Abstract
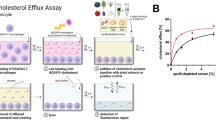
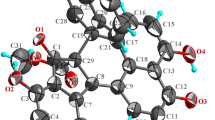
Cholesterol homeostasis is tightly controlled process that involves a variety of regulators including liver X receptors (LXR). Agonists of LXR are expected to increase cholesterol efflux, lower LDL, and raise HDL levels. Screening of a natural product library of microbial extracts using a LXR-scintillation proximity assay (SPA) binding assay and bioassay-guided fractionation of a number of fungal extracts led to the isolation of five ergostane and a cycloartane derivative. These compounds exhibited IC50 value ranging 0.5∼9 µM in the binding assay for α-receptor and a number of these showed in vitro agonist activity in the coactivator association assays but lacked the cell based LXR activation. The isolation and LXR activity of these compounds are described.
Similar content being viewed by others
Article PDF
References
Willy PJ, Umesono K, Ong ES, Evans RM, Heyman RA, Mangelsdorf DJ . LXR, a nuclear receptor that defines a distinct retinoid response pathway. Gene Dev 9: 1033–1045 ( 1995)
Miyata KS, McCaw SE, Patel HV, Rachubinski RA, Capone JP . The orphan nuclear hormone receptor LXRalpha interacts with the peroxisome proliferator-activated receptor and inhibits peroxisome proliferator signaling. J Biol Chem 271: 9189–9192 ( 1996)
Shinar DM, Endo N, Rutledge SJ, Vogel R, Rodan GA, Schmidt A . NER, a new member of the gene family encoding the human steroid hormone nuclear receptor. Gene 147: 273–276 ( 1994)
Repa JJ, Mangelsdorf DJ . The liver X receptor gene team: potential new players in atherosclerosis. Nat Med 8: 1243–1248 ( 2002)
Janowski BA, Willy PJ, Devi TR, Falck JR, Mangelsdorf DJ . An oxysterol signalling pathway mediated by the nuclear receptor LXRα. Nature 383: 728–731 ( 1996)
Lehmann JM, Kliewer SA, Moore LB, Smith-Oliver TA, Oliver BB, Su JL, Sundseth SS, Winegar DA, Blanchard DE, Spencer TA, Wilson TM . Activation of the nuclear receptor LXR by oxysterols defines a new hormone response pathway. J Biol Chem 272: 3137–3140 ( 1997)
Janowski BA, Grogan MJ, Jones SA, Wisely GB, Kliewer SA, Corey EJ, Mangelsdorf DJ . Structural requirements of ligands for the oxysterol liver X receptors LXRalpha and LXRbeta. Proc Natl Acad Sci USA 96: 266–271 ( 1999)
Fu X, Menke JG, Chen Y, Zhou G, MacNaul KL, Wright SD, Sparrow CP, Lund EG . 27-Hydroxycholesterol is an endogenous ligand for liver X receptor in cholesterol-loaded cells. J Biol Chem 276: 38378–38387 ( 2001)
Joseph SB, McKilligin E, Pei L, Watson MA, Collins AR, Laffitte BA, Chen M, Noh G, Goodman J, Hagger GN, Tran J, Tippin TK, Wang X, Lusis AJ, Hsueh WA, Law RE, Collins JL, Willson TM, Tontonoz P . Synthetic LXR ligand inhibits the development of atherosclerosis in mice. Proc Natl Acad Sci USA 99: 7604–7609 ( 2002)
Singh SB, Ondeyka JG, Liu W, Chen S, Chen TS, Li X, Bouffard A, Dropinski J, Jones AB, McCormick S, Hayes N, Wang J, Sharma N, MacNaul K, Hernandez M, Chao YS, Baffic J, Lam MH, Burton C, Sparrow CP, Menke JG . Discovery and development of dimeric podocarpic acid leads as potent agonists of liver X receptor with HDL raising activity in mice and hamsters. Bioorg Med Chem Lett 15: 2824–2828 ( 2005)
Herath K, Jayasuriya H, Ondeyka JG, Guan Z, Borris RP, Stijfhoorn E, Stevenson D, Wang J, Sharma N, MacNaul K, Menke JG, Ali A, Schulman MJ, Singh SB . Guttiferone I, a new prenylated benzophenone from Garcinia humilis as a liver X receptor ligand. J Nat Prod 68: 617–619 ( 2005)
Jayasuriya H, Herath KB, Ondeyka JG, Guan Z, Borris RP, Tiwari S, de Jong W, Chavez F, Moss J, Stevenson DW, Beck HT, Slattery M, Zamora N, Schulman M, Ali A, Sharma N, MacNaul K, Hayes N, Menke JG, Singh SB . Diterpenoid, steroid and triterpenoid agonists of liver X receptors from diversified terrestrial plants and marine sources. J Nat Prod 2005 ( in press)
