Abstract
Significant advances in the biology and treatment of Hodgkin lymphoma (HL) have been accomplished over the past decades. In a landmark study, DeVita and colleagues showed that half of patients with advanced-stage HL experienced long-term disease-free survival following treatment with a four-drug chemotherapy regimen. Subsequent reports and randomized clinical trials conducted over the past 40 years have defined prognostic categories and refined the treatment options for patients with early-stage and advanced-stage HL. New treatment concepts and regimens have continued to increase the cure rate of HL, while other analyses have documented the acute and long-term morbid and potentially fatal side effects of HL therapy. Increased knowledge of HL biology has been gained, in particular, much has been learnt about the genetic and phenotypic characteristics of malignant cells and the varied oncogenic signaling pathways involved in HL. Continued translational research is needed to improve the long-term survival and to lessen the toxicities associated with therapy. Furthermore, continued clinical-trial involvement by oncologists and patients is imperative to further advance the field of HL.
Key Points
-
Preclinical translational studies have demonstrated that the Reed-Sternberg cells of classical Hodgkin lymphoma (HL) and the lymphocytic and histiocytic cells of nodular lymphocyte predominant Hodgkin lymphoma (NLPHL) are derived from B-cell origins and that several anti-apoptotic signaling pathways are involved in the survival of these malignant cells
-
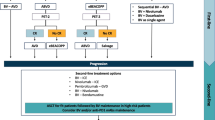
Patients with stage I–II, (i.e. 'early stage') HL should usually be treated with combined-modality therapy comprising chemotherapy (ABVD) followed by involved-field radiotherapy
-
Chemotherapy alone is a treatment option for early-stage HL, especially where the risks of acute and/or long-term radiotherapy are deemed unacceptable
-
At present, ABVD is the internationally accepted standard regimen for advanced-stage HL, and is the regimen against which all experimental combinations should be tested; other chemotherapy programs such as Stanford V, and the more-intensive regimen, BEACOPP, represent treatment options for HL and data from randomized trials continues to be analyzed
-
Acute treatment-related toxicities (i.e. bleomycin-associated lung toxicity) and chronic treatment-related toxicities (secondary cancers, cardiovascular disease) should be considered when therapy choices are made, especially when treating HL in elderly patients
-
For advancement in the outcome of HL, it is imperative that international collaborative efforts continue and that oncologists offer all patients with HL the opportunity to participate in clinical trials
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
DeVita Jr VT and Carbone PP (1967) Treatment of Hodgkin's disease. Med Ann Dist Columbia 36: 232–234
Re D et al. (2005) From Hodgkin disease to Hodgkin lymphoma: biologic insights and therapeutic potential. Blood 105: 4553–4560
Kuppers R et al. (1994) Hodgkin disease: Hodgkin and Reed-Sternberg cells picked from histological sections show clonal immunoglobulin gene rearrangements and appear to be derived from B cells at various stages of development. Proc Natl Acad Sci USA 91: 10962–10966
Marafioti T et al. (2000) Hodgkin and Reed-Sternberg cells represent an expansion of a single clone originating from a germinal center B-cell with functional immunoglobulin gene rearrangements but defective immunoglobulin transcription. Blood 95: 1443–1450
Schwering I et al. (2003) Loss of the B-lineage-specific gene expression program in Hodgkin and Reed-Sternberg cells of Hodgkin lymphoma. Blood 101: 1505–1512
