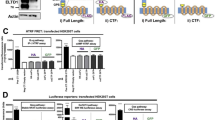
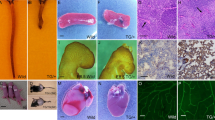
Abstract
Tie2 belongs to the receptor tyrosine kinase family and functions as a receptor for Angiopoietin-1 (Ang1). Gene-targeting analyses of either Ang1 or Tie2 in mice reveal a critical role of Ang1–Tie2 signalling in developmental vascular formation. It remains elusive how the Tie2 signalling pathway plays distinct roles in both vascular quiescence and angiogenesis. We demonstrate here that Ang1 bridges Tie2 at cell–cell contacts, resulting in trans-association of Tie2 in the presence of cell–cell contacts. In clear contrast, in isolated cells, extracellular matrix-bound Ang1 locates Tie2 at cell–substratum contacts. Furthermore, Tie2 activated at cell–cell or cell–substratum contacts leads to preferential activation of Akt and Erk, respectively. Microarray analyses and real-time PCR validation clearly show the differential gene expression profile in vascular endothelial cells upon Ang1 stimulation in the presence or absence of cell–cell contacts, implying downstream signalling is dependent upon the spatial localization of Tie2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Yancopoulos, G. D. et al. Vascular-specific growth factors and blood vessel formation. Nature 407, 242–248 (2000).
Mammoto, T. et al. Angiopoietin-1 requires p190RhoGAP to protect against vascular leakage in vivo. J. Biol. Chem. 282, 23910–23918 (2007).
Jho, D. et al. Angiopoietin-1 opposes VEGF-induced increase in endothelial permeability by inhibiting TRPC1-dependent Ca2 influx. Circ. Res. 96, 1282–1290 (2005).
Brindle, N. P., Saharinen, P. & Alitalo, K. Signaling and functions of angiopoietin-1 in vascular protection. Circ. Res. 98, 1014–1023 (2006).
Baffert, F., Le, T., Thurston, G. & McDonald, D. M. Angiopoietin-1 decreases plasma leakage by reducing number and size of endothelial gaps in venules. Am. J. Physiol. Heart Circ. Physiol. 290, H107–H118 (2005).
Gamble, J. R. et al. Angiopoietin-1 is an antipermeability and anti-inflammatory agent in vitro and targets cell junctions. Circ. Res. 87, 603–607 (2000).
Cho, C. H. et al. Designed angiopoietin-1 variant, COMP-Ang1, protects against radiation-induced endothelial cell apoptosis. Proc. Natl Acad. Sci.USA 101, 5553–5558 (2004).
Kwak, H. J., So, J. N., Lee, S. J., Kim, I. & Koh, G. Y. Angiopoietin-1 is an apoptosis survival factor for endothelial cells. FEBS Lett. 448, 249–253 (1999).
Papapetropoulos, A. et al. Angiopoietin-1 inhibits endothelial cell apoptosis via the Akt/Survivin pathway. J. Biol. Chem. 275, 9102–9105 (2000).
Thurston, G. et al. Angiopoietin-1 protects the adult vasculature against plasma leakage. Nature Med. 6, 460–463 (2000).
Thurston, G. et al. Leakage-resistant blood vessels in mice transgenically overexpressing angiopoietin-1. Science 286, 2511–2514 (1999).
Master, Z. et al. Dok-R plays a pivotal role in angiopoietin-1-dependent cell migration through recruitment and activation of Pak. EMBO J. 20, 5919–5928 (2001).
Kim, I. et al. Angiopoietin-1 induces endothelial cell sprouting through the activation of focal adhesion kinase and plasmin secretion. Circ. Res. 86, 952–959 (2000).
Cho, C. H. et al. COMP-Ang1: A designed angiopoietin-1 variant with nonleaky angiogenic activity. Proc. Natl Acad. Sci. USA 101, 5547–5552 (2004).
Asahara, T. et al. Tie2 receptor ligands, angiopoietin-1 and angiopoietin-2, modulate VEGF-induced postnatal neovascularization. Circ. Res. 83, 233–240 (1998).
Yoon, M. J. et al. Localization of Tie2 and phospholipase D in endothelial caveolae is involved in angiopoietin-1-induced MEK/ERK phosphorylation and migration in endothelial cells. Biochem. Biophys. Res. Commun. 308, 101–105 (2003).
Lin, P. et al. Inhibition of tumor angiogenesis using a soluble receptor establishes a role for Tie2 in pathologic vascular growth. J. Clin. Invest. 100, 2072–2078 (1997).
Lin, P. et al. Antiangiogenic gene therapy targeting the endothelium-specific receptor tyrosine kinase Tie2. Proc. Natl Acad. Sci. USA 95, 8829–8834 (1998).
