Abstract
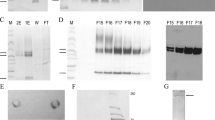
Two forms of human interleukin–1 have been expressed in E. coli. Acid extraction of the cellular suspensions solubilizes rIL–1α and rIL–1β from inclusion bodies while precipitating most of the E. coli proteins. This leads to a facile purification scheme that provides sufficient quantities of highly active IL–1 to undertake biological characterization of these important immunoregulatory molecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mizel, S.B. 1982. Interleukin 1 and T cell activation. Immunol. Rev. 63:51–72.
Oppenheim, J.J. and Gery, I. 1982. Interleukin 1 is more than an interleukin. Immunol. Today 3:113–118.
Rosenwasser, L.J., Dinarello, C.A. and Rosenthal, A.S. 1979. Adherent-cell function in murine T-lymphocyte antigen recognition. IV. Enhancement of murine T-cell antigen recognition by human leukocyte pyrogen. J. Exp. Med. 150:709–714.
Farrar, J.J. and Koopman, W.J. 1978. Characterization of macrophage and lymphocyte-derived mitogenic factors and their effect on the antibody response in vitro, p. 325–346. In: Biology of the Lymphokines. S. Cohen, E. Pick and J. J. Oppenheim (eds.). Academic Press, New York.
Rosenberg, S.A., Lotze, M.T., Muul, L.M., Leitman, S., et al. 1985. Observations on the systemic administration of autologous lymphokine-activated killer cells and recombinant interleukin-2 to patients with metastatic cancer. Nature 313:1485–1492.
Fontana, A.H., Hengartner, E., Weber, K., Fehr, P.J. Grob and Cohen, G. 1982. Interleukin 1 activity in the synovial fluid of patients with rheumatoid arthritis. Rheumatoid Int. 2:49–53.
Gowen, M., Wood, D.D., Ihrie, E.J., McGuire, M.K.B., and Russell, G.G. 1983. An interleukin 1 like factor stimulates bone resorption in vitro. Nature 306:378–380.
March, C.J., Mosley, B., Larsen, A., Cerretti, D.P., et al. 1985. Cloning, sequence, and expression of two distinct human interleukin-1 complementary DNAs. Nature 315:641–647.
Maniatis, T., Ptashne, M., Backmann, K., Kleid, D., Flashman, S., Jeffrey, A. and Mauer, R. 1975. Recognition sequences of repressor and polymerase in the operators of bacteriophage lambda. Cell 51:107–113.
Schoner, B.E., Hsiung, H.M., Belagaje, R.M., Mayne, N.G. and Schoner, R.G. 1984. Role of mRNA translational efficiency in bovine growth hormone expression in Escherchia coli. Proc. Natl. Acad. Sci. USA 81:5403–5407.
Baughman, G. and Nomura, M. 1983. Localization of the target site for translational regulation of the L11 operon and direct evidence for translational coupling in Escherichia coli. Cell 34:979–988.
Bernard, H.U., Ramaut, E., Hershfield, M.V., Das, H.K., Helinski, D.R., Yanofsky, C. and Franklin, N. 1979. Construction of plasmid cloning vehicles that promote gene expression from the bacteriophage lambda PL promoter. Gene 5:59–76.
Maniatis, T., Fritsch, E.F. and Sambrook, J. 1982. Molecular Cloning, A Laboratory Manual. Cold Spring Harbor Laboratory, New York.
Gubler, U., Chua, A.O., Stern, A.S., Hellmann, C.P., et al. 1986. Recombinant human interleukin 1α: Purification and biological characterization. J. Immunol. 136:2492–2497.
Kronheim, S.R., March, C.J., Erb, S.K., Conlon, P.J., Mochizuki, D.Y. and Hopp, T.P. 1985. Human interleukin-1, purification to homogeneity. J. Exp. Med. 161:490–502.
Wolff, S.M. Auron, P.E., Dinarello, C.A. Rosenwasser, L.J., Webb, A.C. and Rich, A. 1986. Clone controversy at Immunex. Nature 319:270.
Taniguchi, T., Matsui, H., Fujita, T., Takaoka, C., Kashima, N., Yoshimoto, R. and Hamuro, J. 1983. Structure and expression of a cloned cDNA for human interleukin-2. Nature 302:305–310.
Marquis, D., Smolec, J.M. and Katz, D.H. 1985. J. Cell. Biochem., Supplement 9B, Abstract 990, 221.
Jay, G., Khoury, G., Seth, A.K. and Jay, E. 1981. Construction of a general vector for efficient expression of mammalian proteins in bacteria: Use of a synthetic ribosome binding site. Proc. Natl. Acad. Sci. USA 78:5543–5548.
Mott, J.E., Grant R.A., Ito, Y.S. and Platt, T. 1985. Maximizing gene expression from plasmid vectors containing the λ PL promoter: Strategies for overproducing transcription termination factor rho. Proc. Natl. Acad. Sci. USA 82:88–92.
Shine, J. and Dalgarno, L. 1975. Determinant of cistron specificity in bacterial ribosomes. Nature 254:34–38.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kronheim, S., Cantrell, M., Deeley, M. et al. Purification and Characterization of Human Interleukin–1 Expressed in Escherichia coli. Nat Biotechnol 4, 1078–1082 (1986). https://doi.org/10.1038/nbt1286-1078
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1286-1078