Abstract
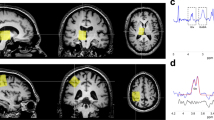
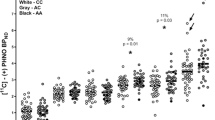
Animal studies suggest that development of substance dependence is associated with dopaminergic activity in striatum and the limbic system. Several genetic studies indicate that allele A1 is associated with both D2 receptor density and alcoholism, although these findings have remained controversial. We studied striatal dopamine (DA) re-uptake site densities in 48 subjects (19 healthy controls, 19 habitually impulsive violent alcoholics, and 10 non-violent alcoholics) with single photon emission computed tomography (SPECT) using iodine-123-I a belled 2β-carbomethoxy-3β (4-iodophenyl)tropane, (β-CIT) as a tracer. Blind quantitative analysis revealed that the striatal DA transporter density was markedly lower in non-violent alcoholics than in healthy controls (P < 0.001), while violent alcoholics had slightly higher DA transporter densities than controls (P < 0.10). The results indicate that both types of alcoholics have alterations in striatal dopaminergic system, though these occur in opposite directions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Koob, G.F. Drugs of abuse: Anatomy, pharmacology and function of reward pathways. Trends pharmacol. Sci. 13, 177–184 (1992).
Päivärinta, P. Predisposition to high ethanol drinking. Role of ethanol reward, and relationship to central control of eating and aggressiveness in rodents [dissertation]. (Alko, Helsinki, 1993).
Rossetti, Z.L., D'Aquila, P.S., Hmaidan, Y., Gessa, G.L. & Serra, G. Repeated treatment with imipramine potentiates cocaine-induced dopamine release and motor stimulation. Eur. J. Pharmacol. 201, 243–245 (1991).
Sorensen, S.M., Jumphreys, T.M., Taylor, V.L. & Schmidt, C.J. 5-HT2 receptor antagonists reverse amphetamine-induced slowing of dopaminergic neurons by interfering with stimulated dopamine synthesis. J. Pharmac. exp. Ther. 260, 872–878 (1992).
Spanagel, R., Herz, A., Bals-Kubik, R. & Shippenberg, T.S. β-Endorphin-induced locomotor stimulation and reinforcement are associated with an increase in dopamine release in the nucleus accumbens. Psychopharmacology (Berlin) 104, 51–56 (1991).
Yoshimoto, K., McBride, W.J., Lumeng, L. & Li, T-K. Alcohol stimulates the release of dopamine and serotonin in the nucleus accumbens. Alcohol 9, 17–22 (1991).
Kalivas, P.W. Neurotransmitter regulation of dopamine neurons in the ventral tegmental area. Brain Res. Revs 18, 75–113 (1993).
Dyr, W., McBride, W.J., Lumen, L., Li, T-K. & Murphy, J.M. Effects of D1 and D2 dopamine receptor agents on ethanol consumption in the high-alcohol-drinking (HAD) line of rats. Alcohol 10, 207–212 (1993).
Caine, S.B. & Koob, G.F. Modulation of cocaine self-administration in the rat through D3 dopamine receptors. Science 260, 1814–1816 (1993).
Blum, K. et al. Allelic association of human dopamine D2 receptor gene in alcoholism. JAMA 263, 2055–2060 (1990).
Comings, D.E. et al. The dopamine D2 receptor locus as a modifying gene in neuropsychiatric disorders. JAMA 266, 1793–1800 (1991).
Smith, S.S. et al. Genetic vulnerability of drug abuse: The D2 dopamine receptor TagI B1 restriction fragment length polymorphism appears more frequently in polysubstance abusers. Arch. gen. Psychiat 49, 723–727 (1992).
Gelernter, J. et al. No association between an allele at the D2 dopamine receptor gene (DRD2) and alcoholism. JAMA 266, 1801–1807 (1991).
Goldman, D. et al. D2 dopamine receptor genotype and cerebrospinal fluid ho-movanillic acid, 5-hydyroxyindoleacetic acid and 3-methoxy-4-hydroxy-phenylglycol in alcoholics in Finland and the United States. Ada psychiat. scand. 86, 351–357 (1993).