Peters, K. G. et al. Functional significance of Tie2 signaling in the adult vasculature. Recent Prog. Horm. Res. 59, 51–71 (2004).
Wong, A. L. et al. Tie2 expression and phosphorylation in angiogenic and quiescent adult tissues. Circ. Res. 81, 567–574 (1997).
Carmeliet, P. et al. Targeted deficiency or cytosolic truncation of the VE-cadherin gene in mice impairs VEGF-mediated endothelial survival and angiogenesis. Cell 98, 147–157 (1999).
Zanetti, A. et al. Vascular endothelial growth factor induces SHC association with vascular endothelial cadherin: a potential feedback mechanism to control vascular endothelial growth factor receptor-2 signaling. Arterioscler. Thromb. Vasc. Biol. 22, 617–622 (2002).
Lampugnani, M. G., Orsenigo, F., Gagliani, M. C., Tacchetti, C. & Dejana, E. Vascular endothelial cadherin controls VEGFR-2 internalization and signaling from intracellular compartments. J. Cell Biol. 174, 593–604 (2006).
Zaidel-Bar, R., Cohen, M., Addadi, L. & Geiger, B. Hierarchical assembly of cell-matrix adhesion complexes. Biochem. Soc. Trans. 32, 416–420 (2004).
Geiger, B., Bershadsky, A., Pankov, R. & Yamada, K. M. Transmembrane crosstalk between the extracellular matrix--cytoskeleton crosstalk. Nature Rev. Mol. Cell Biol 2, 793–805 (2001).
Cascone, I., Napione, L., Maniero, F., Serini, G. & Bussolino, F. Stable interaction between α5β1 integrin and Tie2 tyrosine kinase receptor regulates endothelial cell response to Ang-1. J. Cell Biol. 170, 993–1004 (2005).
Wulfkuhle, J. D. et al. Domain analysis of supervillin, an F-actin bundling plasma membrane protein with functional nuclear localization signals. J. Cell Sci. 112, 2125–2136 (1999).
Adams, C. L., Nelson, W. J. & Smith, S. J. Quantitative analysis of cadherin-catenin-actin reorganization during development of cell-cell adhesion. J. Cell Biol. 135, 1899–1911 (1996).
Kim, I. et al. Angiopoietin-1 regulates endothelial cell survival through the phosphatidylinositol 3′-Kinase/Akt signal transduction pathway. Circ. Res. 86, 24–29 (2000).
Kanda, S., Miyata, Y., Mochizuki, Y., Matsuyama, T. & Kanetake, H. Angiopoietin 1 is mitogenic for cultured endothelial cells. Cancer Res. 65, 6820–6827 (2005).
Teichert-Kuliszewska, K. et al. Biological action of angiopoietin-2 in a fibrin matrix model of angiogenesis is associated with activation of Tie2. Cardiovasc. Res. 49, 659–670 (2001).
Weber, C. C. et al. Effects of protein and gene transfer of the angiopoietin-1 fibrinogen-like receptor-binding domain on endothelial and vessel organization. J. Biol. Chem. 280, 22445–22453 (2005).
Dallabrida, S. M., Ismail, N., Oberle, J. R., Himes, B. E. & Rupnick, M. A. Angiopoietin-1 promotes cardiac and skeletal myocyte survival through integrins. Circ. Res. 96, e8-24 (2005).
Aplin, A. E., Short, S. M. & Juliano, R. L. Anchorage-dependent regulation of the mitogen-activated protein kinase cascade by growth factors is supported by a variety of integrin α chains. J. Biol. Chem. 274, 31223–31228 (1999).
Short, S. M., Talbott, G. A. & Juliano, R. L. Integrin-mediated signaling events in human endothelial cells. Mol. Biol. Cell 9, 1969–1980 (1998).
Schlaepfer, D. D. & Mitra, S. K. Multiple connections link FAK to cell motility and invasion. Curr. Opin. Genet. Dev. 14, 92–101 (2004).
Parsons, J. T. Focal adhesion kinase: the first ten years. J. Cell Sci. 116, 1409–1416 (2003).
Eliceiri, B. P., Klemke, R., Stromblad, S., & Cheresh, D. A. Integrin αV β3 requirement for sustained mitogen-activated protein kinase activity during angiogenesis. J. Cell Biol. 140, 1255–1263 (1998).
Daly, C. et al. Angiopoietin-1 modulates endothelial cell function and gene expression via the transcription factor FKHR (FOXO1). Genes Dev. 18, 1060–1071 (2004).
Chen, J. et al. Akt1 regulates pathological angiogenesis, vascular maturation and permeability in vivo. Nature Med. 11, 1188–1196 (2005).
