Abstract
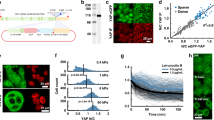
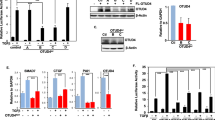
Basic fibroblast growth factor (bFGF or FGF-2) exerts its pleiotropic activities both as an exogenous and an intracellular factor. FGF-1 and FGF-2 are prototypes for this dual signalling, but the mechanisms of their intracellular actions remain unknown. Here we show that Translokin, a cytoplasmic protein of relative molecular mass 55,000 (Mr 55K), interacts specifically with the 18K form of FGF-2. Translokin is ubiquitously expressed and colocalizes with the microtubular network. As Translokin does not interact with FGF-1, we used a strategy based on FGF-1–FGF-2 chimaeras to map the interacting regions in FGF-2 and to generate Nb1a2, a non-interacting variant of FGF-2. Although most of the FGF-2 properties are preserved in Nb1a2, this variant is defective in intracellular translocation and in stimulating proliferation. The fusion of a nuclear localization signal to Nb1a2 restores its mitogenic activity and its nuclear association. Inhibiting Translokin expression by RNA interference reduces the translocation of FGF-2 without affecting the intracellular trafficking of FGF-1. Our data show that the nuclear association of internalized FGF-2 is essential for its mitogenic activity and that Translokin is important in this translocation pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ornitz, D.M. & Itoh, N. Fibroblast growth factors. Genome Biol. 2, 3005.1–3005.12 (2001). http://genomebiology.com/2001/2/3/reviews/3005
Bikfalvi, A., Klein, S., Pintucci, G. & Rifkin, D.B. Biological roles of fibroblast growth factor-2. Endocr. Rev. 18, 26–45 (1997).
Prats, H. et al. High molecular mass forms of basic fibroblast growth factor are initiated by alternative CUG codons. Proc. Natl Acad. Sci. USA 86, 1836–1840 (1989).
Arnaud, E. et al. A new 34-kilodalton isoform of human fibroblast growth factor 2 is cap dependently synthesized by using a non-AUG start codon and behaves as a survival factor. Mol. Cell. Biol. 19, 505–514 (1999).
Bugler, B., Amalric, F. & Prats, H. Alternative initiation of translation determines cytoplasmic or nuclear localization of basic fibroblast growth factor. Mol. Cell. Biol. 11, 573–577 (1991).
Johnson, D.E. & Williams, L.T. Structural and functional diversity in the FGF receptor multigene family. Adv. Cancer Res. 60, 1–41 (1993).
Cross, M.J. & Claesson-Welsh, L. FGF and VEGF function in angiogenesis: signalling pathways, biological responses and therapeutic inhibition. Trends Pharmacol. Sci. 22, 201–207 (2001).
Reiland, J. & Rapraeger, A.C. Heparan sulfate proteoglycan and FGF receptor target basic FGF to different intracellular destinations. J. Cell Sci. 105, 1085–1093 (1993).
Roghani, M. & Moscatelli, D. Basic fibroblast growth factor is internalized through both receptor-mediated and heparan sulfate-mediated mechanisms. J. Biol. Chem. 267, 22156–22162 (1992).
Baldin, V., Roman, A.M., Bosc-Bierne, I., Amalric, F. & Bouche, G. Translocation of bFGF to the nucleus is G1 phase cell cycle specific in bovine aortic endothelial cells. EMBO J. 9, 1511–1517 (1990).
Patry, V., Arnaud, E., Amalric, F. & Prats, H. Involvement of basic fibroblast growth factor NH2 terminus in nuclear accumulation. Growth Factors 11, 163–174 (1994).
Patry, V., Bugler, B., Maret, A., Potier, M. & Prats, H. Endogenous basic fibroblast growth factor isoforms involved in different intracellular protein complexes. Biochem. J. 326, 259–264 (1997).
Nagase, T. et al. Prediction of the coding sequences of unidentified human genes. III. The coding sequences of 40 new genes (KIAA0081–KIAA0120) deduced by analysis of cDNA clones from human cell line KG-1. DNA Res. 2, 37–43 (1995).
Bailly, K., Soulet, F., Leroy, D., Amalric, F. & Bouche, G. Uncoupling of cell proliferation and differentiation activities of basic fibroblast growth factor. FASEB J. 14, 333–344 (2000).
Zhu, X. et al. Three-dimensional structures of acidic and basic fibroblast growth factors. Science 251, 90–93 (1991).
Pages, G. et al. Mitogen-activated protein kinases p42mapk and p44mapk are required for fibroblast proliferation. Proc. Natl Acad. Sci. USA 90, 8319–8323 (1993).
Kanda, S. et al. Phosphatidylinositol 3′-kinase-independent p70S6 kinase activation by fibroblast growth factor receptor-1 is important for proliferation but not differentiation of endothelial cells. J. Biol. Chem. 272, 23347–23353 (1997).
