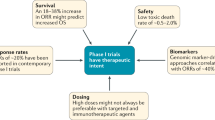
Abstract
Abundant preclinical and clinical data suggest that cancer is a preventable disease; however, few, if any, of the medical interventions available are completely without adverse effects or risk to the patient. This statement is especially true with regard to phenotypically healthy but high-risk populations. Balancing the risks from interventions with the risk of future life-threatening disease is particularly challenging because no standardized methodologies to perform such calculations exist. It is critical, therefore, to establish a framework for determining when interventions show sufficient promise of efficacy and sufficient safety to justify their testing in clinical trials. Systematic review of all available preclinical, epidemiological, and clinical data, along with a mechanistic understanding of the biology of the disease under study, is mandatory before clinical trials are embarked upon. This Review identifies the issues that are critical for decision-making when clinical trials in human beings are being contemplated and provides a framework that can be applied in making these decisions.
Key Points
-
Selection of agents for cancer prevention clinical trials requires a thorough understanding of the potential efficacy and toxic effects associated with specific agents
-
Evidence of effectiveness at cancer prevention comes from investigation of mechanisms, in vitro and animal in vivo experiments, epidemiological case–control and cohort studies, and clinical trials (including both early-phase cancer prevention trials and secondary end point analysis of trials performed for other indications)
-
The risk:benefit balance can be shifted towards benefit by the development of trials in appropriately high-risk cohorts and use of low-toxicity regional, combination, or intermittent drug delivery regimens whenever possible
-
Understanding the molecular mechanisms and the temporal sequence of events that result in carcinogenesis is critical to the choice of agents and the design of prevention trials
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sporn MB and Liby KT (2005) Cancer chemoprevention: scientific promise, clinical uncertainty. Nat Clin Pract Oncol 2: 518–525
Kelloff GJ et al. (2006) Progress in chemoprevention drug development: the promise of molecular biomarkers for prevention of intraepithelial neoplasia (IEN) and cancer (AACR Cancer Prevention Task Force). Clin Cancer Res 12: 3661–3697
Lippman SM and Heymach JV (2007) The convergent development of molecular-targeted drugs for cancer treatment and prevention. Clin Cancer Res 13: 4035–4041
Vogel V et al. (2006) Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes. JAMA 295: 2727–2741
The ATBC Study Group (1994) The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. The Alpha-Tocopherol Beta Carotene Cancer Prevention Study Group. N Engl J Med 330: 1029–1035
Weinstein IB (2002) Addiction to oncogenes—the Achilles heal of cancer. Science 297: 63–64
Cohen MH et al. (2005) U.S. Food and Drug Administration drug approval summary: conversion of imatinib mesylate (STI571; Gleevec) tablets from accelerated approval to full approval. Clin Cancer Res 11: 12–19
Blanke CD and Corless CL (2005) State-of-the art therapy for gastrointestinal stromal tumors. Cancer Invest 23: 274–280
Dy GK et al. (2005) A phase II trial of imatinib (STI571) in patients with c-kit expressing relapsed small-cell lung cancer: a CALGB and NCCTG study. Ann Oncol 16: 1811–1816
Burger H et al. (2003) Lack of c-kit exon 11 activating mutations in c-KIT/CD117-positive SCLC tumour specimens. Eur J Cancer 39: 793–799
Piazza GA et al. (1997) Sulindac sulfone inhibits azoxymethane-induced colon carcinogenesis in rats without reducing prostaglandin levels. Cancer Res 57: 2909–2915
Wick M et al. (2002) Peroxisome proliferator-activated receptor-gamma is a target of nonsteroidal anti-inflammatory drugs mediating cyclooxygenase-independent inhibition of lung cancer cell growth. Mol Pharmacol 62: 1207–1214
Corpet DE and Pierre F (2005) How good are rodent models of carcinogenesis in predicting efficacy in humans? A systematic review and meta-analysis of colon chemoprevention in rats, mice and men. Eur J Cancer 41: 1911–1922
Green JE and Hudson T (2005) The promise of genetically engineered mice for cancer prevention studies. Nat Rev Cancer 5: 184–198
Hursting SD et al. (2005) The utility of genetically altered mouse models for nutrition and cancer chemoprevention research. Mutat Res 576: 80–92
Jacoby RF et al. (2000) The cyclooxygenase-2 inhibitor celecoxib is a potent preventive and therapeutic agent in the Min mouse model of adenomatous polyposis. Cancer Res 60: 5040–5044
Steinbach G et al. (2000) The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N Engl J Med 342: 1946–1952