Re D et al. (2001) Oct-2 and Bob-1 deficiency in Hodgkin and Reed Sternberg cells. Cancer Res 61: 2080–2084
Stein H et al. (2001) Down-regulation of BOB.1/OBF.1 and Oct2 in classical Hodgkin disease but not in lymphocyte predominant Hodgkin disease correlates with immunoglobulin transcription. Blood 97: 496–501
Marafioti T et al. (1997) Origin of nodular lymphocyte-predominant Hodgkin's disease from a clonal expansion of highly mutated germinal-center B cells. N Engl J Med 337: 453–458
Ohno T et al. (1997) Clonality in nodular lymphocyte-predominant Hodgkin's disease. N Engl J Med 337: 459–465
Caldwell RG et al. (1998) Epstein-Barr virus LMP2A drives B cell development and survival in the absence of normal B cell receptor signals. Immunity 9: 405–411
Dutton A et al. (2004) Expression of the cellular FLICE-inhibitory protein (c-FLIP) protects Hodgkin's lymphoma cells from autonomous Fas-mediated death. Proc Natl Acad Sci USA 101: 6611–6616
Thomas RK et al. (2002) Constitutive expression of c-FLIP in Hodgkin and Reed-Sternberg cells. Am J Pathol 160: 1521–1528
Kashkar H et al. (2003) XIAP-mediated caspase inhibition in Hodgkin's lymphoma-derived B cells. J Exp Med 198: 341–347
Sanchez-Aguilera A et al. (2006) Tumor microenvironment and mitotic checkpoint are key factors in the outcome of classic Hodgkin lymphoma. Blood 108: 662–668
Duhmke E et al. (2001) Low-dose radiation is sufficient for the noninvolved extended-field treatment in favorable early-stage Hodgkin's disease: long-term results of a randomized trial of radiotherapy alone. J Clin Oncol 19: 2905–2914
Engert A et al. (2003) Involved-field radiotherapy is equally effective and less toxic compared with extended-field radiotherapy after four cycles of chemotherapy in patients with early-stage unfavorable Hodgkin's lymphoma: results of the HD8 trial of the German Hodgkin's Lymphoma Study Group. J Clin Oncol 21: 3601–3608
Noordijk EM et al. (2006) Combined-modality therapy for clinical stage I or II Hodgkin's lymphoma: long-term results of the European Organisation for Research and Treatment of Cancer H7 randomized controlled trials. J Clin Oncol 24: 3128–3135
Meyer RM et al. (2005) Randomized comparison of ABVD chemotherapy with a strategy that includes radiation therapy in patients with limited-stage Hodgkin's lymphoma: National Cancer Institute of Canada Clinical Trials Group and the Eastern Cooperative Oncology Group. J Clin Oncol 23: 4634–4642
Specht L et al. (1998) Influence of more extensive radiotherapy and adjuvant chemotherapy on long-term outcome of early-stage Hodgkin's disease: a meta-analysis of 23 randomized trials involving 3,888 patients. International Hodgkin's Disease Collaborative Group. J Clin Oncol 16: 830–843
Hagenbeek A et al. (2000) Three cycles of MOPP/ABV hybrid and involved-field irradiation is more effective than subtotal nodal irradiation in favorable supradiaphragmatic clinical stages I-II Hodgkin's disease: preliminary results of the EORTC-GELA H8-F randomized trial in 543 patients [abstract #575]. Blood 96
Noordijk EM et al. (2005) First results of the EORTC-GELA H9 randomized trials: the H9-F trial (comparing 3 radiation dose levels) and H9-U trial (comparing 3 chemotherapy schemes) in patients with favorable or unfavorable early stage Hodgkin's lymphoma. J Clin Oncol 23: 561S
Engert A et al. (2005) Combined modality treatment of two or four cycles of ABVD followed by involved field radiotherapy in the treatment of patients with early stage Hodgkin's lymphoma: update interim analysis of the randomised HD10 Study of the German Hodgkin Study Group (GHSG) [abstract #2673]. Blood 106
