Abstract
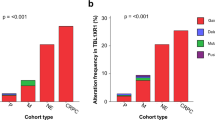
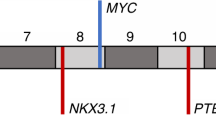
The Mxi1 protein negatively regulates Myc oncoprotein activity and thus potentially serves a tumour suppressor function. MXI1 maps to chromosome 10q24–q25, a region that is deleted in some cases of prostate cancer. We have detected mutations in the retained MXI1 alleles in four primary prostate tumours with 10q24–q25 deletions. Two tumours contained inactivating mutations, whereas two others contained the identical missense mutation. Fluorescence in situ hybridization also demonstrated loss of one MXI1 allele in an additional tumour lacking chromosome 10 abnormalities. MXI1 thus displays allelic loss and mutation in some cases of prostate cancer that may contribute to the pathogenesis or neoplastic evolution of this common malignancy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dalla-Favera, R., Martirotti, S., Galio, R.C., Erickson, J. & Croce, C.M. Translocation and rearrangement of the c-myc oncogene locus in human undifferentiated B-cell lymphomas. Science. 219, 963–965 (1983).
Schwab, M. et al. Enhanced expression of the human gene N-myc consequent to amplification of DNA may contribute to malignant progression of neuroWastoma. Proc. natn. Acad. Sci. U.S.A. 81, 4940–4944 (1984).
Nau, M. et al. L-myc, a new myc-related gene amplified and expressed in human small cell lung cancer. Nature. 318, 69–73 (1985).
Adams, J.M. et al. The c-myc oncogene driven by Immunoglobulin enhancer induces lymphoid malignancy in transgenlc mice. Nature 318, 533–538 (1985).
Murre, C., McCaw, P.S. & Baltimore, D. A new DMA binding and dimerizatlon motif in immunoglobulin, enhancer binding, daughterless, MyoD, and myc proteins. Cell 56, 777–783 (1989).
Murrem, C. et al. Interactions between heterotogous helix-loop-helix proteins generate complexes that bind specifically to a common DNA sequence. Cell 58, 537–544 (1989).
Olson, E.N. MyoD family: a paradigm for development? Genes Dev. 4, 1454–1461 (1990).
Davis, R.L., Cheng, P.F., Lassar, A.B. & Weintraub, H., MyoD DNA binding domain contains a recognition code for muscle-specific gene expression. Cell 60, 733–746 (1990).
Lassar, A.B. et al. Functional activity of myogenic HLH protreins requires hetero-oligomerization with E12/E47-like proteins In vivo. Cell 66, 305–315 (1991).
Weintraub, H. et al. The myoD gene family: nodal point during specification of the muscle cell lineage. Science 251, 761–766 (1991).
Kato, G.J., Barrett, J., Villa-Garcia, M. & Dang, C. An amino terminal c-myc domain required for neoplastic transformation activates transcription. Molec. Cell. Biol. 10, 5914–6920 (1990).
Blackwood, E.M. & Eisenmann, R.N. Max: a helix-loop-helix-zipper protein that forms a sequence-specific DNA binding complex with myc. Science 251, 1211–1217 (1991).
Prendergast, G.C., Lawe, D. & Ziff, E.B. Association of myn, the murine homolog of max, with c-myc stimulates methylatton-sensitive DNA binding and ras cotransormation. Cell 66, 395–407 (1991).
Ayer, D.E., Kretzner, L. & Elsenmann, R.N. Mad: a heteradimeric partner for Max that antagonizes myc transcriptlonal activity. Cell 72, 211–222 (1993).
Zervos, A., Gyuris, J. & Brent, R., Mxl1, a protein that specifically interacts with max to bind myc-max recognition sites. Cell 72, 223–232 (1993).
Smith, M.J., Charron-Prochownik, D.C. & Prochownik, E.V. The leucine zipper of c-myc is required for the full inhibition of erythroleukemia differentiation. Molec. Cell. Biol. 10, 5333–5339 (1990).
