Abstract
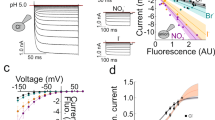
Neurotransmitter-sodium symporters (NSS), targets for psychostimulants and therapeutic drugs, have a critical role in neurotransmission. Whereas eukaryotic NSS show chloride-dependent transport, bacterial NSS feature Cl−-independent substrate transport. Recently we showed that mutation of an acidic residue near one of the sodium ion–binding sites in LeuT of Aquifex aeolicus or Tyt1 of Fusobacterium nucleatum renders substrate binding and/or transport Cl− dependent. We reasoned that the negative charge—provided either by Cl− or by the transporter itself—is required for substrate translocation. Here we show that Tyt1 reconstituted in proteoliposomes is strictly dependent on the Na+ gradient and is stimulated by an inside negative membrane potential and by an inversely oriented proton gradient. Notably, Na+/substrate symport elicited H+ efflux, indicative of Na+/substrate symport–coupled H+ antiport. Mutations that render the transport phenotype Cl− dependent essentially abolish the pH dependence. We propose unifying features of charge balance by all NSS members with similar mechanistic features but different molecular solutions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Rudnick, G. Mechanisms of biogenic amine neurotransmitter transporters. in Neurotransmitter Transporters. Structure, Function, and Regulation (ed. Reith, M.E.A.) 25–52 (Humana Press Inc., Totowa, New Jersey, USA, 2002).
Amara, S.G. & Sonders, M.S. Neurotransmitter transporters as molecular targets for addictive drugs. Drug Alcohol Depend. 51, 87–96 (1998).
Iversen, L. Neurotransmitter transporters and their impact on the development of psychopharmacology. Br. J. Pharmacol. 147 (Suppl. 1), S82–S88 (2006).
Gu, H., Wall, S.C. & Rudnick, G. Stable expression of biogenic amine transporters reveals differences in inhibitor sensitivity, kinetics, and ion dependence. J. Biol. Chem. 269, 7124–7130 (1994).
Norregaard, L. & Gether, U. The monoamine neurotransmitter transporters: structure, conformational changes and molecular gating. Curr. Opin. Drug Discov. Dev. 4, 591–601 (2001).
Torres, G.E., Gainetdinov, R.R. & Caron, M.G. Plasma membrane monoamine transporters: structure, regulation and function. Nat. Rev. Neurosci. 4, 13–25 (2003).
Keynan, S. & Kanner, B.I. γ-Aminobutyric acid transport in reconstituted preparations from rat brain: coupled sodium and chloride fluxes. Biochemistry 27, 12–17 (1988).
Wadiche, J.I., Amara, S.G. & Kavanaugh, M.P. Ion fluxes associated with excitatory amino acid transport. Neuron 15, 721–728 (1995).
Androutsellis-Theotokis, A. et al. Characterization of a functional bacterial homologue of sodium-dependent neurotransmitter transporters. J. Biol. Chem. 278, 12703–12709 (2003).
Yamashita, A. et al. Crystal structure of a bacterial homologue of Na+/Cl−-dependent neurotransmitter transporters. Nature 437, 215–223 (2005).
Quick, M. et al. State-dependent conformations of the translocation pathway in the tyrosine transporter Tyt1, a novel neurotransmitter:sodium symporter from Fusobacterium nucleatum. J. Biol. Chem. 281, 26444–26454 (2006).
Quick, M. & Javitch, J.A. Monitoring the function of membrane transport proteins in detergent-solubilized form. Proc. Natl. Acad. Sci. USA 104, 3603–3608 (2007).
Zomot, E. et al. Mechanism of chloride interaction with neurotransmitter:sodium symporters. Nature 449, 726–730 (2007).
Loo, D.D. et al. Perturbation analysis of the voltage-sensitive conformational changes of the Na+/glucose cotransporter. J. Gen. Physiol. 125, 13–36 (2005).