Ferme C et al. (2005) Four ABVD and involved-field radiotherapy in unfavorable supradiaphragmatic clinical Stages (CS) I-II Hodgkin's lymphoma (HL): preliminary results of the EORTC-GELA H9-U trial [abstract #813]. Blood 106
Ferme C et al. (2000) MOPP/ABV hybrid and irradiation in unfavorable supradiaphragmatic clinical stages I–II Hodgkin's disease: comparison of three treatment modalities. Preliminary results of the EORTC-GELA H8-U randomized trial in 995 patients [abstract #576]. Blood 96
Klimm BC et al. (2005) Comparison of BEACOPP and ABVD chemotherapy in intermediate stage Hodgkin's lymphoma: results of the fourth interim analysis of the HD 11 trial of the GHSG [abstract #6507]. J Clin Oncol 23
Press OW et al. (2001) Phase III randomized intergroup trial of subtotal lymphoid irradiation versus doxorubicin, vinblastine, and subtotal lymphoid irradiation for stage IA to IIA Hodgkin's disease. J Clin Oncol 19: 4238–4244
Straus DJ et al. (2004) Results of a prospective randomized clinical trial of doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) followed by radiation therapy (RT) versus ABVD alone for stages I, II, and IIIA nonbulky Hodgkin disease. Blood 104: 3483–3489
Dores GM et al. (2002) Second malignant neoplasms among long-term survivors of Hodgkin's disease: a population-based evaluation over 25 years. J Clin Oncol 20: 3484–3494
Swerdlow AJ et al. (2000) Risk of second malignancy after Hodgkin's disease in a collaborative British cohort: the relation to age at treatment. J Clin Oncol 18: 498–509
Travis LB et al. (2005) Cumulative absolute breast cancer risk for young women treated for Hodgkin lymphoma. J Natl Cancer Inst 97: 1428–1437
Ng AK et al. (2002) Second malignancy after Hodgkin disease treated with radiation therapy with or without chemotherapy: long-term risks and risk factors. Blood 100: 1989–1996
van Leeuwen FE et al. (2000) Long-term risk of second malignancy in survivors of Hodgkin's disease treated during adolescence or young adulthood. J Clin Oncol 18: 487–497
Adams MJ et al. (2004) Cardiovascular status in long-term survivors of Hodgkin's disease treated with chest radiotherapy. J Clin Oncol 22: 3139–3148
Bowers DC et al. (2005) Stroke as a late treatment effect of Hodgkin's disease: a report from the Childhood Cancer Survivor Study. J Clin Oncol 23: 6508–6515
Aleman BMP et al. (2007) Late cardiotoxicity after treatment for Hodgkin lymphoma. Blood 109: 1878–1886
Swerdlow AJ et al. (2007) Myocardial infarction mortality risk after treatment for Hodgkin disease: a collaborative British cohort study. J Natl Cancer Inst 99: 206–214
Laskar S et al. (2004) Consolidation radiation after complete remission in Hodgkin's disease following six cycles of doxorubicin, bleomycin, vinblastine, and dacarbazine chemotherapy: is there a need? J Clin Oncol 22: 62–68
Bonadonna G et al. (1986) Alternating non-cross-resistant combination chemotherapy or MOPP in stage IV Hodgkin's disease: a report of 8-year results. Ann Intern Med 104: 739–746
Longo DL et al. (1986) Twenty years of MOPP therapy for Hodgkin's disease. J Clin Oncol 4: 1295–1306
Nicholson WM et al. (1970) Combination chemotherapy in generalized Hodgkin's disease. Br Med J 3: 7–10
Sutcliffe SB et al. (1978) MVPP chemotherapy regimen for advanced Hodgkin's disease. Br Med J 1: 679–683
Hancock BW (1986) Randomised study of MOPP (mustine, Oncovin, procarbazine, prednisone) against LOPP (Leukeran substituted for mustine) in advanced Hodgkin's disease. British National Lymphoma Investigation. Radiother Oncol 7: 215–221
McElwain TJ et al. (1977) A combination of chlorambucil, vinblastine, procarbazine and prednisolone for treatment of Hodgkin's disease. Br J Cancer 36: 276–280
