Abstract
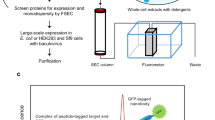
The preparation of pure and homogeneous membrane proteins or membrane protein complexes is time consuming, and the yields are frequently insufficient for structural studies. To circumvent these problems we established an indirect immunoaffinity chromatography method based on engineered Fv fragments. cDNAs encoding the variable domains of hybridoma-derived antibodies raised against various membrane proteins were cloned and expressed in Escherichia coli. The Fv fragments were engineered to serve as bifunctional adaptor molecules. The Fv fragment binds to the epitope of the membrane protein, while the Strep tag affinity peptide, which was fused to the carboxy-terminus of the VH chain, immobilizes the antigen-Fv complex on a streptavidin sepharose column. The usefulness of this technique is illustrated with membrane protein complexes from Paracoccus denitrificans, namely, the cytochrome c oxidase (EC 1.9.3.1), the ubiquinol:cytochrome c oxidoreductase (EC 1.10.2.2), and subcomplexes or individual subunits thereof. These membrane proteins were purified simply by combining the crude P. denitrificans membrane preparation with the E. coli periplasmic cell fraction containing the corresponding Fv fragment, followed by solubilization and streptavidin affinity chromatography. Pure and highly active membrane protein complexes were eluted in the Fv-bound form using diaminobiotin for mild competitive displacement of the Strep tag. The affinity column could thus be reused under continuous operation for several months. Five to 10 mg of membrane protein complexes could be obtained without any detectable impurities within five hours.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Yang, X. and Trumpower, B.L. 1986. Purification of a three-subunit ubiquinol-cytochrome c oxidoreductase complex from Paracoccus denitrificans. J. Biol. Chem. 261: 12282–12289.
Hendler, R.W., Pardhasaradhi, K., Reynafarje, B. and Ludwig, B. 1991. Comparison of energy-transducing capabilities of the two-and three-subunit cytochromes aa3 from Paracoccus denitrificans and the 13-subunit beef heart enzyme. Biophys. J. 60: 415–423.
Bill, K., Broger, C. and Azzi, A. 1982. Affinity chromatography purification of cytochrome c oxidase and b-c1 complex from beef heart mitochondria. Biochim. Biophys. Acta 679: 28–34.
Schertler, G.F.X. 1992. Overproduction of membrane proteins. Curr. Opin. Struct. Biol. 2: 534–544.
Nygren, P.-A., Stahl, S. and Uhlen M. 1994. Engineering proteins to facilitate bioprocessing. Trends Biotechnol. 2: 184–188.
Cuatrecases, P., Wilchek, M. and Anfinsen, C.B. 1968. Selective enzyme purification by affinity chromatography. Proc. Natl. Acad. Sci. USA 61: 636–643.
Köhler, G. and Milstein, C. 1975. Continous cultures of fused cells secreting antibody of predefined specificity. Nature 256: 495–497.
Secher, D.S. and Burke, D.C. 1980. A monoclonal antibody for large-scale purification of human leukocyte interferon. Nature 285: 446–450.
Campbell, D.H., Luescher, E. and Lerman, L.S. 1951. Immunologic adsorbents. I. Isolation of antibody by means of a cellulose-protein antigen. Proc. Nad. Acad. Sci. 37: 575–578.
Gurvich, A.E. and Drizlikh, G.I. 1964. Use of antibodies on an insoluble support for specific detection of radioactive antigens. Nature 203: 648–649.
Berry, M.J., Davies, J., Smith, C.G. and Smith, I. 1991. Immobilization of Fv antibody fragments on porous silica and their utility in affinity chromatography. J. Chromatogr. 587: 161–169.
Livingstone, D.M. 1974. Immunoaffinity chromatography of proteins. Methods Enzymol. 34: 723–731.
Singh, P., Lewis, S.D. and Schafer, J.A. 1979. A support for affinity chromatography that covalently binds amino groups via a cleavable connector arm. Arch. Biochem. Biophys. 193: 284–293.
Boyle, M.D.P. 1990. Bacterial Immunoglobulin-Binding Proteins. Academic Press, New York.
Derrick, J.P. and Wigley, D.B. 1992. Crystal structure of a streptococcal protein G domain bound to an Fab fragment. Nature 359: 752–754.
Hofmann, K., Finn, F.M. and Kiso, Y. 1978. Avidin-biotin affinity columns. General methods for attaching biotin to peptides and proteins. J. Am. Chem. Soc. 100: 3585–3590.
Updyke, T.V. and Nicolson, G.L. 1986. Immunoaffinity isolation of membrane antigens with biotinylated monoclonal antibodies and streptavidin-agarose. Methods Enzymol. 121: 717–725.
Skerra, A. and Plückthun, A. 1988. Assembly of a functional immunoglobulin Fv fragment in Escherichia coli. Science 240: 1038–1041.
Skerra, A. 1993. Bacterial expression of immunoglobulin fragments. Curr. Opin. Immunol. 5: 256–262.
Schmidt, T.G.M. and Skerra, A. 1993. The random peptide libray-assisted engineering of a carboxy-terminal affinity peptide, useful for the detection and purification of a functional Ig Fv fragment. Protein Eng. 6: 109–122.