Goldberg, N.R. et al. Probing conformational changes in neurotransmitter transporters: a structural context. Eur. J. Pharmacol. 479, 3–12 (2003).
Beuming, T., Shi, L., Javitch, J.A. & Weinstein, H. A comprehensive structure-based alignment of prokaryotic and eukaryotic neurotransmitter/Na+ symporters (NSS) aids in the use of the LeuT structure to probe NSS structure and function. Mol. Pharmacol. 70, 1630–1642 (2006).
Quick, M. & Jung, H. Aspartate 55 in the Na+/proline permease of Escherichia coli is essential for Na+-coupled proline uptake. Biochemistry 36, 4631–4636 (1997).
Shi, L. et al. The mechanism of a neurotransmitter:sodium symporter–inward release of Na+ and substrate is triggered by substrate in a second binding site. Mol. Cell 30, 667–677 (2008).
Mehler, E.L. & Guarnieri, F. A self-consistent, microenvironment modulated screened coulomb potential approximation to calculate pH-dependent electrostatic effects in proteins. Biophys. J. 77, 3–22 (1999).
Forrest, L.R. et al. Identification of a chloride ion binding site in Na+/Cl− dependent transporters. Proc. Natl. Acad. Sci. USA 104, 12761–12766 (2007).
Pace, C.N., Grimsley, G.R. & Scholtz, J.M. Protein ionizable groups: pK values and their contribution to protein stability and solubility. J. Biol. Chem. 284, 13285–13289 (2009).
Mitchell, P. Molecule, group and electron translocation throught natural membranes. Biochem. Soc. Symp. 22, 142–168 (1962).
Jung, H., Tebbe, S., Schmid, R. & Jung, K. Unidirectional reconstitution and characterization of purified Na+/proline transporter of Escherichia coli. Biochemistry 37, 11083–11088 (1998).
Quick, M. & Wright, E.M. Employing Escherichia coli to functionally express, purify, and characterize a human transporter. Proc. Natl. Acad. Sci. USA 99, 8597–8601 (2002).
Hall, J.A. & Pajor, A.M. Functional reconstitution of SdcS, a Na+-coupled dicarboxylate carrier protein from Staphylococcus aureus. J. Bacteriol. 189, 880–885 (2007).
Sun, A.Q. et al. Identification of functionally relevant residues of the rat ileal apical sodium-dependent bile acid cotransporter. J. Biol. Chem. 281, 16410–16418 (2006).
Smirnova, I.N., Kasho, V. & Kaback, H.R. Protonation and sugar binding to LacY. Proc. Natl. Acad. Sci. USA 105, 8896–8901 (2008).
Capaldi, R.A. & Aggeler, R. Mechanism of the F1F0-type ATP synthase, a biological rotary motor. Trends Biochem. Sci. 27, 154–160 (2002).
Hilgemann, D.W. & Lu, C.C. GAT1 (GABA:Na+:Cl−) cotransport function. Database reconstruction with an alternating access model. J. Gen. Physiol. 114, 459–475 (1999).
Loo, D.D. et al. Role of Cl- in electrogenic Na+-coupled cotransporters GAT1 and SGLT1. J. Biol. Chem. 275, 37414–37422 (2000).
Erreger, K., Grewer, C., Javitch, J.A. & Galli, A. Currents in response to rapid concentration jumps of amphetamine uncover novel aspects of human dopamine transporter function. J. Neurosci. 28, 976–989 (2008).
Rudnick, G. & Nelson, P.J. Platelet 5-hydroxytryptamine transport, an electroneutral mechanism coupled to potassium. Biochemistry 17, 4739–4742 (1978).
Rudnick, G. & Clark, J. From synapse to vesicle: the reuptake and storage of biogenic amine neurotransmitters. Biochim. Biophys. Acta 1144, 249–263 (1993).
Keyes, S.R. & Rudnick, G. Coupling of transmembrane proton gradients to platelet serotonin transport. J. Biol. Chem. 257, 1172–1176 (1982).