Bakemeier RF et al. (1984) BCVPP chemotherapy for advanced Hodgkin's disease: evidence for greater duration of complete remission, greater survival, and less toxicity than with a MOPP regimen. Results of the Eastern Cooperative Oncology Group study. Ann Intern Med 101: 447–456
Bonadonna G et al. (1975) Combination chemotherapy of Hodgkin's disease with adriamycin, bleomycin, vinblastine, and imidazole carboxamide versus MOPP. Cancer 36: 252–259
Santoro A et al. (1987) Long-term results of combined chemotherapy–radiotherapy approach in Hodgkin's disease: superiority of ABVD plus radiotherapy versus MOPP plus radiotherapy. J Clin Oncol 5: 27–37
Jones SE et al. (1983) Comparison of adriamycin-containing chemotherapy (MOP-BAP) with MOPP-bleomycin in the management of advanced Hodgkin's disease: a Southwest Oncology Group Study. Cancer 51: 1339–1347
Viviani S et al. (1996) Alternating versus hybrid MOPP and ABVD combinations in advanced Hodgkin's disease: ten-year results. J Clin Oncol 14: 1421–1430
Glick JH et al. (1998) MOPP/ABV hybrid chemotherapy for advanced Hodgkin's disease significantly improves failure-free and overall survival: the 8-year results of the intergroup trial. J Clin Oncol 16: 19–26
Somers R et al. (1994) A randomized study in stage IIIB and IV Hodgkin's disease comparing eight courses of MOPP versus an alteration of MOPP with ABVD: a European Organization for Research and Treatment of Cancer Lymphoma Cooperative Group and Groupe Pierre-et-Marie-Curie controlled clinical trial. J Clin Oncol 12: 279–287
Canellos GP et al. (1992) Chemotherapy of advanced Hodgkin's disease with MOPP, ABVD, or MOPP alternating with ABVD. N Engl J Med 327: 1478–1484
Duggan DB et al. (2003) Randomized comparison of ABVD and MOPP/ABV hybrid for the treatment of advanced Hodgkin's disease: report of an intergroup trial. J Clin Oncol 21: 607–614
Horning SJ et al. (2002) Stanford V and radiotherapy for locally extensive and advanced Hodgkin's disease: mature results of a prospective clinical trial. J Clin Oncol 20: 630–637
Gobbi PG et al. (2005) ABVD versus modified Stanford V versus MOPPEBVCAD with optional and limited radiotherapy in intermediate- and advanced-stage Hodgkin's lymphoma: final results of a multicenter randomized trial by the Intergruppo Italiano Linfomi. J Clin Oncol 23: 9198–9207
Radford JA et al. (2002) ChlVPP/EVA hybrid versus the weekly VAPEC-B regimen for previously untreated Hodgkin's disease. J Clin Oncol 20: 2988–2994
Diehl V et al. (2003) Standard and increased-dose BEACOPP chemotherapy compared with COPP-ABVD for advanced Hodgkin's disease. N Engl J Med 348: 2386–2395
Sieber M et al. (2003) 14-day variant of the bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone regimen in advanced-stage Hodgkin's lymphoma: results of a pilot study of the German Hodgkin's Lymphoma Study Group. J Clin Oncol 21: 1734–1739
Aleman BMP et al. (2003) Involved-field radiotherapy for advanced Hodgkin's lymphoma. N Engl J Med 348: 2396–2406
Diehl V et al. (1995) Further chemotherapy versus low-dose involved-field radiotherapy as consolidation of complete remission after six cycles of alternating chemotherapy in patients with advanced Hodgkin's disease. Ann Oncol 6: 901–910
Raemaekers J et al. (1997) Patients with stage III/IV Hodgkin's disease in partial remission after MOPP/ABV chemotherapy have excellent prognosis after additional involved-field radiotherapy: interim results from the ongoing EORTC-LCG and GPMC phase III trial. The EORTC Lymphoma Cooperative Group and Groupe Pierre-et-Marie-Curie. Ann Oncol 8 (Suppl 1): 111–114
