Abstract
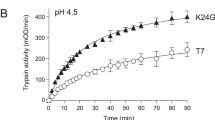
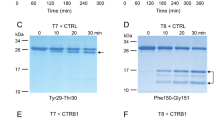
Chronic pancreatitis is a common inflammatory disease of the pancreas. Mutations in the genes encoding cationic trypsinogen (PRSS1)1 and the pancreatic secretory trypsin inhibitor (SPINK1)2 are associated with chronic pancreatitis. Because increased proteolytic activity owing to mutated PRSS1 enhances the risk for chronic pancreatitis, mutations in the gene encoding anionic trypsinogen (PRSS2) may also predispose to disease. Here we analyzed PRSS2 in individuals with chronic pancreatitis and controls and found, to our surprise, that a variant of codon 191 (G191R) is overrepresented in control subjects: G191R was present in 220/6,459 (3.4%) controls but in only 32/2,466 (1.3%) affected individuals (odds ratio 0.37; P = 1.1 × 10−8). Upon activation by enterokinase or trypsin, purified recombinant G191R protein showed a complete loss of trypsin activity owing to the introduction of a new tryptic cleavage site that renders the enzyme hypersensitive to autocatalytic proteolysis. In conclusion, the G191R variant of PRSS2 mitigates intrapancreatic trypsin activity and thereby protects against chronic pancreatitis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Whitcomb, D.C. et al. Hereditary pancreatitis is caused by a mutation in the cationic trypsinogen gene. Nat. Genet. 14, 141–145 (1996).
Witt, H. et al. Mutations in the gene encoding the serine protease inhibitor, Kazal type 1 are associated with chronic pancreatitis. Nat. Genet. 25, 213–216 (2000).
Scheele, G., Bartelt, D. & Bieger, W. Characterization of human exocrine pancreatic proteins by two-dimensional isoelectric focusing/SDS gel electrophoresis. Gastroenterology 80, 461–473 (1981).
Chen, J.M. & Férec, C. Human trypsins. in Handbook of Proteolytic Enzymes 2nd edn. (eds. Barrett, A.J. et al.) 1489–1493 (Elsevier, London, 2004).
Colomb, E. & Figarella, C. Comparative studies on the mechanism of activation of the two human trypsinogens. Biochim. Biophys. Acta 571, 343–351 (1979).
Colomb, E., Guy, O., Deprez, P., Michel, R. & Figarella, C. The two human trypsinogens: catalytic properties of the corresponding trypsins. Biochim. Biophys. Acta 525, 186–193 (1978).
Rowen, L., Koop, B.F. & Hood, L. The complete 685-kilobase DNA sequence of the human beta T cell receptor locus. Science 272, 1755–1762 (1996).
Kénesi, E., Katona, G. & Szilágyi, L. Structural and evolutionary consequences of unpaired cysteines in trypsinogen. Biochem. Biophys. Res. Commun. 309, 749–754 (2003).
Witt, H., Luck, W. & Becker, M. A signal peptide cleavage site mutation in the cationic trypsinogen gene is strongly associated with chronic pancreatitis. Gastroenterology 117, 7–10 (1999).
Barrett, J.C., Fry, B., Maller, J. & Daly, M.J. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–265 (2005).
Gaboriaud, C., Serre, L., Guy-Crotte, O., Forest, E. & Fontecilla-Camps, J.-C. Crystal structure of human trypsin 1: unexpected phosphorylation of Tyr151. J. Mol. Biol. 259, 995–1010 (1996).
Guex, N. & Peitsch, M.C. SWISS-MODEL and the Swiss-PdbViewer: An environment for comparative protein modeling. Electrophoresis 18, 2714–2723 (1997).
Sahin-Tóth, M. Human cationic trypsinogen. Role of Asn-21 in zymogen activation and implications in hereditary pancreatitis. J. Biol. Chem. 275, 22750–22755 (2000).
Sahin-Tóth, M. & Tóth, M. Gain-of-function mutations associated with hereditary pancreatitis enhance autoactivation of human cationic trypsinogen. Biochem. Biophys. Res. Commun. 278, 286–289 (2000).
Kukor, Z., Tóth, M. & Sahin-Tóth, M. Human anionic trypsinogen. Properties of autocatalytic activation and degradation and implications in pancreatic diseases. Eur. J. Biochem. 270, 2047–2058 (2003).
Acknowledgements
We thank the many patients who have participated in this study. We are especially grateful to V. Kielstein (Magdeburg, Germany) and L. Klomp (University Medical Center Utrecht, Utrecht, The Netherlands) for providing blood samples from control subjects. The initial experiments were supported by the DFG (Wi 2036/1-1). This work was supported by the Sonnenfeld-Stiftung, Berlin, Germany (to H.W.), the US National Institutes of Health (NIH) (grant DK058088 to M.S.-T.), INSERM (Institut National de la Santé et de la Recherche Médicale) and the Programme Hospitalier de Recherche Clinique (grant PHRC R 08-04 to C.F.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
Distribution of G191R in the different patient groups. (PDF 49 kb)
Supplementary Table 2
Characteristics of patients. (PDF 59 kb)
Rights and permissions
About this article
Cite this article
Witt, H., Sahin-Tóth, M., Landt, O. et al. A degradation-sensitive anionic trypsinogen (PRSS2) variant protects against chronic pancreatitis. Nat Genet 38, 668–673 (2006). https://doi.org/10.1038/ng1797
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1797
This article is cited by
-
Expanding ACMG variant classification guidelines into a general framework
Human Genomics (2022)
-
Trypsinogen (PRSS1 and PRSS2) gene dosage correlates with pancreatitis risk across genetic and transgenic studies: a systematic review and re-analysis
Human Genetics (2022)
-
Genetic Testing in Acute and Chronic Pancreatitis
Current Treatment Options in Gastroenterology (2022)
-
Prevalence and Risk Factors for Osteopathy in Chronic Pancreatitis
Digestive Diseases and Sciences (2021)
-
A novel homozygous nonsense mutation in the CA2 gene (c.368G>A, p.W123X) linked to carbonic anhydrase II deficiency syndrome in a Chinese family
Metabolic Brain Disease (2021)