Abstract
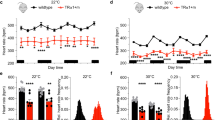
Maintenance of blood glucose by the liver is normally initiated by extracellular regulatory molecules such as glucagon and vasopressin triggering specific hepatocyte receptors to activate the cAMP or phosphoinositide signal transduction pathways, respectively. We now show that the normal ligand-receptor regulators of blood glucose in the liver can be bypassed using an adenovirus vector expressing the mouse pituitary thyrotropin releasing hormone receptor (TRHR) cDNA ectopically in rat liver in vivo. The ectopically expressed TRHR links to the phosphoinositide pathway, providing a means to regulate liver function with TRH, an extracellular ligand that does not normally affect hepatic function. Administration of TRH to these animals activates the phosphoinositide pathway, resulting in a sustained rise in blood glucose. It should be possible to use this general strategy to modulate the differentiated functions of target organs in a wide variety of pathologic states.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gammeltoft, S. & Kahn, R.C. Hormone signaling via membrane receptors. In Endocrinology. (eds DeGroot, LJ. et al.) 17–65 (W.B. Saunders Company, Philadelphia, 1995).
Gershengorn, M.C. & Perlman, J.H. Second messenger signaling pathways: phosphatidyl inositol and calcium. In Endocrinology. (eds DeGroot, LJ. et al.) 66–76 (W.B. Saunders Company, Philadelphia, 1995).
Habener, J.F. Cyclic AMP second messenger signaling pathway. In Endocrinology (eds DeGroot, L.J. et al.) 77–92 (W.B. Saunders Com pany, Philadelphia, 1995).
Walsh, D.A. & Van Patten, S.M. Multiple pathway signal transduction by the cAMP-dependent protein kinase. FASEB J. 8, 1227–1236 (1994).
Exton, J.H. Mechanisms of Hormonal Regulation of Hepatic Glucose Metabolism. Diabetes Metab. Rev. 3, 163–183 (1987).
Raymond, J.R. Hereditary and acquired defects in signaling through the hormone-receptor-G protein complex. Am. J. Physiol. 266, F163–74 (1994).
Wilkin, T.J. Receptor autoimmunity in endocrine disorders. N. Engl. J. Med. 323, 1318–1324 (1990).
Tsunoda, Y., Ca2+ signaling and crosstalk in stimulus secretion coupling. Biochim. Biophys. Acta. 1154, 105–156 (1993).
Birnbaumer, L. et al. Roles of g proteins in coupling of receptors to ionic channels and other effector systems. Crit. Rev. Biochem. Mol. Biol. 25, 225–244 (1990).
Gershengorn, M.C., Heinflink, M., Nussenzveig, D.R., Hinkle, P.M. & Falck-Pedersen, E. Thyrotropin-releasing hormone (TRH) receptor number determines the size of the TRH-responsive phosphoinositide pool. J. Biol. Chem. 269, 6779–6783 (1994).
Bringhurst, F.R. et al. Cloned, stably expressed parathyroid hormone (pth)/pth-related peptide receptors activate multiple messenger signals and biological responses in Ilc-pk1 kidney cells. Endocrinology 132, 2090–2098 (1993).
Kaneko, K.J., Gelinas, C. & Gorski, J. Activation of the silent progesterone receptor gene by ectopic expression of estrogen receptors in a rat fibroblast cell line. Biochemistry 32, 8348–8359 (1993).
Gershengorn, M.C. Thyrotropin-releasing hormone receptor: cloning and regulation of its expression. Recent Prog. Horm. Res. 48, 341–363 (1993).
Scanlon, M.F. & Hall, R. Thyrotropin-releasing hormone: basic and clinical aspects. In Endocrinology. (eds DeGroot, L.J. et al.) 192–207 (W.B. Saunders, Philadelphia, 1995).
Heinflink, M., Nussenzveig, D.R., Friedman, A.M. & Gershengorn, M.C. Thyrotropin-releasing hormone receptor activation does not elevate intracellular cyclic adenosine 3',5'-monophosphate in cells expressing high levels of receptors. J. Clin. Endocrinol. Metab. 79, 650–652 (1994).
Vale, W., Rivier, J. & Burgus, R. Synthetic TRF (Thyrotropin Releasing Factor) Ana logues: II. pGlu-N3imMe-His-Pro-NH2: A Synthetic Analogue with Specific Activity Greater then that of TRF. Endocrinology 89, 1485–1488 (1971).
Herz, J. & Gerard, R.D. Adenovirus-mediated transfer of low density lipoprotein receptor gene acutely accelerates cholesterol in normal mice. Proc. Natl. Acad. Sci. USA 90, 2812–2816 (1993).
Pilkis, S.J. & Granner, O.K., Physiology of the Regulation of Hepatic Gluco neogenesis and Glycolysis. Annu. Rev. Physiol. 54, 885–909 (1992).
Young, A.A., Cooper, G.J.S., Carlo, R., Rink, T.J. & Wang, M.-W. Response to intrave nous injections of amylin and glucagon in fasted, fed, and hypoglycemic rats. Am. J. Physiol. 264, E943–E950 (1993).
