Abstract
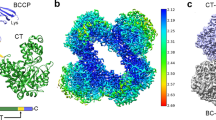
The structure of the holo enzyme shows that the four chemically identical sub-units are arranged with almost perfect 222 symmetry. There are indications, however, that the active centre regions might only be related in pairs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Velick, S. F., and Furfine, C., in The Enzymes (edit. by Boyer, P. D., Lardy, H., and Myrbäck, K.), 7, 243 (Academic Press, New York, 1963).
Allison, W. S., and Kaplan, N. O., J. Biol. Chem., 239, 2140 (1964).
Perham, R. N., and Harris, J. I., J. Mol. Biol., 7, 316 (1963).
Jones, G. M. T., and Harris, J. I., FEBS Lett., 22, 185 (1972).
Watson, H. C., and Banaszak, L. J., Nature, 204, 918 (1964).
Wassarman, P. M., and Watson, H. C., FEBS Lett., 18, 51 (1970).
Campbell, J. W., Duée, E., Hodgson, G., Mercer, W. D., Stammers, D. K., Wendell, P. L., Muirhead, H., and Watson, H. C., Cold Spring Harbor Symp. Quant. Biol., 36, 165 (1971).
Baranowski, T., and Wolny, M., Acta Biol. Med. Germ., 11, 651 (1963).
Racker, E., and Krimsky, I., Nature, 169, 1043 (1952).
Fox, J. B., and Dandliker, W. B., J. Biol. Chem., 221, 1005 (1956).
Gorjunov, A. I., and Andreeva, N. S., Molekul. Biolog., 1, 261 (1967)
Mathews, B. W., J. Mol. Biol., 33, 491 (1968).
Bokhoven, C., Schoone, J. C., and Bijvoet, J. B., Acta Cryst., 4, 275 (1951).
Green, D. W., Ingram, V. M., and Perutz, M. F., Proc. Roy. Soc., A, 225, 287 (1954).
Kendrew, J. C., Bodo, G., Dintzis, H. M., Parrish, R. G., Wyckoff, H. W., and Phillips, D. C., Nature, 181, 662 (1958).
Watson, H. C., Shotton, D. M., Cox, J. M., and Muirhead, H., Nature, 225, 806 (1970).
Lipscomb, W. N., Coppola, J. C., Hartsuck, J. A., Ludwig, M. L., Muirhead, H., Searl, J., and Steitz, J. A., J. Mol. Biol., 19, 423 (1966).
Blow, D. M., and Crick, F. H. C., Acta Cryst., 12, 794 (1959).
Wolny, M., Acta Biochim. Polon., 15, 137 (1968).
Adams, M. J., McPherson, A., Rossmann, M. G., Schevitz, R. W., and Wonacott, A. J., J. Mol. Biol., 51, 31 (1970).
Adams, M. J., Haas, D. J., Jeffery, B. A., McPherson, A., Mermall, H. L., Rossmann, M. G., Schevitz, R. W., and Wonacott, A. J., J. Mol. Biol., 41, 159 (1969).
Adams, M. J., Ford, G. C., Koekoek, R., Lentz, P. J., McPherson, A., Rossmann, M. G., Smiley, I. E., Schevitz, R. W., and Wonacott, A. J., Nature, 227, 1098 (1970).
Tsernoglou, D., Hill, E., and Banaszak, L. J., J. Mol. Biol. (in the press).
Conway, A., and Koshland, D. E., Biochemistry, 7, 4011 (1968).
Trentham, D. R., Biochem. J., 109, 603 (1968).
Bernhard, S. A., and MacQuarrie, R. A. (in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WATSON, H., DUÉE, E. & MERCER, W. Low Resolution Structure of Glyceraldehyde 3-Phosphate Dehydrogenase. Nature New Biology 240, 130–133 (1972). https://doi.org/10.1038/newbio240130a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/newbio240130a0