bakovic, S., Geuna, M. & Naldini, L. Gene transfer by lentiviral vectors is limited by nuclear translocation and rescued by HIV-1 pol sequences. Nat. Genet. 25, 217–222 (2000).
Kikuchi, T., Worgall, S., Singh, R., Moore, M.A. & Crystal, R.G. Dendritic cells genetically modified to express CD40 ligand and pulsed with antigen can initiate antigen-specific humoral immunity independent of CD4+ T cells. Nat. Med. 6, 1154–1159 (2000).
Acknowledgements
We thank K. Nakayama and Q. Jiang for help in construction of trans-splicing vectors; A. Busch for flow cytometry analysis; N. Hackett and A. Cieciuch for real-time RT-PCR analysis; K. Kasuya for histological analysis; T. Kafri for providing plasmid; and N. Mohamed for help preparing this manuscript. These studies were supported, in part, by U01 HL66952, the Will Rogers Memorial Fund and The Malcolm Hewitt Wiener Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Schematic illustration of trans-splicer plasmid and demonstration of correction of mutated CD40L by trans-splicing in vitro. (PDF 162 kb)
Supplementary Fig. 2
Identification of the most efficient hybridization domain as assessed by in vitro trans-splicing-mediated correction of murine CD40L pre-mRNA. (PDF 53 kb)
Supplementary Fig. 3
Assessment of restoration of function following in vitro trans-splicing-mediated correction of CD40L pre-mRNA. (PDF 111 kb)
Rights and permissions
About this article
Cite this article
Tahara, M., Pergolizzi, R., Kobayashi, H. et al. Trans-splicing repair of CD40 ligand deficiency results in naturally regulated correction of a mouse model of hyper-IgM X-linked immunodeficiency. Nat Med 10, 835–841 (2004). https://doi.org/10.1038/nm1086
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1086
This article is cited by
-
Reprogramming the Dynamin 2 mRNA by Spliceosome-mediated RNA Trans-splicing
Molecular Therapy - Nucleic Acids (2016)
-
Construction and validation of an RNA trans-splicing molecule suitable to repair a large number of COL7A1 mutations
Gene Therapy (2016)
-
Trans-splicing repair of mutant p53 suppresses the growth of hepatocellular carcinoma cells in vitro and in vivo
Scientific Reports (2015)
-
Repair of Rhodopsin mRNA by Spliceosome-Mediated RNA Trans -Splicing: A New Approach for Autosomal Dominant Retinitis Pigmentosa
Molecular Therapy (2015)
-
Cis -splicing and Translation of the Pre- Trans -splicing Molecule Combine With Efficiency in Spliceosome-mediated RNA Trans -splicing
Molecular Therapy (2014)