Goldman, D. et al. D2 dopamine receptor genotype, linkage disequilibrium and dopamine duction in Finnish, American Indian and U.S. Caucasian alcoholics and the population association approach in psychogenetics. Alcohol, din. exp. Res. 17, 199–204 (1993).
Cloninger, C.R. D2 dopamine receptor gene is associated but not linked with alcoholism. JAMA 266, 1833–1834 (1991).
Gelernter, J., Goldman, D. & Risen, N., Al allele at the D2 dopamine receptor gene and alcoholism: A reappraisal. JAMA 269, 1673–1677 (1993).
Uhl, G., Blum, K., Noble, E. & Smith, S. Substance abuse vulnerability and D2 receptor genes. Trends Neurosci. 16, 83–88 (1993).
Noble, E.P., Blum, K., Ritschie, T., Montgomery, A. & Sheridan, P.J. Allelic association of the D2 dopamine receptor gene with receptor-binding characteristics in alcoholism. Arch. gen. Psychiat 48, 643–647 (1991).
Hietala, J. et al. Striatal D2 dopamine receptor binding characteristics in vivo in patients with alcohol dependence. Psychopharmacology 116, 285–290 (1994).
Uhl, G.R. Neurotransmitter transporter (plus): A promising new gene family. Trends. Neurosci. 15, 265–268 (1992).
Kuikka, J.T. et al. Initial experience with SPET examinations using [123I]2β-carbomethoxy-3β(4-iodophenyl)tropane ([123I]β-CIT) in human brain. Eur. J. nucl. Med. 20, 783–786 (1993).
Farde, L. et al. PET-study of [11C] β-CIT binding to monoamine transporters in the monkey and human brain. Synapse 16, 93–103 (1994).
Brücke, T. et al. SPECT imaging of dopamine and serotonin transporters with [123I]β-CIT. Binding kinetics in the human brain. J. neural. Trans. 94, 137–146 (1993).
Innis, R. et al. SPECT imaging demonstrates loss of striatal dopamine transporters in Parkinson's disease. Proc. natn. Acad. Sci. U.S.A. 90, 11965–11969 (1993).
Kuikka, J.T., Bergström, K.A., Ahonen, A. & Länsimies, E. The dosimetry of iodine-123 labelled 2βP-carbomethoxy-3β-(4-iodophenyl)tropane. Eur. J. nucl. Med. 21, 53–56 (1994).
Lycke, E., Modigh, K. & Roos, B.E. Aggression in mice associated with changes in the monoamine-metabolism of the brain. Experientia 25, 951–953 (1969).
Gejman, P.V. et al. No structural mutation in the dopamine D2 receptor gene in alcoholism or schizophrenia. JAMA 271, 204–208 (1994).
Cloninger, C.R. Neurogenetic adaptive mechanisms in alcoholism. Science 236, 410–416 (1987).
Bergstöm, K.A., Kuikka, J.T., Ahonen, A. & Vanninen, E. E.[123I]β-CIT, a tracer for dopamine and serotonin reuptake sites, preparation and preliminary SPET studies in humans. J. nucl. biol. Med. 37, 1425–1428 (1994).
Kuikka, J.T., Tenhunen-Eskelinen, M., Jurvelin, J. & Kiiliäinen, H. Physical performance of the Siemens MultiSPECT 3 gamma camera. Nucl. Med. Commun. 14, 490–497 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tiihonen, J., Kuikka, J., Bergström, K. et al. Altered striatal dopamine re-uptake site densities in habitually violent and non-violent alcoholics. Nat Med 1, 654–657 (1995). https://doi.org/10.1038/nm0795-654
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0795-654
This article is cited by
-
Effects of sedative drug use on the dopamine system: a systematic review and meta-analysis of in vivo neuroimaging studies
Neuropsychopharmacology (2019)
-
Characterization of a dopamine transporter polymorphism and behavior in Belgian Malinois
BMC Genetics (2013)
-
Involvement of nucleus accumbens dopamine D1 receptors in ethanol drinking, ethanol-induced conditioned place preference, and ethanol-induced psychomotor sensitization in mice
Psychopharmacology (2012)