Abstract
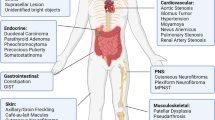
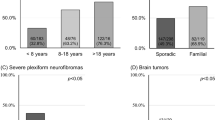
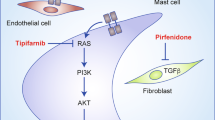
Neurofibromatosis type 1 (NF1) is a common autosomal dominant disorder characterized by progressive and variable involvement of tissues predominantly derived from the neural crest and a predisposition toward malignancies. The NF1 gene encodes neurofibromin, a GTPase–activating protein containing a GAP–related domain (NF1–GRD) that is capable of down–regulating ras by stimulating its intrinsic GTPase activity. We report a homozygous deletion of most of NF1 in one of eight malignant melanoma cell lines leading to loss of detectable mRNA and protein, as well as the apparent absence of protein and mRNA in another melanoma. This data suggests that NF1 can function as a tumour suppressor gene in the development or progression of malignant melanoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Martuza, R.L. et al. Melanin macroglobules as a cellular marker of neurofibromatosis: a quantitative study. J. Invest. Derm. 85, 347–50 (1985).
Riccardi, V.M. & Eichner, J.E. Neurofibromatosis: Phenotype, Natural History and Pathogenesis (John Hopkins, Baltimore, 1986).
Bolande, R.P. Neurofibromatosis - the quintessential neurocristopathy: pathogenetic concepts and relationships. Adv. Neurol. 29, 67–75 (1981).
Wallace, M.R. et al. Type 1 neurofibromatosis gene: Identification of a large transcript disrupted in three NF1 patients. Science 249, 181–189 (1990).
Gutmann, D.H., Wood, D.L. & Collins, F.S. Identification of the neurofibromatosis type 1 gene product. Proc. natn. Acad. Sci. U.S.A. 88, 9658–9662 (1991).
DeClue, J.E., Cohen, B.D. & Lowy, D.R. Identification and characterization of the neurofibromatosis type 1 protein product. Proc. natn. Acad. Sci. U.S.A. 88, 9914–9918 (1991).
Daston, M.M. et al. The protein product of the neurofibromatosis type 1 gene is expressed at highest abundance in neurons, Schwann cells, and oligodendrocytes. Neuron 8, 415–28 (1992).
Xu, G. et al. The neurofibromatosis type 1 gene encodes a protein related to GAP. Cell 62, 599–608 (1990).
Ballester, R. et al. The NF1 locus encodes a protein functionally related to mammalian GAP and yeast IRA proteins. Cell 63, 851–559 (1990).
Martin, G.A. et al. The GAP-related domain of the neurofibromatosis type 1 gene product interacts with ras p21. Cell 63, 843–849 (1990).
Viskochil, D. et al. Deletions and atranslocation breakpoint interrupt a cloned gene at the neurofibromatosis type 1 locus. Cell 62, 187–192 (1990).
Upadhyaya, M. et al. A 90 kb DNA deletion associated with neurofibromatosis type 1. J. med. Genet. 27, 738–741 (1990).
Cawthon, R.M. et al. A major segment of the neurofibromatosis type 1 gene: cDNA sequence, gene structure, and point mutations. Cell 62, 193–201 (1990).
Wallace, M.R. et al. A de novo Alu insertion results in neurofibromatosis type 1. Nature 353, 864–866 (1991).
Li, Y. et al. Somatic mutations in the neurofibromatosis 1 gene in human tumors. Cell 69, 275–281 (1992).
Herlyn, M. et al. Biology of tumor progression in human melanocytes. Lab Invest 56, 461–474 (1987).
Dracopoli, N.C., Houghton, A.N. & Old, L.J. Loss of polymorphic restriction fragments in malignant melanoma: implications for tumor heterogeneity. Proc. natn. Acad. Sci. U.S.A. 82, 1470–1474 (1985).
Dracopoli, N.C., Alhadeff, B., Houghton, A.N. & Old, L.J. Loss of heterozygosity at autosomal and X-linked loci during tumor progression in a patient with melanoma. Cancer Res. 47, 3995–4000 (1987).
Fountain, J.W. et al. Homozygous deletions within human chromosome band 9p21 in melanoma. Proc. natn. Acad. Sci. U.S.A. 89, 10557–10561 (1992).
Dracopoli, N.C. et al. Loss of alleles from distal short arm of chromosome 1 occurs late in melanoma tumor progression. Proc. natn. Acad. Sci. U.S.A. 86, 4614–4618 (1989).
