Abstract
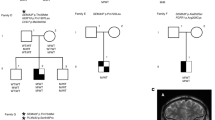
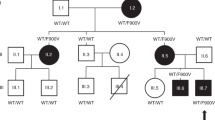
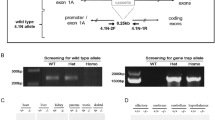
The family of basic helix-loop-helix (bHLH) genes comprises transcription factors involved in many aspects of growth and development. We have previously described two bHLH transcription factors, Nhlh1 and Nhlh2 (originally named NSCL1 and NSCL2)1–3. The nucleotide and predicted protein sequences of Nhlh1 and Nhlh2 are homologous within their bHLH domain where there are only three conservative amino acid differences2,3. During murine embryogenesis, Nhlh1 and Nhlh2 share an overlapping but distinct pattern of expression in the developing nervous system1,2. To improve our understanding of the role of these genes during neurogenesis, we have generated mice containing targeted deletions of both genes and here describe our results for Nhlh2. Loss of Nhlh2 results in a disruption of the hypothalamic-pituitary axis in mice. Male Nhlh2−/ − mice are microphallic, hypogonadal and infertile with alterations in circulating gonadotropins, a defect in spermatogenesis and a loss of instinctual male sexual behaviour. Female Nhlh2−/ − mice reared alone are hypogonadal, but when reared in the presence of males, their ovaries and uteri develop normally and they are fertile. Both male and female homozygotes exhibit progressive adult-onset obesity. Nhlh2 is expressed in the ventral-medial and lateral hypothalamus, Rathke's pouch and in the anterior lobe of the adult pituitary. Our results support a role for Nhlh2 in the onset of puberty and the regulation of body weight metabolism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Begley, C.G. et al. Molecular characterization of NSCL, a gene encoding a helix-loop-helix protein expressed in the developing nervous system. Proc. Natl. Acad. Sci. USA 89, 38–42 (1992).
Gobel, V., Lipkowitz, S., Kozak, C.A. & Kirsch, I.R. NSCL-2: A basic domain helix-loop-helix gene expressed in early neurogenesis. Cell Growth Differ. 3, 143–148 (1992).
Lipkowitz, S. et al. A comparative structural characterization of the human IMSCL-1 and NSCL-2 genes. J. Biol. Chem. 267, 21065–21071 (1992).
Rugh, R. The Mouse. Its reproduction and development 1–43 (Oxford University Press, Oxford, 1990).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 425–32 (1994).
Tartaglia, L.A. et al. Identification and expression cloning of a leptin receptor, OB-R. Cell 83, 1263–1271 (1995).
Noben-Trauth, K., Naggert, J.K., North, M.A. & Nishina, P.M. A candidate gene for the mouse mutation, tubby. Nature 380, 534–538 (1996).
Matochik, J.A., Sipos, M.L., Nyby, J.G. & Barfield, R.J. Intracranial androgenic activation of male-typical behaviors in house mice: motivation verses performance. Behav. Brain Res. 60, 141–149 (1994).
McGill, T.E. Sexual behavior in three inbred strains of mice. Behavior 19, 341–350 (1961).
Crowley, W.R. et al. Neuroendocrine control of human reproduction in the male. Recent Prog. Horm. Res. 47, 27–67 (1991).
Culler, M.D. & Negro-Vilar, A. Evidence that pulsatile follicle-stimulating hormone secretion is independent of endogenous luteinizing hormone-releasing hormone. Endocrinology 118, 609–612 (1986).
Lee, S.L. et al. Luteinizing hormone deficiency and female infertility in mice lacking the transcription factor NGF1-A (Egr-1). Science 273, 1219–1221 (1996).
Cattanach, B.M., Iddon, C.A., Charlton, H.M., Chiappa, S.A. & Fink, G. Gonadotrophin-releasing hormone deficiency in a mutant mouse with hypogonadism. Nature 269, 338–340 (1977).
Halpin, D.M., Charlton, H.M. & Faddy, M.J. Effects of gonadotrophin deficiency on follicular development in hypogonadal (hpg) mice. J. Reprod. Fertil. 78, 119–125 (1986).
Bronson, F.H. & Macmillan, B. in Pheromones and Reproduction in Mammals. 175–196 (Academic Press, New York, 1983).
Garcia de Yebenes, E., Li, S., Fournier, A., St-Pierre, S. & Pelletier, G. Regulation of proopriomelanocortin gene expression by neuropeptide Y in the rat arcuate nucleus. Brain Res. 674, 112–116 (1995).
Japon, M.A., Rubinstein, M. & Low, M.J. In situ hybridization analysis of anterior pituitary hormone gene expression during fetal mouse development. J. Histochem. Cytochem 42, 1117–1125 (1994).
