Abstract
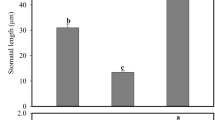
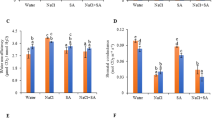
Transgenic tobacco seedlings that overexpress a cDNA encoding an enzyme with both glutathione S-transferase (GST) and glutathione peroxidase (GPX) activity had GST- and GPX-specific activities approximately twofold higher than wild-type seedlings. These GST/GPX overexpressing seedlings grew significantly faster than control seedlings when exposed to chilling or salt stress. During chilling stress, levels of oxidized glutathione (GSSG) were significantly higher in transgenic seedlings than in wild-types. Growth of wild-type seedlings was accelerated by treatment with GSSG, while treatment with reduced glutathione or other sulfhydryl-reducing agents inhibited growth. Therefore, overexpression of GST/GPX can stimulate seedling growth under chilling and salt stress, and this effect could be caused by oxidation of the glutathione pool.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Coles, B. and Ketterer, B. 1990. The role of glutathione and glutathione S-trans-ferases in chemical carcinogenesis. CRC Crit. Rev. Biochem. 25: 47–70.
Mannervik, B. and Danielson, U.H. 1988. Glutathione transferases: structure and catalytic activity. CRC Crit. Rev. Biochem. 23: 283–337.
Pickett, B. and Lu, A.Y.H., 1989. S-transferases: gene structure, regulation, and biological function. Annu. Rev. Biochem. 58: 743–76.
Hatzios, K.K. 1989. pp. 65ndash;101 in Crop safeners for herbicides: development, uses and mechanisms of action. Hatzios, K. and Hoagland, R. (eds.) Academic Press Inc., New York, NY.
Ulmasov, T., Ohmiya, A., Hagen, G., and Guilfoyle, T. 1995. The soybean GH2/4 gene that encodes a glutathione s-transferase has a promoter that is activated by a wide range of chemical agents. Plant Physiol. 108: 919–927.
Roxas, V.P. 1996. Molecular analysis and expression of GST-coding genes in tobacco. Ph.D. Dissertation, Texas Tech University, Lubbock, TX.
Fuerst, E.P., Irzyk, G.P., and Miller, K.D. 1993. Partial characterization of GST isozymes induced by the herbicide safener benoxacor in maize. Plant Physiol. 102: 795–802.
Berhane, K., Widersten, M., Engstrom, A., Kozarich, J.W., and Mannervik, B. 1994. Detoxification of base propenals and other α,β unsaturated aldehyde products of radical reactions and lipid peroxidation by GSTs. Proc. Natl. Acad. Sci. USA 91: 1480–1484.
Bartling, D., Radzio, R., Steiner, U., and Weiler, E. 1993. A glutathione S-transferase with glutathione-peroxidase activity from Arabidopsis thaliana: molecular cloning and functional characterization. Eur. J. Biochem. 216: 579–586.
Criqui, M.C., Jamet, E., Parmentier, Y., Marbach, J., Durr, A., and Fleck, J. 1992. Isolation and characterization of a plant cDNA showing homology to animal glutathione peroxidases. Plant Mol. Biol. 18: 623–627.
Beeor-Tzahar, T., Ben-Hayyim, G., Holland, D., Faltin, Z., and Eshdat, Y. 1993. A stress-associated protein is a distinct plant phospholipid hydroperoxide glutathione peroxidase. FEBS Lett. 366: 151–155.
Allen, R.D. 1995. Dissection of oxidative stress tolerance using transgenic plants. Plant Physiol. 107: 1–7.
Bowler, C., Slooten, L., Vandenbranden, S., De Rycke, R., Botterman, J., Sybesma, C., et al. 1991. Manganese superoxide dismutase can reduce cellular damage mediated by oxygen radicals in transgenic plants. EMBO J. 10: 1723–1732.
Sen Gupta, A., Webb, R.P., Holaday, A.S., and Allen, R.D. 1993. Overexpression of superoxide dismutase protects plants from oxidative stress. Plant Physiol. 103: 1067–1073.
Van Camp, W., Capiau, K., Van Montague, M., Inzé, D., and Slooten, L. 1996. Enhancement of oxidative stress tolerance in transgenic tobacco plants overproducing Fe-superoxide dismutase in chloroplasts. Plant Physiol. 112: 1703–1714.