Hunte, C. et al. Structure of a Na+/H+ antiporter and insights into mechanism of action and regulation by pH. Nature 435, 1197–1202 (2005).
Arkin, I.T. et al. Mechanism of Na+/H+ antiporting. Science 317, 799–803 (2007).
Guptaroy, B. et al. A juxtamembrane mutation in the N-terminus of the dopamine transporter induces preference for an inward-facing conformation. Mol. Pharmacol. 75, 514–524 (2009).
Sasaki, J. et al. Conversion of bacteriorhodopsin into a chloride ion pump. Science 269, 73–75 (1995).
Breeuwer, P., Drocourt, J., Rombouts, F.M. & Abee, T. A novel method for continuous determination of the intracellular pH in bacteria with the internally conjugated fluorescent probe 5 (and 6-)-carboxyfluorescein succinimidyl ester. Appl. Environ. Microbiol. 62, 178–183 (1996).
Kano, K. & Fendler, J.H. Pyranine as a sensitive pH probe for liposome interiors and surfaces. pH gradients across phospholipid vesicles. Biochim. Biophys. Acta 509, 289–299 (1978).
Indiveri, C., Tonazzi, A., Stipani, I. & Palmieri, F. The purified and reconstituted ornithine/citrulline carrier from rat liver mitochondria: electrical nature and coupling of the exchange reaction with H+ translocation. Biochem. J. 327, 349–355 (1997).
Warshel, A. Calculations of enzymatic reactions: calculations of pKa, proton transfer reactions, and general acid catalysis reactions in enzymes. Biochemistry 20, 3167–3177 (1981).
Bashford, D. & Karplus, M. pKa's of ionizable groups in proteins: atomic detail from a continuum electrostatic model. Biochemistry 29, 10219–10225 (1990).
Screpanti, E. & Hunte, C. Discontinuous membrane helices in transport proteins and their correlation with function. J. Struct. Biol. 159, 261–267 (2007).
Acknowledgements
We thank B. Kanner for helpful discussion, M.A. Gawinovicz of the Columbia University Medical Center Protein Core Facility for the MALDI-TOF analysis of Tyt1 and L. Chung for technical assistance. Computations were performed on the Ranger at the Texas Advanced Computing Center (TG-MCB090022) and the computational infrastructure of the Institute for Computational Biomedicine at Weill Cornell Medical College. This work was supported by US National Institutes of Health grants DA022413 and DA17293 (J.A.J.), P01 DA012923 (H.W.), DA015170 (E.L.M.) and K99 DA023694 (L.S.).
Author information
Authors and Affiliations
Contributions
Y.Z. and M.Q. designed and performed transport and binding studies; L.S. and E.L.M. designed, carried out, and analyzed the computational simulations and pKa calculations. H.W. and J.A.J. helped to design experiments and analyze data related to the computational and biochemical studies, respectively. Y.Z., M.Q., L.S., E.L.M., H.W. and J.A.J. contributed to writing and editing the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Methods, Supplementary Table 1 and Supplementary Figures 2–3 (PDF 184 kb)
Rights and permissions
About this article
Cite this article
Zhao, Y., Quick, M., Shi, L. et al. Substrate-dependent proton antiport in neurotransmitter:sodium symporters. Nat Chem Biol 6, 109–116 (2010). https://doi.org/10.1038/nchembio.284
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.284
This article is cited by
-
Elucidating the Mechanism Behind Sodium-Coupled Neurotransmitter Transporters by Reconstitution
Neurochemical Research (2022)
-
The Lepidopteran KAAT1 and CAATCH1: Orthologs to Understand Structure–Function Relationships in Mammalian SLC6 Transporters
Neurochemical Research (2022)
-
Quantifying secondary transport at single-molecule resolution
Nature (2019)
-
A partially-open inward-facing intermediate conformation of LeuT is associated with Na+ release and substrate transport
Nature Communications (2018)
-
Single-molecule fluorescence studies on the conformational change of the ABC transporter MsbA
Biophysics Reports (2018)