Herath KB, Jayasuriya H, Guan Z, Schulman M, Ruby C, Sharma N, MacNaul K, Menke JG, Kodali S, Galgoci A, Wang J, Singh SB . Anthrabenzoxocinones from Streptomyces sp. as liver X receptor ligands and antibacterial agents. J Nat Prod 2005 ( in press)
Menke JG, Macnaul KL, Hayes NS, Baffic J, Chao Y-S, Elbrecht A, Kelly LJ, Lam MH, Schmidt A, Sahoo S, Wang J, Wright SD, Xin P, Zhou G, Moller DE, Sparrow CP . A novel liver X receptor agonist establishes species differences in the regulation of cholesterol 7α-hydroxylase (CYP7a). Endocrinology 143: 2548–2558 ( 2002)
Chobot V, Opletal L, Jahodar L, Patel AV, Dacke CG, Blunden G . Ergosta-4,6,8,22-tetraen-3-one from the edible fungus, Pleurotus ostreatus (oyster fungus). Phytochemistry 45: 1669–1671 ( 1997)
Raederstorff D, Rohmer M . The action of the systemic fungicides tridemorph and fenpropimorph on sterol biosynthesis by the soil amoeba Acanthamoeba ployphaga. Eur J Biochem 164: 421–426 ( 1987)
Raederstorff D, Rohmer M . Sterol biosynthesis via cycloartenol and other biochemical features related to photosynthetic phyla in the amoebe Naegleria lovaniensis and Naegleria gruberi. Eur J Biochem 164: 427–434 ( 1987)
Akihisa T, Kokke WCMC, Kimura Y, Tamura T . Isokarounidiol [D:C-Friedooleana-6,8-diene-3alpha,29-diol]: the first naturally occuring triterpene with a delta6,8-conjugated diene system. Iodine-mediated dehydrogenation and isomerization of its diacetate. J Org Chem 58: 1959–1962 ( 1993)
Bisseret P, Adam H, Rohmer M . Structure elucidation of ring B aromatic sterols of the soid amoeba Acanthamoeba polyphaga. J Chem Soc Chem Commun 693–695 ( 1987)
Ponce MA, Ramirez JA, Galagovsky LR, Gros EG, Erra-Balsells R . A new look into the reaction between ergosterol and singlet oxygen in vitro. Photochem Photobiol Sci 1: 749–756 ( 2002)
Akihisa T, Shimizu N, Tamura T, Matsumoto T . (24S)-14alpha,24-dimethyl-9beta,19-cyclo-5alpha-cholest-25-en-3beta-ol: A new sterol and other sterols in Musa sapientum. Lipids 21: 494–497 ( 1986)
Akihisa T, Kimura Y, Kokke WCMC, Takase S, Yasukawa K, Jin-Nai A, Tamura T . 4-Epicycloeucalenone and 4-epicyclomusalenone: Two 3-oxo-28-norcycloartanes from fruit peel of Musa sapientum L. Chem Pharm Bull 45: 744–746 ( 1997)
Spencer TA, Li D, Russel JS, Collins JL, Bledsoe RK, Consler TG, Moore LB, Galardi CM, McKee DD, Moore JT, Watson MA, Parks DJ, Lambert MH, Willson TM . Pharmacophore analysis of the nuclear oxysterol receptor LXRa. J Med Chem 44: 886–897 ( 2001)
Peláez F, Collado J, Arenal F, Basilio A, Cabello A, Díez Matas MT, García JB, González Del Val A, González V, Gorrochategui J, Hernández P, Martín I, Platas G, Vicente F . Mycol Res 102: 755–761 ( 1998)
Sparrow CP, Baffic J, Lam M-H, Lund EG, Adams AD, Fu X, Hayes N, Jones AB, Macnaul KL, Ondeyka J, Singh S, Wang J, Zhou G, Moller DE, Wright SD, Menke JG . A potent synthetic LXR agonist is more effective than cholesterol loading at inducing ABCA1 mRNA and stimulating cholesterol efflux. J Biol Chem 277: 10021–10027 ( 2002)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ondeyka, J., Jayasuriya, H., Herath, K. et al. Steroidal and Triterpenoidal Fungal Metabolites as Ligands of Liver X Receptors. J Antibiot 58, 559–565 (2005). https://doi.org/10.1038/ja.2005.76
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2005.76
Keywords
This article is cited by
-
Secondary Metabolites of Fungus Penicillium thomii Associated with Eelgrass Zostera marina
Chemistry of Natural Compounds (2018)
-
Pleiotropic Effects of a Schweinfurthin on Isoprenoid Homeostasis
Lipids (2011)