Abstract
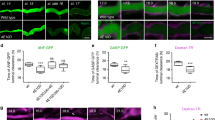
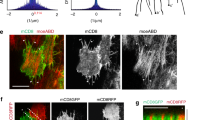
The actin cytoskeleton orders cellular space and transduces many of the forces required for morphogenesis. Here we combine genetics and cell biology to identify genes that control the polarized distribution of actin filaments within the Drosophila follicular epithelium. We find that profilin and cofilin regulate actin-filament formation throughout the cell cortex. In contrast, CAP—a Drosophila homologue of Adenylyl Cyclase Associated Proteins—functions specifically to limit actin-filament formation catalysed by Ena at apical cell junctions. The Abl tyrosine kinase also collaborates in this process. We therefore propose that CAP, Ena and Abl act in concert to modulate the subcellular distribution of actin filaments in Drosophila.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Pruyne, D. & Bretscher, A. Polarization of cell growth in yeast. J. Cell Sci. 113, 571–585 (2000).
Mitchison, T. J. & Cramer, L. P. Actin-based cell motility and cell locomotion. Cell 84, 371–379 (1996).
Vasioukhin, V., Bauer, C., Yin, M. & Fuchs, E. Directed actin polymerization is the driving force for epithelial cell–cell adhesion. Cell 100, 209–2019 (2000).
Lanier, L. M. & Gertler, F. B. From Abl to actin: Abl tyrosine kinase and associated proteins in growth cone motility. Curr. Opin. Neurobiol. 10, 80–87 (2000).
Wegner, A. Head to tail polymerization of actin. J. Mol. Biol. 108, 139–150 (1976).
Pollard, T. D. Rate constants for the reactions of ATP- and ADP-actin with the ends of actin filaments. J. Cell Biol. 103, 2747–2754 (1986).
Pollard, T. D., Blanchoin, L. & Mullins, R. D. Actin dynamics. J. Cell Sci. 114, 3–4 (2001).
Beckerle, M. C. Spatial control of actin filament assembly: lessons from Listeria. Cell 95, 741–748 (1998).
Laurent, V. et al. Role of proteins of the Ena/VASP family in actin-based motility of Listeria monocytogenes. J. Cell Biol. 144, 1245–1258 (1999).
Machesky, L. M. & Gould, K. L. The Arp2/3 complex: a multifunctional actin organizer. Curr. Opin. Cell Biol. 11, 117–121 (1999).
Ahern-Djamali, S. M. et al. Identification of profilin and Src homology 3 domains as binding partners for Drosophila enabled. Proc. Natl Acad. Sci. USA 96, 4977–4982 (1999).
Goldschmidt-Clermont, P. J. et al. The control of actin nucleotide exchange by thymosin beta 4 and profilin. A potential regulatory mechanism for actin polymerization in cells. Mol. Biol. Cell 3, 1015–1024 (1992).
Yang, C. et al. Profilin enhances Cdc42-induced nucleation of actin polymerization. J. Cell Biol. 150, 1001–1012 (2000).
Gieselmann, R. & Mann, K. ASP-56, a new actin sequestering protein from pig platelets with homology to CAP, an adenylate cyclase-associated protein from yeast. FEBS Lett. 298, 149–153 (1992).
Freeman, N. L., Chen, Z., Horenstein, J., Weber, A. & Field, J. An actin monomer binding activity localizes to the carboxyl-terminal half of the Saccharomyces cerevisiae cyclase-associated protein. J. Biol. Chem. 270, 5680–5685 (1995).
Amberg, D. C., Basart, E. & Botstein, D. Defining protein interactions with yeast actin in vivo. Nature Struct. Biol. 2, 28–35 (1995).
Gottwald, U., Brokamp, R., Karakesisoglou, I., Schleicher, M. & Noegel, A. A. Identification of a cyclase-associated protein (CAP) homologue in Dictyostelium discoideum and characterization of its interaction with actin. Mol. Biol. Cell 7, 261–272 (1996).
Bamburg, J. R. Proteins of the ADF/cofilin family: essential regulators of actin dynamics. Annu. Rev. Cell. Dev. Biol. 15, 185–230 (1999).
Spradling, A. in The development of Drosophila melanogaster (eds Bate, M. & Arias, A. M.) 1–70 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1993).
Muller, H. A. Genetic control of epithelial cell polarity: lessons from Drosophila. Dev. Dyn. 218, 52–67 (2000).
Duffy, J. B., Harrison, D. A. & Perrimon, N. Identifying loci required for follicular patterning using directed mosaics. Development 125, 2263–2271 (1998).
Sutherland, J. D. & Witke, W. Molecular genetic approaches to understanding the actin cytoskeleton. Curr. Opin. Cell Biol. 11, 142–151 (1999).
Baum, B., Li, W. & Perrimon, N. A cyclase-associated protein regulates actin and cell polarity during Drosophila oogenesis and in yeast. Curr. Biol. 10, 964–973 (2000).