Bouche, G. et al. Basic fibroblast growth factor enters the nucleolus and stimulates the transcription of ribosomal genes in ABAE cells undergoing G0–G1 transition. Proc. Natl Acad. Sci. USA 84, 6770–6774 (1987).
Clarke, W.E., Berry, M., Smith, C., Kent, A. & Logan, A. Coordination of fibroblast growth factor receptor 1 (FGFR1) and fibroblast growth factor-2 (FGF-2) trafficking to nuclei of reactive astrocytes around cerebral lesions in adult rats. Mol. Cell. Neurosci. 17, 17–30 (2001).
Joy, A. et al. Nuclear accumulation of FGF-2 is associated with proliferation of human astrocytes and glioma cells. Oncogene 14, 171–183 (1997).
Elbashir, S.M. et al. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411, 494–498 (2001).
Keresztes, M. & Boonstra, J. Import(ance) of growth factors in(to) the nucleus. J. Cell Biol. 145, 421–424 (1999).
Imamura, T. et al. Recovery of mitogenic activity of a growth factor mutant with a nuclear translocation sequence. Science 249, 1567–1570 (1990).
Wiedlocha, A., Falnes, P.O., Madshus, I.H., Sandvig, K. & Olsnes, S. Dual mode of signal transduction by externally added acidic fibroblast growth factor. Cell 76, 1039–1051 (1994).
Bonnet, H. et al. Fibroblast growth factor-2 binds to the regulatory β subunit of CK2 and directly stimulates CK2 activity toward nucleolin. J. Biol. Chem. 271, 24781–24787 (1996).
Skjerpen, C.S., Nilsen, T., Wesche, J. & Olsnes, S. Binding of FGF-1 variants to protein kinase CK2 correlates with mitogenicity. EMBO J. 21, 4058–4069 (2002).
Shen, B., Arese, M., Gualandris, A. & Rifkin, D.B. Intracellular association of FGF-2 with the ribosomal protein L6/TAXREB107. Biochem. Biophys. Res. Commun. 252, 524–528 (1998).
Van den Berghe, L . et al. FIF [fibroblast growth factor-2 (FGF-2)-interacting-factor], a nuclear putatively antiapoptotic factor, interacts specifically with FGF-2. Mol. Endocrinol. 14, 1709–1724 (2000).
Reilly, J.F. & Maher, P.A. Importin beta-mediated nuclear import of fibroblast growth factor receptor: role in cell proliferation. J. Cell Biol. 152, 1307–1312 (2001).
Gleizes, P.E., Noaillac-Depeyre, J., Dupont, M.A. & Gas, N. Basic fibroblast growth factor (FGF-2) is addressed to caveolae after binding to the plasma membrane of BHK cells. Eur. J. Cell Biol. 71, 144–153 (1996).
Kudla, A.J. et al. The FGF receptor-1 tyrosine kinase domain regulates myogenesis but is not sufficient to stimulate proliferation. J. Cell Biol. 142, 241–250 (1998).
Huez, I., Bornes, S., Bresson, D., Creancier, L. & Prats, H. New vascular endothelial growth factor isoform generated by internal ribosome entry site-driven CUG translation initiation. Mol. Endocrinol. 15, 2197–2210 (2001).
Estival, A. et al. Differential regulation of fibroblast growth factor (FGF) receptor-1 mRNA and protein by two molecular forms of basic FGF. Modulation of FGFR-1 mRNA stability. J. Biol. Chem. 271, 5663–5670 (1996).
Acknowledgements
We thank G. Bouche for HA–FGF-2 plasmid; D. Bresson and B. Knibiehler for technical advice; and B. Bugler, C. Touriol, A. C. Prats and F. Bayard for discussions and critically reading the manuscript. C.B. was supported by the Association pour la Recherche contre le Cancer; H.L. was supported by an INSERM fellowship. This work was supported by the Association pour la Recherche contre le Cancer and the Ligue Nationale contre le Cancer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figure
Figure S1. Translokin expression is ubiquitous (PDF 833 kb)
Rights and permissions
About this article
Cite this article
Bossard, C., Laurell, H., Van den Berghe, L. et al. Translokin is an intracellular mediator of FGF-2 trafficking. Nat Cell Biol 5, 433–439 (2003). https://doi.org/10.1038/ncb979
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb979
This article is cited by
-
Fibroblast growth factor signaling in axons: from development to disease
Cell Communication and Signaling (2023)
-
Cep57 is a Mis12-interacting kinetochore protein involved in kinetochore targeting of Mad1–Mad2
Nature Communications (2016)
-
A nuclear odyssey: fibroblast growth factor-2 (FGF-2) as a regulator of nuclear homeostasis in the nervous system
Cellular and Molecular Life Sciences (2015)
-
Inhibition of Proliferation of Non-small Cell Lung Cancer Cells by a bFGF Antagonist Peptide
International Journal of Peptide Research and Therapeutics (2014)
-
CEP proteins: the knights of centrosome dynasty
Protoplasma (2013)