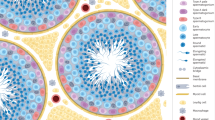
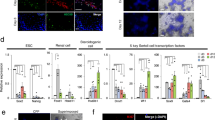
Abstract
Male germline stem cells—spermatogonial stem cells (SSCs)—self-renew and produce large numbers of differentiating germ cells that become spermatozoa throughout postnatal life and transmit genetic information to the next generation. SSCs are the only germline stem cells in adults, because all female germline stem cells cease proliferation before birth. In this article, we first summarize development of SSCs, and then the relation of SSCs to somatic stem cells in tissues and pluripotent stem cells in vitro, such as embryonic stem cells. Next, we describe a transplantation technique in which donor testis cells from a fertile male can be transplanted to the testes of an infertile male where they re-establish spermatogenesis and restore fertility. The transplantation technique has been used to study the biology of SSCs, which made possible the identification of external factors that support in vitro self-renewal and proliferation of mouse and rat SSCs. Since SSCs of all mammalian species examined, including human, can replicate in mouse seminiferous tubules following transplantation, the growth factors required for SSC self-renewal are probably conserved among mammalian species. Culture techniques should therefore soon be available for human SSCs. In the final section, we discuss current and potential approaches for using the transplantation technique and in vitro culture of SSCs in human medicine. Because assisted reproductive techniques to fertilize oocytes with round or elongated spermatids are available, clinical use of cultured human SSCs will be greatly facilitated by development of techniques for in vitro differentiation of SSCs to mature germ cells.
Key Points
-
Spermatogonial stem cells are the only cells in postnatal mammals that undergo self-renewal and transmit genes to subsequent generations
-
In vitro growth-factor requirements for mouse spermatogonial stem cells have been identified using defined culture conditions
-
Because extrinsic factors for self-renewal of spermatogonial stem cells appear to be conserved among many mammalian species, including humans, in vitro culture techniques for human spermatogonial stem cell will probably be developed in the near future
-
Spermatogonial stem-cell transplantation could be used to restore fertility in men following chemotherapy or radiation treatment
-
Development of techniques for the in vitro differentiation of spermatogonial stem cells to functional spermatozoa is a crucial step for the treatment of infertility or germline gene therapy
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Brinster RL and Zimmermann JW (1994) Spermatogenesis following male germ-cell transplantation. Proc Natl Acad Sci USA 91: 11298–11302
Brinster RL and Avarbock MR (1994) Germline transmission of donor haplotype following spermatogonial transplantation. Proc Natl Acad Sci USA 91: 11303–11307
Kubota H et al. (2004) Growth factors essential for self-renewal and expansion of mouse spermatogonial stem cells. Proc Natl Acad Sci USA 101: 16489–16494
Radford J et al. (1999) Fertility after treatment for cancer. Questions remain over ways of preserving ovarian and testicular tissue. BMJ 319: 935–936
Radford J (2003) Restoration of fertility after treatment for cancer. Horm Res 59 (Suppl 1): 21–23
Barzon L et al. (2000) New perspectives for gene therapy in endocrinology. Eur J Endocrinol 143: 447–466
Nagy A et al. (2003) Manipulating the Mouse Embryo, A Laboratory Manual. Cold Spring Harbor: Cold Spring Harbor Laboratory Press
Potten CS and Morris RJ (1988) Epithelial stem cells in vivo. J Cell Sci Suppl 10: 45–62
Reid LM (1990) Stem cell biology, hormone/matrix synergies and liver differentiation. Curr Opin Cell Biol 2: 121–130
Tam PP and Zhou SX (1996) The allocation of epiblast cells to ectodermal and germ-line lineages is influenced by the position of the cells in the gastrulating mouse embryo. Dev Biol 178: 124–132
McLaren A (2003) Primordial germ cells in the mouse. Dev Biol 262: 1–15
Johnson J et al. (2004) Germline stem cells and follicular renewal in the postnatal mammalian ovary. Nature 428: 145–150
Johnson J et al. (2005) Oocyte generation in adult mammalian ovaries by putative germ cells in bone marrow and peripheral blood. Cell 122: 303–315
Telfer EE et al. (2005) On regenerating the ovary and generating controversy. Cell 122: 821–822
de Rooij DG (1998) Stem cells in the testis. Int J Exp Pathol 79: 67–80
Orwig KE et al. (2002) Male germ-line stem cell potential is predicted by morphology of cells in neonatal rat testes. Proc Natl Acad Sci USA 99: 11706–11711
