Abstract
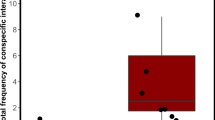
Owing to a lack of basic information on the biology of zebrafish (Danio rerio), lab managers must often base decisions regarding the care and use of this species on anecdotal information. In an effort to provide researchers with context-specific behavioral information, the authors evaluated shoaling and spawning behaviors in small groups of zebrafish. In each shoaling assay, a fish was given a choice to shoal with either a single fish or a group of three fish. Females preferred to shoal with a group of three individuals rather than with a single individual, regardless of the sex of the other fish. Males preferred groups of three males over single males but preferred single females to groups of three females. In spawning assays, zebrafish were placed in breeding tanks in one of three sex ratios (1 male:1 female; 3 males:1 female; 1 male:3 females). Reproductive efficiency did not differ among groups, but aggression (evaluated according to presence of shed scales) was more frequently observed in the male-dominated treatment group.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vascotto, S.G., Beckham, Y. & Kelly, G.M. The zebrafish's swim to fame as an experimental model in biology. Biochem. Cell Biol. 75, 479–485 (1997).
Lawrence, C. The husbandry of zebrafish (Danio rerio): a review. Aquaculture 269, 1–20 (2007).
Spence, R., Gerlach, G., Lawrence, C. & Smith, C. The behaviour and ecology of the zebrafish, Danio rerio . Biol. Rev. 83, 13–34 (2008).
Al-Imari, L. & Gerlai, R. Sight of conspecifics as a reward in associative learning in zebrafish (Danio rerio). Behav. Brain Res. 189, 216–219 (2008).
Gerlach, G., Hodgins-Davis, A., Avolio, C. & Schunter, C. Kin recognition in zebrafish: a 24-hour window for olfactory imprinting. Proc. R. Soc. Lond. B Biol Sci. 275, 2165–2170 (2008).
Marks, C., West, T.N., Bagatto, B. & Moore, F.B. Developmental environment alters conditional aggression in zebrafish. Copeia 4, 901–908 (2005).
Moretz, J.A., Martins, E.P. & Robison, B.D. The effects of early and adult social environment on zebrafish (Danio rerio) behavior. Environ. Biol. Fish. 80, 91–101 (2007).
Brown, C. & Laland, K.N. Social learning in fishes: a review. Fish Fish 4, 280–288 (2003).
Moretz, J.A., Martins, E.P. & Robison, B.D. Behavioral syndromes and the evolution of correlated behavior in zebrafish. Behav. Ecol. 18, 556–562 (2007).
Ruhl, N. & McRobert, S.P. The effect of sex and shoal size on shoaling behaviour in Danio rerio . J. Fish Biol. 67, 1318–1326 (2005).
Engeszer, R.E., Ryan, M.J. & Parichy, D.M. Learned social preference in zebrafish. Curr. Biol. 14, 881–884 (2004).
Sih, A. Predation risk and the evolutionary ecology of reproductive behavior. J. Fish Biol. 45, 111–130 (1994).
Institute for Laboratory Animal Research, National Research Council. Guide for the Care and Use of Laboratory Animals (National Academy Press, Washington, DC, 1996).
McRobert, S.P. & Bradner, J. The influence of body coloration on shoaling preferences in fish. Anim. Behav. 56, 611–615 (1999).
Westerfield, M. The Zebrafish Book: A Guide to the Laboratory Use of Zebrafish (Danio rerio) 3rd edn. (Univ. Oregon Press, Eugene, OR, 1995).
Spence, R. & Smith, C. Male territoriality mediates density and sex ratio effects on oviposition in the zebrafish, Danio rerio . Anim. Behav. 69, 1317–1323 (2005).
Spence, R., Ashton, R. & Smith, C. Oviposition decisions are mediated by spawning site quality in wild and domesticated zebrafish, Danio rerio . Behaviour 144, 953–996 (2007).
Larson, E.T., O'Malley, D.M. & Melloni, R.H. Aggression and vasotocin are associated with dominant-subordinate relationships in zebrafish. Behav. Brain Res. 167, 94–102 (2006).
Portz, D.E., Woodley, C.M. & Cech, J.J. Stress-associated impact of short-term holding on fishes. Rev. Fish Biol. Fisher. 16, 125–170 (2006).
Magurren, A.E. & Garcia, M. Sex differences in behaviour as an indirect consequence of mating system. J. Fish Biol. 57, 839–857 (2000).
Briffa, M. & Sneddon, L.U. Physiological constraints on contest behavior. Funct. Ecol. 21, 627–637 (2007).
Spence, R. & Smith, C. Mating preference of female zebrafish, Danio rerio, in relation to male dominance. Behav. Ecol. 17, 779–783 (2006).
Paull, G.C. et al. Variability in measures of reproductive success in laboratory-kept colonies of zebrafish and implications for studies addressing population-level effects of environmental chemicals. Aquat. Toxicol. 87, 115–126 (2008).
Acknowledgements
We thank the graduate students and faculty at both Saint Joseph's University and Ohio University for their helpful advice on this experiment. We also thank the devoted undergraduates who participated in this experiment at Ohio University: Jess Kauffman, Natalie Buchman and J. J. Soski.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Ruhl, N., McRobert, S. & Currie, W. Shoaling preferences and the effects of sex ratio on spawning and aggression in small laboratory populations of zebrafish (Danio rerio). Lab Anim 38, 264–269 (2009). https://doi.org/10.1038/laban0809-264
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/laban0809-264