Aleman BM et al. (2007) Involved-field radiotherapy for patients in partial remission after chemotherapy for advanced Hodgkin's lymphoma. Int J Radiat Oncol Biol Phys 67: 19–30
Ferme C et al. (2006) Long-term results and competing risk analysis of the H89 trial in patients with advanced-stage Hodgkin lymphoma: a study by the Groupe d'Etude des Lymphomes de l'Adulte (GELA). Blood 107: 4636–4642
Engert A et al. (2006) HD12 randomised trial comparing 8 dose-escalated cycles of BEACOPP with 4 escalated and 4 baseline cycles in patients with advanced stage Hodgkin lymphoma (HL): an analysis of the German Hodgkin Lymphoma Study Group (GHSG). Blood 108: 99A
Franklin J et al. (2006) Second malignancy risk associated with treatment of Hodgkin's lymphoma: meta-analysis of the randomised trials. Ann Oncol 17: 1749–1760
Hasenclever D and Diehl V (1998) A prognostic score for advanced Hodgkin's disease. International Prognostic Factors Project on Advanced Hodgkin's Disease. N Engl J Med 339: 1506–1514
Friedberg JW et al. (2004) FDG-PET is superior to gallium scintigraphy in staging and more sensitive in the follow-up of patients with de novo Hodgkin lymphoma: a blinded comparison. Leuk Lymphoma 45: 85–92
Stumpe KD et al. (1998) Whole-body positron emission tomography using fluorodeoxyglucose for staging of lymphoma: effectiveness and comparison with computed tomography. Eur J Nucl Med 25: 721–728
Torizuka T et al. (2004) Early therapy monitoring with FDG-PET in aggressive non-Hodgkin's lymphoma and Hodgkin's lymphoma. Eur J Nucl Med Mol Imaging 31: 22–28
Hutchings M et al. (2006) FDG-PET after two cycles of chemotherapy predicts treatment failure and progression-free survival in Hodgkin lymphoma. Blood 107: 52–59
Gallamini A et al. (2006) The predictive value of positron emission tomography scanning performed after two courses of standard therapy on treatment outcome in advanced stage Hodgkin's disease. Haematologica 91: 475–481
Zinzani PL et al. (2006) Early positron emission tomography (PET) restaging: a predictive final response in Hodgkin's disease patients. Ann Oncol 17: 1296–1300
Dann EJ et al. (2007) Risk-adapted BEACOPP regimen can reduce the cumulative dose of chemotherapy for standard and high-risk Hodgkin lymphoma with no impairment of outcome. Blood 109: 905–909
Gallamini A et al. (2007) Early interim 2-[18F]fluoro-2-deoxy-D-glucose positron emission tomography is prognostically superior to international prognostic score in advanced-stage Hodgkin's lymphoma: a report from a joint Italian-Danish study. J Clin Oncol 25: 3746–3752
Carde P (1983) A dose and time response analysis of the treatment of Hodgkin's disease with MOPP chemotherapy. J Clin Oncol 1: 146–153
van Rijswijk RE et al. (1989) Dose intensity of MOPP chemotherapy and survival in Hodgkin's disease. J Clin Oncol 7: 1776–1782
Klimm B et al. (2005) Role of hematotoxicity and sex in patients with Hodgkin's lymphoma: an analysis from the German Hodgkin Study Group. J Clin Oncol 23: 8003–8011
Landgren O et al. (2003) Hodgkin's lymphoma in the elderly with special reference to type and intensity of chemotherapy in relation to prognosis. Haematologica 88: 438–444
Chand VK et al. (2006) Neutropenia and febrile neutropenia in patients with Hodgkin's lymphoma treated with doxorubicin (Adriamycin), bleomycin, vinblastine and dacarbazine (ABVD) chemotherapy. Leuk Lymphoma 47: 657–663
Johnson PW et al. (2005) Comparison of ABVD and alternating or hybrid multidrug regimens for the treatment of advanced Hodgkin's lymphoma: results of the United Kingdom Lymphoma Group LY09 Trial (ISRCTN97144519). J Clin Oncol 23: 9208–9218
Rueda A et al. (2001) Secondary prophylactic G-CSF (filgrastim) administration in chemotherapy of stage I and II Hodgkin's lymphoma with ABVD. Leuk Lymphoma 41: 353–358