Wray, S., Grant, P. & Gainer, H. Evidence that cells expressing luteinizing hormone-releasing hormone mRNA in the mouse are derived from progenitor cells in the olfactory placode. Proc. Natl. Acad. Sci. USA 86, 8132–8136 (1989).
Charlton, H.M. & Wood, M.J. Animal models for brain and pituitary gonadal disturbances. Prog. Brain Res. 93, 321–332 (1992).
Parkingson, W.L. & Weingarten, H.P. Dissociative analysis of ventromedial hypothalamic obesity syndrome. Am. J. Physiol. 259, R829–R835 (1990).
Bray, G.A., York, D.A. & Fisler, J.S. Experimental obesity: a homeostatic failure due to a defective stimulation of the sympathetic nervous system. Vitam. Horm. 45, 1–125 (1989).
Tybulewicz, V.I., Crawford, C.E., Jackson, P.K., Bronson, R.T. & Mulligan, R.C. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65, 1153–1163 (1991).
Li, E., Bestor, T.H. & Jaenisch, R. Targeted mutation of the DNA methyltransferase gene results in embryonic lethality. Cell 69, 915–926 (1992).
Love, P.E., Tremblay, M.L. & Westphal, H. Targeting of the T-cell receptor zeta-chain in embryonic stem cells: strategies for generating multiple mutations in a single gene. Proc. Natl. Acad. Sci. USA 89, 9929–9933 (1992).
Laird, P.W. et al. Simplified mammalian DNA isolation procedure. Nucl. Acids Res. 19. 4293–4295 (1991).
Sambrook, J., Fritsch, E.F. & Maniatis, T., A Laboratory Manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, 1989).
Takeuchi, T. et al. Genes encoding pancreatic polypeptide and neuropeptide Y are on human chromosomes 17 and 7. J. Clin. Invest. 77, 1038–1041 (1986).
Oates, E. & Herbert, E. 5′ sequence of porcine and rat pro-opiomelanocortin mRNA: one porcine and two rat forms. J. Biol. Chem. 259, 7421–7425 (1984).
Rehbein, M. et al. The neurohypophseal hormones vasopressin and oxytocin: Precursor structure, synthesis and regulation. Biol. Chem. Hoppe-Seyler 367, 695–704 (1986).
Kleyn, P.W. et al. Identification and characterization of the mouse obesity gene tubby: A member of a novel gene family. Cell 85, 281–290 (1996).
Lopez, F.J., Merchenthaler, I., Ching, M., Wisniewski, M.G. & Negro-Vilar, A. Galanin: a hypothalamic-hypophysiotropic hormone modulating reproductive functions. Proc. Natl. Acad. Sci. USA 88, 4508–4512 (1991).
Ikeda, M., Taga, M., Sakakibara, H., Minaguchi, H. & Vonderhaar, B.K. Detection of messenger RNA for gonadotropin-releasing hormone (GnRH) but not for GnRH receptors in mouse mammary glands. Biochem. Biophys. Res. Comm. 207, 800–806 (1995).
Wray, S., Gahwiler, B.H. & Gainer, H. Slice cultures of LHRH neurons in the presence and absence of brainstem and pituitary. Peptides 9, 1151–1175 (1988).
Kovacic, N. & Parlow, A.F. Alterations in serum FSH-LH ratios in relation to the estrous cycle, pseudopregnancy, and gonadectomy in the mouse. Endocrinology 91, 910–915 (1972).
Hogan, B., Beddington, R., Costantini, F. & Lacy, E. Manipulating the mouse embryo: A laboratory manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, 1994).
Conlon, R.A. & Rossant, J. Exogenous retinoic acid rapidly induces anterior ectopic expression of murine Hox-2 genes in vivo. Development 116, 357–368 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Good, D., Porter, F., Mahon, K. et al. Hypogonadism and obesity in mice with a targeted deletion of the Nhlh2 gene. Nat Genet 15, 397–401 (1997). https://doi.org/10.1038/ng0497-397
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0497-397
This article is cited by
-
Inactivating NHLH2 variants cause idiopathic hypogonadotropic hypogonadism and obesity in humans
Human Genetics (2022)
-
Molecular clues in the regulation of mini‐puberty involve neuronal DNA binding transcription factor NHLH2
Basic and Clinical Andrology (2021)
-
Single cell transcriptome analysis of developing arcuate nucleus neurons uncovers their key developmental regulators
Nature Communications (2019)
-
Genetically modified laboratory mice with sebaceous glands abnormalities
Cellular and Molecular Life Sciences (2016)
-
Molecular regulation of hypothalamic development and physiological functions
Molecular Neurobiology (2016)