Xu, Y. & Yu, Q. Angiopoietin-1, unlike angiopoietin-2, is incorporated into the extracellular matrix via its linker peptide region. J. Biol. Chem. 276, 34990–34998 (2001).
Carlson, T. R., Feng, Y., Maisonpierre, P. C., Mrksich, M. & Morla, A. O. Direct cell adhesion to the angiopoietins mediated by integrins. J. Biol. Chem. 276, 26516–26525 (2001).
Braren, R. et al. Endothelial FAK is essential for vascular network stability, cell survival, and lamellipodial formation. J. Cell Biol. 172, 151–162 (2006).
Shen, T. L. et al. Conditional knockout of focal adhesion kinase in endothelial cells reveals its role in angiogenesis and vascular development in late embryogenesis. J. Cell Biol. 169, 941–952 (2005).
Meadows, K. N., Bryant, P. & Pumiglia, K. Vascular endothelial growth factor induction of the angiogenic phenotype requires Ras activation. J. Biol. Chem. 276, 49289–49298 (2001).
Yana, I. et al. Crosstalk between neovessels and mural cells directs the site-specific expression of MT1-MMP to endothelial tip cells. J. Cell Sci. 120, 1607–1614 (2007).
Nagashima, K. et al. Adaptor protein Crk is required for ephrin-B1-induced membrane ruffling and focal complex assembly of human aortic endothelial cells. Mol. Biol. Cell 13, 4231–4242 (2002).
Sakurai, A. et al. MAGI-1 is required for Rap1 activation upon cell-cell contact and for enhancement of vascular endothelial cadherin-mediated cell adhesion. Mol. Biol. Cell 17, 966–976 (2006).
Acknowledgements
We are grateful to T. Suda (Keio University, Tokyo, Japan) for the Tie2 cDNA, to A. Fukamizu (University of Tsukuba, Tsukuba, Japan) for the Foxo1 cDNA, to K.M. Yamada (National Institute of Health) for GFP–tensin, to J. Nakae (Kobe University Graduate School of Medicine, Kobe, Japan) for the adenovirus encoding Foxo1 mutant, to A. Mizushima, M. Sone, M. Maeoka, and Y. Matsuura for technical assistance, to M. Masuda, H. Hanada, and S. Yamanoto for helpful advice and to J.T. Pearson and J.S. Gutkind for critical reading of the manuscript. This work was supported in part by grants from the Ministry of Education, Science, Sports and Culture of Japan; the Ministry of Health, Labour, and Welfare of Japan; and the Program for the Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation (to S.F., T.M, T.K. N.M.); the Naito Foundation (to S.F.); Takeda Medical Research Foundation (to N.M); and KOSEF through the NRL Program (2004-02376 to G.Y.K) funded by the MOST.
Author information
Authors and Affiliations
Contributions
S. F. and N. M. designed and wrote the paper. S. F. performed the all cell biological and biochemical analysis. K. S. and K. N. helped with the experiments performed by S. F. T. M. and T. K. performed microarray analyses. M. S. and N. T. helped with VEGF-related and Tie2–BaF experiments. H. Z. K. and G. Y. K. prepared several forms of recombinant Ang1.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4, S5, S6, S7, S8 and Supplemntary Methods (PDF 3113 kb)
Supplementary Information
Supplementary Movie 1 (MOV 2802 kb)
Supplementary Information
Supplementary Movie 2 (MOV 2906 kb)
Supplementary Information
Supplementary Movie 3 (MOV 2727 kb)
Supplementary Information
Supplementary Movie 4 (MOV 2926 kb)
Supplementary Information
Supplementary Movie 5 (MOV 3003 kb)
Supplementary Information
Supplementary Movie 6 (MOV 2995 kb)
Supplementary Information
Supplementary Movie 7 (MOV 3030 kb)
Rights and permissions
About this article
Cite this article
Fukuhara, S., Sako, K., Minami, T. et al. Differential function of Tie2 at cell–cell contacts and cell–substratum contacts regulated by angiopoietin-1. Nat Cell Biol 10, 513–526 (2008). https://doi.org/10.1038/ncb1714
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1714
This article is cited by
-
The CLDN5 gene at the blood-brain barrier in health and disease
Fluids and Barriers of the CNS (2023)
-
In vitro inhibition of cancer angiogenesis and migration by a nanobody that targets the orphan receptor Tie1
Cellular and Molecular Life Sciences (2022)
-
Caveolae-mediated Tie2 signaling contributes to CCM pathogenesis in a brain endothelial cell-specific Pdcd10-deficient mouse model
Nature Communications (2021)
-
Cellular crosstalk regulates the aqueous humor outflow pathway and provides new targets for glaucoma therapies
Nature Communications (2021)
-
A systems biology model of junctional localization and downstream signaling of the Ang–Tie signaling pathway
npj Systems Biology and Applications (2021)