Millikin, D., Meese, E., Vogelstein, B., Witkowski, C. & Trent, J. Loss of heterozygosity for loci on the long arm of chromosome 6 in human malignant melanoma. Cancer Res. 51, 5449–5453 (1991).
Bean, M.A. et al. Cell-mediated cytotoxicity for bladder carcinoma: evaluation of a workshop. Cancer Res. 35, 2902–2913 (1975).
Carey, T.E., Takahashi, T., Resnick, L.A., Oettgen, H.F. & Old, L.J. Cell surface antigens of human malignant melanoma: mixed hemadsorption assays for humoral immunity to cultured autologous melanoma cells. J. Proc. natn. Acad. Sci. U.S.A. 73, 3278–3282 (1976).
Fogh, J., Fogh, J.M. & Orfeo, T.J. One hundred and twenty-seven cultured human tumor cell lines producing tumors in nude mice. J. natn. Cancer Inst. 59, 221–226 (1977).
Albino, A.P., Le Strange, R., Oliff, A.I., Furth, M.E. & Old, L.J. Transforming ras genes from human melanoma: a manifestation of tumour heterogeneity? Nature 308, 69–72 (1984).
Marchuk, D.A. et al. cDNA cloning of the type 1 neurofibromatosis gene: complete sequence of the NF1 gene. Genomics 11, 931–940 (1991).
Wallace, M.R., Fountain, J.W., Brereton, A.M. & Collins, F.S. Direct construction of a chromosome-specific Notl linking library from flow-sorted chromosomes. Nucl. Acids Res. 4, 1665–1677 (1989).
White, R. et al. Tightly linked markers for the neurofibromatosis type 1 gene. Genomics 1, 364–67 (1987).
Sardet, C., Franchi, A. & Pouysségur, J. Molecular cloning, primary structure, and expression of the human growth factor-activatable Na+/H+ antiporter. Cell 56, 271–280 (1989).
Becher, R., Gibas, Z., Karakousis, C. & Sandberg, A.A. Nonrandom chromosome changes in malignant melanoma. Cancer Res. 43, 5010–5016 (1983).
Friend, S.H. et al. A human DNA segment with properties of the gene that predisposes to retinoblastoma and osteosarcoma. Nature 323, 643–646 (1986).
Call, K.M. et al. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome 11 Wilms' tumor locus. Cell 60, 509–520 (1990).
Fearon, E.R. & Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 61, 759–767 (1990).
Albino, A.P. et al. Analysis of ras oncogenes in malignant melanoma and precursor lesions: correlation of point mutations with differentiation phenotype. Oncogene 4, 1363–1374 (1989).
Crowe, F.W., Schull, W.T. & Neel, J.F. Clinical, Pathological, and Genetic Study of Multiple Neurofibromatosis (Charles C. Thomas, Springfield, Illinois, 1956).
T'Ang, A., Varley, J.M., Chakraborty, S., Murphree, A.L. & Fung, Y.K. Structural rearrangement of the retinoblastoma gene in human breast carcinoma. Science 242, 263–266 (1988).
Sambrook, J., Fritsch, E.F. & Maniatis, T. in Molecular Cloning. A Laboratory Manual 2nd ed. (Cold Spring Harbor Press, New York, 1989).
Harlow, E. & Lane, D. Antibodies: A Laboratory Manual, 421–470 (Cold Spring Harbor Press, New York, 1988).
Basu, T.N. et al. Aberrant regulation of ras proteins in malignant tumour cells from type 1 neurofibromatosis patients. Nature 356, 713–715 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Andersen, L., Fountain, J., Gutmann, D. et al. Mutations in the neurofibromatosis 1 gene in sporadic malignant melanoma cell lines. Nat Genet 3, 118–121 (1993). https://doi.org/10.1038/ng0293-118
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0293-118
This article is cited by
-
Diverse mechanisms activate the PI 3-kinase/mTOR pathway in melanomas: implications for the use of PI 3-kinase inhibitors to overcome resistance to inhibitors of BRAF and MEK
BMC Cancer (2021)
-
Signal pathways of melanoma and targeted therapy
Signal Transduction and Targeted Therapy (2021)
-
Synergistic melanoma cell death mediated by inhibition of both MCL1 and BCL2 in high-risk tumors driven by NF1/PTEN loss
Oncogene (2021)
-
Whole-genome landscapes of major melanoma subtypes
Nature (2017)
-
Molecular characterisation of cutaneous melanoma: creating a framework for targeted and immune therapies
British Journal of Cancer (2016)