Aono, M., Kubo, A., Saji, H., Tanaka, K., and Kondo, N. 1993. Enhanced tolerance to photooxidative stress of transgenic Nicotiana tabacum with high chloro-plastic glutathione reductase activity. Plant Cell Physiol. 34: 129–136.
Broadbent, P., Creissen, G.P., Kular, B., Wellbum, A.R., and Mullineaux, P. 1995. Oxidative stress responses in transgenic tobacco containing altered levels of glutathione reductase activity. Plant J. 8: 247–255.
Foyer, C.H., Lelandais, M., Galap, C., and Kunert, K.J. 1991. Effects of elevated cytosolic glutathione reductase activity on the cellular glutathione pool and photosynthesis in leaves under normal and stress conditions. Plant Physiol. 97: 863–872.
Droog, F.N.J., Hooykaas, P.J.J., Libbenga, K.R., and van der Zaal, E.J. 1993. Proteins encoded by an auxin-regulated gene family of tobacco share limited but significant homology with glutathione S-transferases and one member indeed shows in vitro GST activity. Plant Mol. Biol. 21: 965–972.
Takahashi, Y. and Nagata, T. 1992. parB: An auxin-regulated gene encoding glutathione S-transferase. Proc. Natl. Acad. Sci. USA 89: 56–59.
Webb, R.W. 1995. Isolation, characterization, and overexpression of pea cytosolic ascorbate peroxidase. Ph.D. Dissertation. Texas Tech University, Lubbock, TX.
Alscher, RG. 1989. Biosynthesis and antioxidant function of glutathione in plants. Physiol. Plant. 77: 457–464.
Brigelius, R. 1985. pp. 243–272 in Oxidative stress. Sies, H. (ed.) Academic Press Inc., London.
Wolin, M.S. and Mohazzab-H, K.M. 1996. pp. 21–48 in Oxidative stress and the molecular biology of antioxidant defenses. Scandalios, J.G. (ed.) Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
Lozano, M., Wong, J.H., Yee, B.C., Peters, A., Kqbrehel, K., and Buchanan, B.B. 1996. New evidence for a role for thioredoxin h in germination and seedling development. Planta 200: 100–106.
Wingate, V.P.M., Lawton, M.A., and Lamb, C.J. 1988. Glutathione causes a massive and selective induction of plant defense genes. Plant Physiol. 87: 206–210.
Habig, W.H. and Jakoby, W.A. 1981. Assays for differentiation of GST. Methods Enzymol. 77: 735–740.
Mozer, T.J., Tiemeier, D.C., and Jaworksi, E.G. 1983. Purification and characterization of corn glutathione S-transferase. Biochem. 22: 1068–1072.
Tappel, A.L. 1978. Glutathione peroxidase and hydroperoxidase. Methods Enzymol. 52: 506–513.
Horsch, R.B., Fry, J.E., Hofman, N.J., Eicholtz, D., Rogers, S.G., and Fraley, R.T. 1985. A simple and general method for transferring genes into plants. Science 227: 119–123.
Griffith, O.W. 1980. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 106: 207–212.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roxas, V., Smith, R., Allen, E. et al. Overexpression of glutathione S-transferase/glutathioneperoxidase enhances the growth of transgenic tobacco seedlings during stress. Nat Biotechnol 15, 988–991 (1997). https://doi.org/10.1038/nbt1097-988
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1097-988
This article is cited by
-
Embryo growth alteration and oxidative stress responses in germinating Cucurbita pepo seeds exposed to cadmium and copper toxicity
Scientific Reports (2024)
-
Uncovering the transcriptional responses of tobacco (Nicotiana tabacum L.) roots to Ralstonia solanacearum infection: a comparative study of resistant and susceptible cultivars
BMC Plant Biology (2023)
-
Effects of low nitrogen on seedling growth, photosynthetic characteristics and antioxidant system of rice varieties with different nitrogen efficiencies
Scientific Reports (2023)
-
Genome-wide analysis of the Glutathione S-Transferase family in wild Medicago ruthenica and drought-tolerant breeding application of MruGSTU39 gene in cultivated alfalfa
Theoretical and Applied Genetics (2022)
-
Metabolic signatures of Arabidopsis thaliana abiotic stress responses elucidate patterns in stress priming, acclimation, and recovery
Stress Biology (2022)