Holdcraft RW and Braun RE (2004) Hormonal regulation of spermatogenesis. Int J Androl 27: 335–342
Meistrich ML and van Beek MEAB (1993) Spermatogonial stem cells. In Cell and Molecular Biology of the Testis, 266–295 (Eds Desjardins C and Ewing LL) New York: Oxford University Press
Brinster RL (2002) Germline stem cell transplantation and transgenesis. Science 296: 2174–2176
Evans MJ and Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292: 154–156
Martin GR (1981) Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci USA 78: 7634–7638
Thomson JA et al. (1998) Embryonic stem cell lines derived from human blastocysts. Science 282: 1145–1147
Smith AG (2001) Embryo-derived stem cells: of mice and men. Annu Rev Cell Dev Biol 17: 435–462
Matsui Y et al. (1992) Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell 70: 841–847
Resnick JL et al. (1992) Long-term proliferation of mouse primordial germ cells in culture. Nature 359: 550–551
Shamblott MJ et al. (1998) Derivation of pluripotent stem cells from cultured human primordial germ cells. Proc Natl Acad Sci USA 95: 13726–13731
Kanatsu-Shinohara M et al. (2004) Generation of pluripotent stem cells from neonatal mouse testis. Cell 119: 1001–1012
Harvey M et al. (1993) Genetic background alters the spectrum of tumors that develop in p53-deficient mice. FASEB J 7: 938–943
Stevens LC (1983) The origin and development of testicular, ovarian, and embryo-derived teratomas. In Teratocarcinoma Stem Cells, 23–36 (Eds Silver LM et al.) Cold Spring Harbor: Cold Spring Harbor Laboratory Press
Bradley A et al. (1984) Formation of germ-line chimaeras from embryo-derived teratocarcinoma cell lines. Nature 309: 255–256
Labosky PA et al. (1994) Mouse embryonic germ (EG) cell lines: transmission through the germline and differences in the methylation imprint of insulin-like growth factor 2 receptor (Igf2r) gene compared with embryonic stem (ES) cell lines. Development 120: 3197–3204
Ogawa T et al. (1997) Transplantation of testis germinal cells into mouse seminiferous tubules. Int J Dev Biol 41: 111–122
Ogawa T et al. (1999) Recipient preparation is critical for spermatogonial transplantation in the rat. Tissue Cell 31: 461–472
Honaramooz A et al. (2002) Germ cell transplantation in pigs. Biol Reprod 66: 21–28
Honaramooz A et al. (2003) Germ cell transplantation in goats. Mol Reprod Dev 64: 422–428
Izadyar F et al. (2003) Autologous and homologous transplantation of bovine spermatogonial stem cells. Reproduction 126: 765–774
Oatley JM et al. (2005) Changes in spermatogenesis and endocrine function in the ram testis due to irradiation and active immunization against luteinizing hormone-releasing hormone. J Anim Sci 83: 604–612
Schlatt S et al. (2002) Germ cell transplantation into X-irradiated monkey testes. Hum Reprod 17: 55–62
Nagano MC (2003) Homing efficiency and proliferation kinetics of male germ line stem cells following transplantation in mice. Biol Reprod 69: 701–707
Ogawa T et al. (2003) Expansion of murine spermatogonial stem cells through serial transplantation. Biol Reprod 68: 316–322
Nagano M et al. (1999) Pattern and kinetics of mouse donor spermatogonial stem cell colonization in recipient testes. Biol Reprod 60: 1429–1436
Dobrinski I et al. (1999) Computer assisted image analysis to assess colonization of recipient seminiferous tubules by spermatogonial stem cells from transgenic donor mice. Mol Reprod Dev 53: 142–148
Zhang X et al. (2003) Genetic analysis of the clonal origin of regenerating mouse spermatogenesis following transplantation. Biol Reprod 69: 1872–1878
Ogawa T et al. (2000) Transplantation of male germ line stem cells restores fertility in infertile mice. Nat Med 6: 29–34
Till JE and McCulloch E (1961) A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat Res 14: 213–222
Blanpain C et al. (2004) Self-renewal, multipotency, and the existence of two cell populations within an epithelial stem cell niche. Cell 118: 635–648
Morrison SJ et al. (1997) Regulatory mechanisms in stem cell biology. Cell 88: 287–298
Tegelenbosch RA and de Rooij DG (1993) A quantitative study of spermatogonial multiplication and stem cell renewal in the C3H/101 F1 hybrid mouse. Mutat Res 290: 193–200
Shinohara T et al. (1999) β1- and α6-integrin are surface markers on mouse spermatogonial stem cells. Proc Natl Acad Sci USA 96: 5504–5509
Shinohara T et al. (2000) Spermatogonial stem cell enrichment by multiparameter selection of mouse testis cells. Proc Natl Acad Sci USA 97: 8346–8351
Kubota H et al. (2003) Spermatogonial stem cells share some, but not all, phenotypic and functional characteristics with other stem cells. Proc Natl Acad Sci USA 100: 6487–6492