Silvestri F et al. (1994) The role of granulocyte colony-stimulating factor (filgrastim) in maintaining dose intensity during conventional-dose chemotherapy with ABVD in Hodgkin's disease. Tumori 80: 453–458
Sleijfer S (2001) Bleomycin-induced pneumonitis. Chest 120: 617–624
Azoulay E et al. (2003) Effect of granulocyte colony-stimulating factor on bleomycin-induced acute lung injury and pulmonary fibrosis. Crit Care Med 31: 1442–1448
Behringer K et al. (2005) Secondary amenorrhea after Hodgkin's lymphoma is influenced by age at treatment, stage of disease, chemotherapy regimen, and the use of oral contraceptives during therapy: a report from the German Hodgkin's Lymphoma Study Group. J Clin Oncol 23: 7555–7564
Evens AM et al. (2007) G-CSF is not necessary to maintain over 99% dose-intensity with ABVD in the treatment of Hodgkin lymphoma: low toxicity and excellent outcomes in a 10-year analysis. Br J Haematol 137: 545–552
Nogova L et al. (2005) Extended field radiotherapy, combined modality treatment or involved field radiotherapy for patients with stage IA lymphocyte-predominant Hodgkin's lymphoma: a retrospective analysis from the German Hodgkin Study Group (GHSG). Ann Oncol 16: 1683–1687
Diehl V et al. (1999) Clinical presentation, course, and prognostic factors in lymphocyte-predominant Hodgkin's disease and lymphocyte-rich classical Hodgkin's disease: report from the European Task Force on Lymphoma Project on Lymphocyte-Predominant Hodgkin's Disease. J Clin Oncol 17: 776–783
Nogova L et al. (2005) Lymphocyte-predominant and classical Hodgkin's lymphoma—comparison of outcomes. Eur J Haematol Suppl 66: 106–110
Ekstrand BC et al. (2003) Rituximab in lymphocyte-predominant Hodgkin disease: results of a phase 2 trial. Blood 101: 4285–4289
Rehwald U et al. (2003) Treatment of relapsed CD20+ Hodgkin lymphoma with the monoclonal antibody rituximab is effective and well tolerated: results of a phase 2 trial of the German Hodgkin Lymphoma Study Group. Blood 101: 420–424
Proctor SJ et al. (2002) Hodgkin's disease in the elderly: current status and future directions. Ann Oncol 13 (Suppl 1): 133–137
Engert A et al. (2005) Hodgkin's lymphoma in elderly patients: a comprehensive retrospective analysis from the German Hodgkin's Study Group. J Clin Oncol 23: 5052–5060
Ballova V et al. (2005) A prospectively randomized trial carried out by the German Hodgkin Study Group (GHSG) for elderly patients with advanced Hodgkin's disease comparing BEACOPP baseline and COPP-ABVD (study HD9 elderly). Ann Oncol 16: 124–131
Martin WG et al. (2005) Bleomycin pulmonary toxicity has a negative impact on the outcome of patients with Hodgkin's lymphoma. J Clin Oncol 23: 7614–7620
Canellos GP et al. (2004) How important is bleomycin in the adriamycin + bleomycin + vinblastine + dacarbazine regimen. J Clin Oncol 22: 1532–1533
Weekes CD et al. (2002) Hodgkin's disease in the elderly: improved treatment outcome with a doxorubicin-containing regimen. J Clin Oncol 20: 1087–1093
Kolstad et al. (2007) Standard CHOP-21 as first line therapy for elderly patients with Hodgkin's lymphoma. Leuk Lymphoma 48: 570–576
Proctor SJ et al. (2005) An international approach to the treatment of Hodgkin's disease in the elderly: launch of the SHIELD study programme. Eur J Haematol Suppl 66: 63–67
Cheson BD (2001) Pharmacology of cancer chemotherapy: miscellaneous chemotherapeutic agents. In Cancer Principles and Practice of Oncology. (Eds De Vita Jr VT et al.) Philadelphia: Lippincott Williams & Wilikins
Matthews JH (1993) Pulmonary toxicity of ABVD chemotherapy and G-CSF in Hodgkin's disease: possible synergy. Lancet 342: 988