Munro, S. and Pelham, H. 1986. An Hsp70-like protein in the ER: Identity with the 78 kd glucose-regulated protein and immunoglobulin heavy chain, binding protein. Cell 46: 291–300.
Skerra, A., Pfitzinger, I. and Pluckthun, A. 1991. The functional expression of antibody Fv fragments in Escherichia coli: Improved vectors and a generally applicable purification technique. Bio/Technology 9: 273–278.
Sambrook, T., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual. 2nd ed. Cold Spring Harbour, New York.
Skerra, A. 1994. A general vector, pASK84, for cloning, bacterial production, and single-step purification of antibody Fab fragments. Gene 141: 79–84.
Orlandi, R., Gülssow, D.H., Jones, P.T. and Winter, G. 1989. Cloning immunoglobulin variable domains for expression by the polymerase chain reaction. Proc. Natl. Acad. Sci. USA 86: 3833–3837.
Kaluza, B., Betzl, G., Shao, H., Diamantstein, T. and Weidle, U.H. 1992. A general method for chimerization of monoclonal antibodies by inverse polymerase chain reaction which conserves authentic N-terminal sequences. Gene 122: 321–328.
Clackson, T., Hoogenboom, H.R., Griffiths, A.D. and Winter, G. 1991. Making antibody fragments using phage display libraries. Nature 352: 624–628.
Uematsu, Y. 1991. A novel and rapid cloning method for the T-Cell receptor variable region sequences. Immunogenetics 34: 174–178.
Kearney, T., Radbruch, A., Liesegang, B. and Rajewsky, K. 1979. A new mouse myeloma cell line that has lost immunoglobulin expression but permits the construction of antibody-secreting hybrid cell lines. J.Immunol. 123: 1548–1558.
Strohal, R., Kroemer, G., Wick, G. and Kofler, R. 1987. Complete variable region sequence of a nonfunctionally rearranged kappa light chain transcribed in the nonsecretor P3-X63-Ag8. 653 myeloma cell line. Nucl. Acids Res. 15: 2771.
Schmidt, T.G.M. and Skerra, A. 1994. One-step affinity purification of bacterially produced proteins by means of the Strep tag and immobilized recombinant core streptavidin. J. Chromatogr. 676: 337–345.
Gill, S.C. and Hippel, P.H. 1989. Calculation of protein extinction coefficients from amino acid sequence data. Anal. Biochem. 182: 319–326.
Berry, E.A. and Trumpower, B.L. 1985. Isolation of ubiquinol oxidase from Paracoccus denitrificans and resolution into cytochrome bc1 and cytochrome c-aa3 complexes. J. Biol. Chem. 260: 2458–2467.
Haltia, T. 1992. Cytochrome c oxidase: Biochemical, genetic and spectroscopic studies using enzyme from Paracoccus denitrificans. Commentationes Physico-Mathematicae et Chemico-Medicae 136 1–88.
Ludwig, B. and Schatz, G. 1980. A two-subunit cytochrome c oxidase (cytochrome aa3) from Paracoccus denitrificans. Proc. Natl. Acad. Sci. USA 77: 196–200.
Schägger, H. and Jagow, G. 1991. Blue native electrophoresis for isolation of membrane protein complexes in enzymatically active form. Anal. Biochem. 199: 223–231.
Hodgson, J. 1993. Expression Systems: A User's Guide. Bio/Technology 11: 887–893.
Hauri, H.P., Quaroni, A. and Isselbacher, K.J. 1980. Monoclonal antibodies to sucrase/isomaltase: Probes for the study of postnatal development and biogenesis of the intestinal microvillus membrane. Proc. Natl. Acad. Sci. USA 77: 6629–6633.
Saiki, R.K., Gelfand, D.H., Stoffel, S., Scharf, S.J., Higuchi, R., Horn, G.T., Mullis, K.B. and Ehrlich, H.A. 1988. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science 239: 487–491.
Sanger, F., Nicklen, S. and Coulson, A.R. 1977. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 74: 5463–5467.
Yanish-Peron, C., Viera, J. and Messing, J. 1985. Improved M13 phage cloning vectors and host strains: Nucleotide sequences of the M13mpl8 and pUC19 vectors. Gene 33: 103–199.
Gerhus, E., Steinrücke, P. and Ludwig, B. 1990. Paracoccus denitrificans cytochrome c1 gene replacement mutants. J. Bacteriol. 172: 2392–2400.
Fling, S.P. and Gregerson, D.S. 1986. Peptide and protein molecular weight determination by electrophoresis using a high-molarity buffer system without urea. Anal. Biochem. 155: 83–88.
Kabat, E.A., Wu, T.T., Perry, H.M., Gottesman, K.S. and Foeller, C. 1991. Sequences of Proteins of Immunological Interest, 5th ed. U.S. Dept. of Health and Human Services, U.S. Goverment Printing Office, Washington, DC.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kleymann, G., Ostermeier, C., Ludwig, B. et al. Engineered Fv Fragments as a Tool for the One-Step Purification of Integral Multisubunit Membrane Protein Complexes. Nat Biotechnol 13, 155–160 (1995). https://doi.org/10.1038/nbt0295-155
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0295-155