Bertagnolli MM et al. (2006) Celecoxib for the prevention of sporadic colorectal adenomas. N Engl J Med 355: 873–884
Arber N et al. (2006) Celecoxib for the prevention of colorectal adenomatous polyps. N Engl J Med 355: 885–895
Peto R et al. (1981) Can dietary beta-carotene materially reduce human cancer rates. Nature 290: 201–208
Dorgan JF and Schatzkin A (1991) Antioxidant micronutrients in cancer prevention. Hematol Oncol Clin North Am 5: 43–68
Omenn GS et al. (1996) Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N Engl J Med 334: 1150–1155
Wang XD et al. (1999) Retinoid signaling and activator protein-1 expression in ferrets given β-carotene supplements and exposed to tobacco smoke. J Natl Cancer Inst 91: 60–66
Anderson WF et al. (2003) Cyclooxygenase inhibition in cancer prevention and treatment. Expert Opin Pharmacother 4: 2193–2204
Baron JA et al. (2003) A randomized trial of aspirin to prevent colorectal adenomas. N Engl J Med 348: 891–899
Sandler RS et al. (2003) A randomized trial of aspirin to prevent colorectal adenomas in patients with previous colorectal cancer. N Engl J Med 348: 883–890
Baron J et al. (2006) A randomized trial of rofecoxib for the chemoprevention of colorectal cancer. Gastroenterology 131: 1674–1682
Chan AT et al. (2007) Aspirin and the risk of colorectal cancer in relation to the expression of COX-2. N Engl J Med 356: 2131–2142
Meyskens FL and Szabo E (2005) Diet and cancer: the disconnect between epidemiology and randomized clinical trials. Cancer Epidemiol Biomarkers Prev 14: 1366–1369
Guarnieri S et al. (2007) Orange juice vs vitamin C: effect on hydrogen peroxide-induced DNA damage in mononuclear blood cells. Br J Nutr 97: 639–643
Fisher B et al. (1989) A randomized clinical trial evaluating tamoxifen in the treatment of patients with node-negative breast cancer who have estrogen-receptor-positive tumors. N Engl J Med 320: 479–484
Cummings SR et al. (1999) The effect of raloxifene on risk of breast cancer in postmenopausal women: results from the MORE randomized trial: multiple outcomes of raloxifene evaluation. JAMA 281: 2189–2197
Fisher B et al. (1998) Tamoxifen for Prevention of Breast Cancer: Report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst 190: 1371–1388
Klein EA et al. (2003) SELECT: the selenium and vitamin E cancer prevention trial. Urol Oncol 21: 59–65
Clark LC et al. (1996) Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. JAMA 276: 1957–1963
Lam S et al. (2002) A randomized phase IIb trial of anethole dithiolethione in smokers with bronchial dysplasia. J Natl Cancer Inst 94: 1001–1009
Seiwert TY et al. (2007) The chemoradiation paradigm in head and neck cancer. Nat Clin Pract Oncol 4: 156–171
Bresalier RS et al. (2005) Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 352: 1092–1102
Nissen SE (2006) Adverse cardiovascular effects of rofecoxib. N Engl J Med 355: 203–204
Nissen SE et al. (2005) Effect of muraglitazar on death and major adverse cardiovascular events in patients with type 2 diabetes mellitus. JAMA 294: 2581–2586
Nissen SE and Wolski K (2007) Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med 356: 2457–2471
Dormandy JA et al. (2005) Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomized controlled trial. Lancet 366: 1279–1289
Gail MH et al. (1989) Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst 81: 1879–1886
Bach PB et al. (2003) Variations in lung cancer risk among smokers. J Natl Cancer Inst 95: 470–478
Spitz MR et al. (2007) A risk model for prediction of lung cancer. J Natl Cancer Inst 99: 715–726
Silverman S Jr et al. (1984) Oral leukoplakia and malignant transformation: a follow-up study of 257 patients. Cancer 53: 563–568
Rosin MP et al. (2000) Use of allelic loss to predict malignant risk for low grade epithelial dysplasia. Clin Cancer Res 6: 357–362
Lam S et al. (2004) A randomized phase IIb trial of Pulmicort turbuhaler (budesonide) in persons with dysplasia of the bronchial epithelium. Clin Cancer Res 10: 6502–6511
Grubbs CJ et al. (2007) Preventive effects of the EGFR inhibitor Iressa alone and in combination with Targretin or Vorozole in the methylnitrosourea (MNU) induced mammary cancer model. Proc Am Assoc Cancer Res 48: 592
Solomon SD et al. (2005) Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N Engl J Med 352: 1071–1080
Thompson IM et al. (2003) The influence of finasteride on the development of prostate cancer. N Engl J Med 349: 215–224
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Szabo, E. Primer: first do no harm—when is it appropriate to plan a cancer prevention clinical trial?. Nat Rev Clin Oncol 5, 348–356 (2008). https://doi.org/10.1038/ncponc1123
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncponc1123