Dirix LY et al. (1994) Pulmonary toxicity and bleomycin. Lancet 344: 56
Adach K et al. (2002) Granulocyte colony-stimulating factor exacerbates the acute lung injury and pulmonary fibrosis induced by intratracheal administration of bleomycin in rats. Exp Toxicol Pathol 53: 501–510
Brusamolino E et al. (2006) Long-term events in adult patients with clinical stage IA-IIA nonbulky Hodgkin's lymphoma treated with four cycles of doxorubicin, bleomycin, vinblastine, and dacarbazine and adjuvant radiotherapy: a single-institution 15-year follow-up. Clin Cancer Res 12: 6487–6493
Illes A et al. (2003) Hypothyroidism and thyroiditis after therapy for Hodgkin's disease. Acta Haematol 109: 11–17
Younes A et al. (2003) A pilot study of rituximab in patients with recurrent, classic Hodgkin disease. Cancer 98: 310–314
Karin M et al. (2004) The IKK NF-kappa B system: a treasure trove for drug development. Nat Rev Drug Discov 3: 17–26
Miller CP et al. (2007) NPI-0052, a novel proteasome inhibitor, induces caspase-8 and ROS-dependent apoptosis alone and in combination with HDAC inhibitors in leukemia cells. Blood 110. 267–277
Zheng B et al. (2004) Induction of cell cycle arrest and apoptosis by the proteasome inhibitor PS-341 in Hodgkin disease cell lines is independent of inhibitor of nuclear factor-kappaB mutations or activation of the CD30, CD40, and RANK receptors. Clin Cancer Res 10: 3207–3215
Younes A et al. (2006) Experience with bortezomib for the treatment of patients with relapsed classical Hodgkin lymphoma. Blood 107: 1731–1732
Boll B et al. (2005) The fully human anti-CD30 antibody 5F11 activates NF-κB and sensitizes lymphoma cells to bortezomib-induced apoptosis. Blood 106: 1839–1842
Georgakis GV et al. (2005) Activity of selective fully human agonistic antibodies to the TRAIL death receptors TRAIL-R1 and TRAIL-R2 in primary and cultured lymphoma cells: induction of apoptosis and enhancement of doxorubicin- and bortezomib-induced cell death. Br J Haematol 130: 501–510
Rosato RR et al. (2003) The histone deacetylase inhibitor MS-275 promotes differentiation or apoptosis in human leukemia cells through a process regulated by generation of reactive oxygen species and induction of p21CIP1/WAF1 1. Cancer Res 63: 3637–3645
Portis T and Longnecker R (2004) Epstein-Barr virus (EBV) LMP2A mediates B-lymphocyte survival through constitutive activation of the Ras/PI3K/Akt pathway. Oncogene 23: 8619–8628
Carde P et al. (1993) Clinical staging versus laparotomy and combined modality with MOPP versus ABVD in early-stage Hodgkin's disease: the H6 twin randomized trials from the European Organization for Research and Treatment of Cancer Lymphoma Cooperative Group. J Clin Oncol 11: 2258–2272
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Evens, A., Hutchings, M. & Diehl, V. Treatment of Hodgkin lymphoma: the past, present, and future. Nat Rev Clin Oncol 5, 543–556 (2008). https://doi.org/10.1038/ncponc1186
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncponc1186
This article is cited by
-
Survival in hematological malignancies in the Nordic countries through a half century with correlation to treatment
Leukemia (2023)
-
Survival trends in hematological malignancies in the Nordic countries through 50 years
Blood Cancer Journal (2022)
-
Changing causes of death in persons with haematological cancers 1975–2016
Leukemia (2022)
-
Impact of the first COVID-19 lockdown in Germany on the rate of acute infections during intensive chemotherapy for Hodgkin lymphoma
Infection (2022)
-
Incidence, mortality and survival in multiple myeloma compared to other hematopoietic neoplasms in Sweden up to year 2016
Scientific Reports (2021)