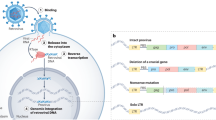
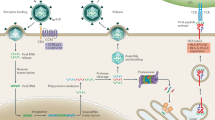
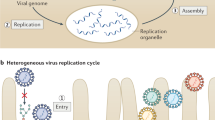
Abstract
Human retroviruses have developed novel strategies for their propagation and survival. A consequence of their success has been the induction of an extraordinarily diverse set of human diseases, including AIDS, cancers and neurological and inflammatory disorders. Early research focused on their characterization, linkage to these diseases, and the mechanisms involved. Research should now aim at the eradication of human retroviruses and on treatment of infected people.
Similar content being viewed by others
Article PDF
References
Temin, H.M. Origin and general nature of retrovirus. The Retroviridae, vol. 1 (ed. Levy, J.A.) (Plenum, New York, 1992).
Coffin, J.M. Structure and classification of retroviruses. The Retroviridae, vol. 1 (ed. Levy, J.A.) (Plenum, New York, 1992).
Gallo, R.C. The story of retroviruses and cancer: from poultry to people. in Virus Hunting 59–81 (Basic Books, New York, 1991).
Lower, R. et al. Identification of human endogenous retroviruses with complex mRNA expression and particle formation. Proc. natn. Acad. Sci. U.S.A. 90, 4480–4484 (1983).
Barré-Sinoussi, F. et al. Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). Science 220, 868–871 (1993).
Popovic, M., Sarngadharan, M.G., Read, E. & Gallo, R.C., Detection, isolation, and continuous production of cytopathic retroviruses (HTLV-III) from patients with AIDS and pre-AIDS. Science 224, 497–500 (1984).
Gallo, R.C. et al. Frequent detection and isolation of cytopathic retroviruses (HTLV-III) from patients with AIDS and at risk for AIDS. Science 224, 500–503 (1984).
Sarngadharan, M.G., Popovic, M., Bruch, L., Schupbach, J. & Gallo, R.C. Antibodies reactive with human T-lymphotropic retroviruses (HTLV-III) in the serum of patients with AIDS. Science 224, 506–508 (1984).
Gallo, R.C. The first human retrovirus. Sci. Am. 255 (Dec), 88–98 (1986).
Poiesz, B.J. et al. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc. natn. Acad. Sci. U.S.A. 77, 7415–7419 (1980).
Miyoshi, I. et al. Type C virus particles in a cord T-cell line derived by co-cultivating normal human cord leukocytes and human leukaemic T cells. Nature 294, 770–771 (1981).
Gallo, R.C. Discovery of a cancer virus: The first human retrovirus. in Virus Hunting 99–115 (Basic Books, New York, 1991).
Franchini, G. Molecular mechanisms of HTLV-I infection. Blood (in the press).
Kalyanaraman, V.S. et al. A new subtype of human T-cell leukemia virus (HTLV-II) associated with a T-cell variant of hairy cell leukemia. Science 218, 571–573 (1982).
Lairmore, M.D. et al. Isolation of human T-cell lymphotropic virus type 2 from Guaymi Indians in Panama. Proc. natn. Acad. Sci. U.S.A. 87, 8840–8844 (1990).
Robert-Guroff, M. et al. Prevalence of antibodies to HTLV-I, -II, and -III in intravenous drug abusers from an AIDS endemic region. JAMA 255, 3133–3137 (1986).
Zagury, D. et al. Long-term cultures of HTLV-III-infected T cells: A model of cytopathology of T-cell depletion in AIDS. Science 231, 850–853 (1986).
Wei, X. et al. Viral dynamics in human immnunodeficiency virus type 1 infection. Nature 373, 117–122 (1995).
Ho, D.D. et al. Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection. Nature 373, 123–126 (1995).
Pantaleo, G., Graziosi, C. & Fauci, A.S. The role of lymphoid organs in the pathogenesis of HIV infection. Semin. Immun. 5, 157–163 (1993).
Embretson, J. et al. Analysis of human immunodeficiency virus-infected tissues by amplification and in situ hybridization reveals latent and permissive infections at single-cell resolution. Proc. natn. Acad. Sci. U.S.A. 90, 357–361 (1993).
Catovsky, D. et al. Adult T-cell lymphoma-leukaemia in Blacks from the West Indies. Lancet 1, 639–643 (1982).
Robert-Guroff, M. et al. Natural antibodies to human retrovirus HTLV in a cluster of Japanese patients with adult T cell leukemia. Science 215, 975–978 (1982).
Bartholomew, C. et al. HTLV-I and tropical spastic paraparesis [letter]. Lancet 2, 99–100 (1986).
Grassmann, R. et al. Transformation to continuous growth of primary human T lymphocytes by human T-cell leukemia virus type I X-region genes transduced by a Herpesvirus saimiri vector. Proc. natn. Acad. Sci. U.S.A. 86, 3351–3355 (1989).
Tanaka, A. et al. Oncogenic transformation by the tax gene of human T-cell leukemia virus type I in vitro. Proc. natn. Acad. Sci. U.S.A. 87, 1071–1075 (1990).
Saggioro, D. et al. Tax protein of human T-lymphotropic virus type I triggers DNA damage. Leuk. Lymphoma 12, 281–286 (1994).
Gessain, A. et al. Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet 2, 407–410 (1985).
Osame, M. et al. HTLV-I associated myelopathy, a new clinical entity [letter]. Lancet 1, 1031–1032 (1986).