Dang, C.V. et al. Intracellular leucine zipper interactions suggest c-myc hetero-oligomerization. Molec. Cell. Biol. 11, 954–962 (1991).
Amati, B. et al. Transcriptional activation by the human c-Myc oncoprotein in yeast requires interactions with Max. Nature 359, 423–426 (1992).
Kretzner, L., Blackwood, E.M. & Eisenman, R.N. Myc and Max proteins possess distinct transcriptional activities. Nature 359, 426–429 (1992).
Amati, B. et al. Oncogenic activity of the c-myc protein requires oligomerization with max. Cell 72, 233–245 (1993).
Amin, C., Wagner, A.J. & Hay, N. Sequence-specific transcriptional activation by Myc and repression by Max. Molec. Cell. Biol. 13, 383–390 (1993).
Gu, W., Chechova, K. & Dalla-Favera, R. Opposite regulation of gene transcription and cell proliferation by c-Myc and Max. Proc. natn. Acad. Sci. U.S.A. 90, 2935 (1993).
Lahoz, E.G., Xu, L., Schreiber, A. & DePinho, R. Suppression of Myc, but not Ela, transformation activity by Max-associated proteins, Mad and Mxi1. Proc. natn. Acad. Sci. U.S.A. 91, 5503–5507 (1994).
Edelhoff, S. et al. Mapping of two genes encoding members of a distinct subfamily of MAX interacting proteins: MAD to human chromosome 2 and mouse chromosome 6, and MXI1 to human chromosome 10 and mouse chromosome 19. Oncogene 9, 665 (1994).
Shapiro, D.N. et al. Assignment of the human MAD and MXI1 genes to chromosomes 2p12–p13 and 10q24–q25. Genomics 23, 282–285 (1994).
Sreekantaiah, C., Baer, M.R., Sole, F., Preisier, H.F. & Sandberg, A.A. Translocation (2;7) (p13;q36) in a case of acute non-lymphocytic leukemia evolving from a myelodysplastic syndrome. Cancer Genet. Cytogenet. 35, 199–204 (1988).
Fleischman, E.W. et al. Chromsomal characteristics of malignant lymphoma. Hum. Genet. 82, 343–348 (1989).
Heisler, C.H., Phillip, P. & Hansen, M.M. B-cell chronic lymphocytic leukaemia: clonal chromosome abnormalities and prognosis In 89 cases. Eur. J. Haematol. 43, 397–403 (1990).
Yoffe, G., Howard-eebles, P.N., Smith, G., Tucker, P.W., Buchanan, G.R. Childhood chronic lymphocytic leukemia with t (2;14) translocation. J. Pediatr. 116, 114–117 (1990).
Fults, D. & Pedone, C. Deletion mapping of the long arm of chromosome 10 In glioblastoma multiforme. Genes Chrom. Cancer. 7, 173–177 (1993).
Bird, M.L., Ueshima, Y., Rowley, J.D., Haren, J.M., Vardiman, J.W. Chromosome abnormalities In B cell chronic lymphocytic leukemia and their clinical correlations. Leukemia 3, 182–191 (1989).
Atkin, N.B. & Baker, M.C. Chromosome study of five cancers in prostate. Hum. Genet. 70, 359–364 (1985).
Lundgren, R. et al. Cytogenetic analysis of 57 primary prostatic adenocarcinomas. Genes Chrom. Cancer. 4, 16–24 (1992).
Brothman, A.R., Peehl, D.M., Patel, A.M. & McNeal, J.E. Frequency and pattern of karyotypic abnormalities in human prostatic cancer. Cancer Res. 50, 3795–3803 (1990).
Arps, S. et al. Cytogenetic survey of 32 cancers of the prostate. Cancer Genet Cytogenet. 66, 93–99 (1993).
Weinberg, R.A. Tumor suppressor genes. Science 254, 1138–1146 (1992).
Levine, A.J. The tumor suppressor genes. Annu. Rev. Biochem. 62, 623–651 (1993).