Jaffe, H.A. et al. Adenovirus-mediated in vivo gene transfer and expression in normal rat liver. Nature Genet. 1, 372–378 (1992).
Li, Q., Kay, M.A., Finegold, M., Startford-Perriaudet, L.D. & Woo, S.L.C. Assessment of recombinant adenoviral vectors for hepatic gene therapy. Hum. Gene Ther. 4, 403–409 (1993).
Milano, C.A. et al. Enhanced myocardial function in transgenic mice overexpressing the β2-adrenergic receptor. Science 264, 582–586 (1994).
Milano, C.A. et al. Myocardial expression of a constitutively active alpha 1 b-adrenergic receptor in transgenic mice induces cardiac hypertrophy. Proc. Natl. Acad. Sci. USA 91, 10109–10113 (1994).
Bond, R.A. et al. Physiological effects of inverse agonists in transgenic mice with myo cardial overexpression of the beta 2-adrenoceptor. Nature. 374, 272–276 (1995).
Spencer, D.M., Wandless, T.J., Schreiber, S.L. & Crabtree, G.R., Transduction with Synthetic Ligands. Science 262, 1019–1024 (1993).
Grossman, M. et al. Successful ex vivo gene therapy directed to liver in a patient with familial hypercholesterolaemia. Nature Genet. 6, 335–341 (1994).
Kozarsky, K.F., McKinley, D.R., Austin, L.L., Raper, S.E., Stratford-Perricaudet, L.D. & Wilson, J.M. In vivo correction of low density lipoprotein receptor deficiency in the watanabe heritable hyperlipidemic rabbit with recombinant adenoviruses. J. Biol. Chem. 269, 13695–13702 (1994).
Hems, D.A., Whitton, P.O. & Ma, G.Y. Metabolic actions of vasopressin, glucagon and adrenalin in the intact rat. Biochim. Biophys. Acta. 411, 155–164 (1975).
Buwalda, B., Nyakas, C., Koolhaas, J.M. & Bohus, B. Neuroendocrine and behavioral effects of vasopressin in resting and mild stress conditions. Physiol. Behav. 54, 947–953 (1993).
Lefkowitz, R.J. & Premont, R.T. Diseased G protein-coupled receptors. J. Clin. Invest. 92, 2089 (1993).
Spiegel, A.M., Weinstein, L.S. & Shenker, A. Abnormalities in g protein-coupled signal transduction pathways in human disease. J. Clin. Invest. 92, 1119–1125 (1993).
Naparstek, Y. & Plotz, P.H. The role of autoantibodies in autoimmune disease. Annu. Rev. Immunol. 11, 79–104 (1993).
Rogers, S.W. et al. Autoantibodies to glutamate receptor glur3 in rasmussen's encephali tis. Science 265, 648–651 (1994).
Falck-Pedersen, E., Heinflink, M., Alvira, M., Nussenzveig, D.R. & Gershengorn, M.C. Expression of thyrotropin-releasing hormone receptors by adenovirus-mediated gene transfer reveals that thyrotropin releasing hormone desensitization is cell specific. Mol. Pharmacol. 45, 684–689 (1994).
Hersh, J., Crystal, R.G. & Bewig, B. Modulation of gene expression after replication-deficient, recombinant adenovirus-mediated gene transfer by the product of a second adenovirus vector. Gene Therapy 2, 124–131 (1995).
Rosenfeld, M.A. et al. Adenovirus-mediated transfer of a recombinant alpha 1 - antitryp sin gene to the lung epithelium in vivo. Science 252, 431–434 (1991).
Rosenfeld, M.A. et al. In vivo transfer of the human cystic fibrosis transmembrane con ductance regulator gene to the airway epithelium. Cell 68, 143–155 (1992).
Gershengorn, M.C. Bihormonal regulation of the thyrotropin-releasing hormone receptor in mouse pituitary thyrotropic tumor cells in culture. J. Clin. Invest 62, 937–943 (1978).
Imai, A. & Gershengorn, M.C. Measurement of lipid turnover in response to thyrotro pin-releasing hormone. Meth. Enzymol. 141, 100–101 (1987).
Mitruka, B.M., Rawnsley, H.M. In Clinical Biochemical and Hematological Reference Values in Normal Experimental Animals. 117–246 (Masson, New York, 1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolff, G., Mastrangeli, A., Heinflink, M. et al. Ectopic expression of thyrotropin releasing hormone (TRH) receptors in liver modulates organ function to regulate blood glucose by TRH. Nat Genet 12, 274–279 (1996). https://doi.org/10.1038/ng0396-274
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0396-274
This article is cited by
-
Coexpression of p21WAF1/CIP1 in adenovirus vector transfected human primary hepatocytes prevents apoptosis resulting in improved transgene expression
Gene Therapy (2003)
-
Adenovirus-mediated gene transfer of P16INK4/CDKN2 into bax-negative colon cancer cells induces apoptosis and tumor regression in vivo
Cancer Gene Therapy (2002)
-
Complex-trait genetics: emergence of multivariate strategies
Nature Reviews Neuroscience (2002)