Benlali A., Draskovic, I., Hazelett D. J. & Treisman J. E. act up controls actin polymerization to alter cell shape and restrict hedgehog signaling in the Drosophila eye disc. Cell 101, 271–281 (2000).
Gertler, F. B. et al. enabled, a dosage-sensitive suppressor of mutations in the Drosophila Abl tyrosine kinase, encodes an Abl substrate with SH3 domain-binding properties. Genes Dev. 9, 521–533 (1995).
Gertler, F. B., Niebuhr, K., Reinhard, M., Wehland, J. & Soriano, P. Mena, a relative of VASP and Drosophila Enabled, is implicated in the control of microfilament dynamics. Cell 87, 227–239 (1996).
Ahern-Djamali, S. M. et al. Mutations in Drosophila enabled and rescue by human vasodilator-stimulated phosphoprotein (VASP) indicate important functional roles for Ena/VASP homology domain 1 (EVH1) and EVH2 domains. Mol. Biol. Cell 9, 2157–2171 (1998).
Freeman, N. L. et al. A conserved proline-rich region of the Saccharomyces cerevisiae cyclase-associated protein binds SH3 domains and modulates cytoskeletal localization. Mol. Cell. Biol. 16, 548–556 (1996).
Comer, A. R., Ahern-Djamali, S. M., Juang, J. L., Jackson, P. D. & Hoffmann, F. M. Phosphorylation of Enabled by the Drosophila Abelson tyrosine kinase regulates the in vivo function and protein–protein interactions of Enabled. Mol. Cell. Biol. 18, 152–160 (1998).
Wills, Z., Bateman, J., Korey, C. A., Comer, A. & Van Vactor, D. The tyrosine kinase Abl and its substrate enabled collaborate with the receptor phosphatase Dlar to control motor axon guidance. Neuron 22, 301–312 (1999).
Gunsalus, K. C. et al. Mutations in twinstar, a Drosophila gene encoding a cofilin/ADF homologue, result in defects in centrosome migration and cytokinesis. J. Cell Biol. 131, 1243–1259 (1995).
Cooley, L., Verheyen, E. & Ayers, K. chickadee encodes a profilin required for intercellular cytoplasm transport during Drosophila oogenesis. Cell 69, 173–184 (1992).
Theriot, J. A. & Mitchison, T. J. The three faces of profilin. Cell 75, 835–838 (1993).
Verheyen, E. M. & Cooley, L. Profilin mutations disrupt multiple actin-dependent processes during Drosophila development. Development 120, 717–728 (1994).
Bilder, D., Li, M. & Perrimon, N. Cooperative regulation of cell polarity and growth by Drosophila tumor suppressors. Science 289, 113–116 (2000).
Reinhard, M. et al. The 46/50 kDa phosphoprotein VASP purified from human platelets is a novel protein associated with actin filaments and focal contacts. EMBO J. 11, 2063–2070 (1992).
Goode, S. & Perrimon, N. Inhibition of patterned cell shape change and cell invasion by Discs large during Drosophila oogenesis. Genes Dev. 11, 2532–2544 (1997).
Koleske, A. J. et al. Essential roles for the Abl and Arg tyrosine kinases in neurulation. Neuron 21, 1259–1272 (1998).
Winter, C. G. et al. Drosophila Rho-associated kinase (Drok) links Frizzled-mediated planar cell polarity signaling to the actin cytoskeleton. Cell 105, 81–91 (2001).
Chou, T. B. & Perrimon, N. The autosomal FLP-DFS technique for generating germline mosaics in Drosophila melanogaster. Genetics 144, 1673–1679 (1996).
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993).
Acknowledgements
We thank Zak Wills, Jack Bateman, David Van Vactor and David Bilder for help throughout this project; T. Schupbach, K. Gunsalus, D. St Johnston and the Bloomington Stock Center for Drosophila stocks, and D. Brenton, M. Hoffmann and E. Knust for antibodies; and B. Stronach, S. Noselli, D. Bilder, M. Schober, D. Van Vactor, A. Kiger and F. Gertler for their comments on the manuscript. B.B. was supported by EMBO, HFSP and HHMI, and N.P. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baum, B., Perrimon, N. Spatial control of the actin cytoskeleton in Drosophila epithelial cells. Nat Cell Biol 3, 883–890 (2001). https://doi.org/10.1038/ncb1001-883
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1001-883
This article is cited by
-
Hedgehog morphogen gradient is robust towards variations in tissue morphology in Drosophila
Scientific Reports (2023)
-
Transmission of cytokinesis forces via E-cadherin dilution and actomyosin flows
Nature (2017)
-
E-cadherin roles in animal biology: A perspective on thyroid hormone-influence
Cell Communication and Signaling (2016)
-
Domestication-driven Gossypium profilin 1 (GhPRF1) gene transduces early flowering phenotype in tobacco by spatial alteration of apical/floral-meristem related gene expression
BMC Plant Biology (2016)
-
Drosophila follicle stem cells are regulated by proliferation and niche adhesion as well as mitochondria and ROS
Nature Communications (2012)