Kalllo, O., Syrjanen, S., Tervahuauta, S. & Syrjanen, K. A simple method for isolation of DNA from formalin-fixed pafaffin-embedded samples for PCR. J. Virol. Meth. 35, 39–47 (1991).
Padgett, R.A., Grabowski, P.J., Konaraka, M.M., Seiber, S. & Sharp, P.A. Splicing of messenger RNA precursors. Annu. Rev. Biochem. 55, 1119–1150 (1986).
Kazazian, H.H. & Boehm, C.D. Molecular basis and prenatal diagnosis of β-thalassemia. Blood. 72, 1107–1116 (1988).
Saiki, R.K. et al. Primer-directed enzymatic amplifications of DNA with a thermostable DNA polymerase. Science 230, 487–491 (1988).
Prochownik, E.V. & Van Antwerp, M.E. Differential patterns of DNA binding by myc and max proteins. Proc. natn. Acad. Sci. U.S.A. 90, 960–964 (1993).
Epstein, I. in Prostate Biopsy Interpretation. Biopsy Interpretation Series. (ed. Silverberg, S.G ) 39–129 (Raven Press, New York, 1989).
Sidransky, D. et al. Clonal expansion of p53 mutant cells in association with brain tumor progression. Nature 355, 646–847 (1992).
Marshall, R. et al. Rearrangement and expression of p53 in the chronic phase and blast crisis of chronic myetogenous leukemia. Blood 75, 180–189 (1990).
Nakai, H., Misawa, S., Toguchida, J., Yandell, D.W. & Ishizaki, K. Frequent p53 gene mutations in blast crisis of chronic myetogenous leukemia, especially In myetoid crisis harboring loss of chromsome 17p. Cancer Res. 52, 6588–6593 (1992).
Baker, S.J. et al. p53 gene mutations occur in combination with 17p allelic deletions as late events in colorectal tumorigenesis. Cancer Res. 50, 7717–7722 (1990).
Frankel, R.H., Bayona, W., Koslow, M. & Newcomb, E.W. p53 mutations in human malingnant glomas: comparison of loss of heterzygosity with mutation frequency. Cancer Res. 52, 1427–1433 (1992).
Brennan, T.J. & Olson, E.N. Myogenin resides In the nucleus and acquires high affinity for a conserved enhancer element on heterodemerizatton. Genes Dev. 4, 582–595 (1990).
Dang, C.V., Dolde, C., Gillison, M.L. & Kato, G.J. Discrimination between related DNA sites by a single amino acid residue of Myc-related basic helix-loop-helix protein. Proc. natn. Acad. Sci. U.S.A. 88, 599–601 (1992).
Van Antwerp, M.E., Chen, D.G., Chang, C. & Prochownik, E.V. A point mutation in the MyoD basic domain imparts c-myc-like properties. Proc. natn. Acad. Sci. U.S.A. 88, 9010–9014 (1992).
Kunket, T.A. Rapid and efficient site-specific mutagenesis without phenotypic selections. Proc. natn. Acad. Sci. U.S.A. 82, 488–492 (1985).
Jones, E., Zhu, X.L., Rohr, L.R., Stephenson, R.A. & Brothman, A.R. Aneusomy of chromosomes 7 and 17 detected by FISH in prostate cancer and the effects of selection in vitro. Genes Chrom. Cancer. 11, 163–170 (1994).
Zervos, A., Gyuris, J. & Brent, R., Errata. Cell. 79, 389 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eagle, L., Yin, X., Brothman, A. et al. Mutation of the MXI1 gene in prostate cancer. Nat Genet 9, 249–255 (1995). https://doi.org/10.1038/ng0395-249
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0395-249
This article is cited by
-
A cell cycle centric view of tumour dormancy
British Journal of Cancer (2023)
-
DNA methylation-based chromatin compartments and ChIP-seq profiles reveal transcriptional drivers of prostate carcinogenesis
Genome Medicine (2017)