Kubota H et al. (2004) Culture conditions and single growth factors affect fate determination of mouse spermatogonial stem cells. Biol Reprod 71: 722–731
Ryu BY et al. (2004) Phenotypic and functional characteristics of spermatogonial stem cells in rats. Dev Biol 274: 158–170
Russell LD et al. (1990) Histological and Histopathological Evaluation of the Testis. Clearwater: Cache River Press
Clouthier DE et al. (1996) Rat spermatogenesis in mouse testis. Nature 381: 418–421
Dobrinski I et al. (1999) Transplantation of germ cells from rabbits and dogs into mouse testes. Biol Reprod 61: 1331–1339
Dobrinski I et al. (2000) Germ cell transplantation from large domestic animals into mouse testes. Mol Reprod Dev 57: 270–279
Oatley JM et al. (2004) Biological activity of cryopreserved bovine spermatogonial stem cells during in vitro culture. Biol Reprod 71: 942–947
Nagano M et al. (2001) Primate spermatogonial stem cells colonize mouse testes. Biol Reprod 64: 1409–1416
Nagano M et al. (2002) Long-term survival of human spermatogonial stem cells in mouse testes. Fertil Steril 78: 1225–1233
Kanatsu-Shinohara M et al. (2003) Long-term proliferation in culture and germline transmission of mouse male germline stem cells. Biol Reprod 69: 612–616
Nagano M et al. (2003) Maintenance of mouse male germ line stem cells in vitro. Biol Reprod 68: 2207–2214
Kubota H and Reid LM (2000) Clonogenic hepatoblasts, common precursors for hepatocytic and biliary lineages, are lacking classical major histocompatibility complex class I antigen. Proc Natl Acad Sci USA 97: 12132–12137
Barnes D and Sato G (1980) Serum-free cell culture: a unifying approach. Cell 22: 649–655
Chang HY et al. (2002) Diversity, topographic differentiation, and positional memory in human fibroblasts. Proc Natl Acad Sci USA 99: 12877–12882
Matzuk MM (2004) Germ-line immortality. Proc Natl Acad Sci USA 101: 16395–16396
Ryu BY et al. (2005) Conservation of spermatogonial stem cell self-renewal signaling between mouse and rat. Proc Natl Acad Sci USA 102: 14302–14307
Avarbock MR et al. (1996) Reconstitution of spermatogenesis from frozen spermatogonial stem cells. Nat Med 2: 693–696
Blackhall FH et al. (2002) Semen cryopreservation, utilisation and reproductive outcome in men treated for Hodgkin's disease. Br J Cancer 87: 381–384
Jahnukainen K et al. (2001) Intratesticular transplantation of testicular cells from leukemic rats causes transmission of leukemia. Cancer Research 61: 706–710
Fujita K et al. (2005) Transplantation of spermatogonial stem cells isolated from leukemic mice restores fertility without inducing leukemia. J Clin Invest 115: 1855–1861
Geijsen N et al. (2004) Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature 427: 148–154
Rideout WM III et al. (2002) Correction of a genetic defect by nuclear transplantation and combined cell and gene therapy. Cell 109: 17–27
Kolb AF et al. (2005) Site-directed genome modification: nucleic acid and protein modules for targeted integration and gene correction. Trends Biotechnol 23: 399–406
Urnov FD et al. (2005) Highly efficient endogenous human gene correction using designed zinc-finger nucleases. Nature 435: 646–651
Shinohara T et al. (2000) Functional analysis of spermatogonial stem cells in Steel and cryptorchid infertile mouse models. Dev Biol 220: 401–411
Acknowledgements
We thank Jon Oatley for helpful comments on the manuscript and James Hayden for assistance in developing the figures. Financial support for the research was from the National Institute of Child Health and Human Development Grant 044445; and the Robert J Kleberg Jr, and Helen C Kleberg Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Kubota, H., Brinster, R. Technology Insight: in vitro culture of spermatogonial stem cells and their potential therapeutic uses. Nat Rev Endocrinol 2, 99–108 (2006). https://doi.org/10.1038/ncpendmet0098
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpendmet0098
This article is cited by
-
Postnatal development of mouse spermatogonial stem cells as determined by immunophenotype, regenerative capacity, and long-term culture-initiating ability: a model for practical applications
Scientific Reports (2024)
-
Advances of three-dimensional (3D) culture systems for in vitro spermatogenesis
Stem Cell Research & Therapy (2023)
-
Current Progress in Stem Cell Therapy for Male Infertility
Stem Cell Reviews and Reports (2023)
-
Germline stem cells in human
Signal Transduction and Targeted Therapy (2022)
-
Conserved and non-conserved characteristics of porcine glial cell line-derived neurotrophic factor expressed in the testis
Scientific Reports (2018)