Waldmann, T.A. Anti-IL-2 receptor monoclonal antibody (anti-Tac) treatment of T-cell lymphoma. Important Adv. Oncol. 131–141 (1994).
Gill, P.S. et al. Treatment of adult T-cell leukemia-lymphoma with a combination of interferon alfa and zidovudine. New Engl.J. Med. 332, 1744–1748 (1995).
Hermine, O. et al. Brief report: treatment of adult T-cell leukemia-lymphoma with zidovudine and interferon alfa. New Engl. J. Med. 332, 1749–1751 (1995).
Haseltine, W.A. & Wong-Staal, F. The molecular biology of the AIDS virus. Sci. Am. 259 (Oct.), 52–62 (1988).
Cullen, B.R. & Greene, W.C. Functions of the auxiliary gene products of the human immunodeficiency virus type 1. Virology 178, 1–5 (1990).
Ensoli, B., Barillari, G., Salahuddin, S.Z., Gallo, R.C. & Wong-Staal, F. Tat protein of HIV-1 stimulates growth of cells derived from Kaposi's sarcoma lesions of AIDS patients. Nature 345, 84–86 (1990).
Li, C.J., Friedman, D.J., Wang, C., Metelev, V. & Pardee, A.B. Induction of apoptosis in uninfected lymphocytes by HIV-1 Tat protein. Science 268, 429–431 (1995).
Rogel, M.E., Wu, L.I. & Emerman, M. The human immunodeficiency virus type I vpr gene prevents cell proliferation during chronic infection. J. Virol. 69, 882–888 (1995).
Bukrinsky, M.I. et al. A nuclear localization signal within HIV-1 matrix protein that governs infection of non-dividing cells. Nature 365, 666–669 (1993).
Bukrinsky, M.I. et al. Active nuclear import of human immunodeficiency virus type 1 preintegration complexes. Proc. natn. Acad. Sci. U.S.A. 89, 6580–6584 (1992).
Balotta, C., Lusso, P., Crowley, R., Gallo, R.C. & Franchini, G. Antisense phosphorothioate oligodeoxynucleotides targeted to the vpr gene inhibit human immunodeficiency virus type 1 replication in primary human macrophages. J. Virol. 67, 4409–4414 (1993).
Saksela, K., Cheng, G. & Baltimore, D. Proline-rich (PxxP) motifs in HIV-1 Nef bind to SH3 domains of a subset of Src kinases and are required for the enhanced growth of Nef+ viruses but not for down-regulation of CD4. EMBO J. 14, 484–491 (1995).
Baur, A.S. et al. HIV-1 Nef leads to inhibition or activation of T cells depending on its intracellular localization. Immunity 1, 373–384 (1994).
Strebel, K., Klimkait, T. & Martin, M.A. A novel gene of HIV-1, vpu, and its 16-kilodalton product. Science 241, 1221–1223 (1988).
Zagury, J.F. et al. Identification of CD4 and major histocompatibility complex functional peptide sites and their homology with oligopeptides from human immunodeficiency virus type 1 glycoprotein gpl20: Role in AIDS pathogenesis. Proc. natn. Acad. Sci. U.S.A. 90, 7573–7577 (1993).
Finkel, T.H. et al. Apoptosis occurs predominantly in bystander cells and not in productively infected cells of HIV- and SIV-infected lymph nodes. Nature Med. 1, 129–134 (1995).
Pantaleo, G. & Fauci, A.S. Apoptosis in HIV infection. Nature Med. 1, 118–120 (1995).
Turano, A. et al. Natural human antibodies to interferon-interfere with the immunomodulating activity of the lymphokine. Proc. natn. Acad. Sci. U.S.A. 89, 4447–4451 (1992).
Chang, Y. et al. Identification of herpesvirus-like DNA sequences in AIDS-as-sociated Kaposi's sarcoma. Science 266, 1865–1869 (1994).
Nakamura, S. et al. Kaposi's sarcoma cells: Long-term culture with growth factor from retrovirus-infected CD4+ T cells. Science 242, 426–430 (1988).
Ensoli, B. et al. Synergy between basic fibroblast growth factor and HIV-1 Tat protein in induction of Kaposi's sarcoma. Nature 371, 674–680 (1994).
Ensoli, B. & Gallo, R.C., Kaposi's sarcoma: A new perspective of its pathogenesis and treatment. Proc. Assoc. Am. Physicians 107, 8–18 (1995).
Lunardi-Iskandar, Y. et al. Tumorigenesis and metastasis of neoplastic Kaposi's sarcoma cell line in immunodeficient mice blocked by a human pregnancy hormone. Nature 375, 64–68 (1995).
Siegal, B. et al. Kaposi sarcoma in immunosuppression. Possibly the result of a dual viral infection. Cancer 65, 492–498 (1990).
Salahuddin, S.Z. et al. Isolation of a new virus, HBLV, in patients with lymphoproliferative disorders. Science 234, 596–601 (1986).
Lori, F. et al. Hydroxyurea as an inhibitor of human immunodeficiency virus-type 1 replication. Science 266, 801–805 (1994).
Baba, T.W. et al. Pathogenicity of live, attenuated SIV after mucosal infection of neonatal macaques. Science 267, 1820–1825 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gallo, R. Human retroviruses in the second decade: A personal perspective. Nat Med 1, 753–759 (1995). https://doi.org/10.1038/nm0895-753
Issue Date:
DOI: https://doi.org/10.1038/nm0895-753