Abstract
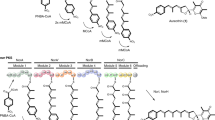
Epothilones, potent cytotoxic agents and potential anticancer drugs, are complex polyketides produced by a modular polyketide synthase (PKS). The epothilone PKS genes were introduced and expressed in Myxococcus xanthus and engineered to generate novel unnatural natural products which can be used as new scaffolds for chemical modification. Inactivation of the KR domain in module 6 of the epo PKS resulted in accumulation of 9-oxoepothilone D and its isomer 8-epi-9-oxoepothilone D as the major products. Modification of the KR domain in module 4 resulted in the production of the expected compound 12,13-dihydro-13-oxoepothilone C in trace amounts, and the unexpected compound 11,12-dehydro-12,13-dihydro-13-oxoepothilone D as the major product. The other expected compound, 12,13-dihydro-13-oxoepothilone D, was not detected. The unexpected 13-oxo derivative produced indicates that the ER domain of module 5 has substrate-specificity requirements and suggests a second enzymatic role for the domain.
Similar content being viewed by others
Article PDF
References
Gerth K, Bedorf N, Höfle G, Irschik H, Reichenbach H . Antifungal and cytotoxic compounds from Sorangium cellulosum (Myxobacteria)—Production, physico-chemical and biological properties. J Antibiot 49: 560–563 ( 1996)
Tang L, Shah S, Chung L, Carney JR, Katz L, Khosla C, Julien B . Cloning and heterologous expression of the epothilone gene cluster. Science 287: 640–642 ( 2000)
Hardt I, Steinmetz H, Gerth K, Sasse F, Reichenbach H, Höfle G . New natural epothilones from Sorangium cellulosum, strains So ce90/B2 and So ce90/D13: isolation, structure elucidation, and SAR studies. J Nat Prod 64: 847–856, 2001
Höfle G, Bedorf N, Steinmetz H, Schumberg D, Gerth K, Reichenbach H . Epothilones A and B-novel 16-membered macrolides with cytotoxic activity: isolation, crystal structure, and conformation in solution. Angew Chem Int Ed Engl 35: 1567–1569 ( 1996)
Bollag D, McQueney P, Zhu J, Hensens O, Koupal L, Liesch J, Goetz M, Lazarides E, Woods C . Epothilones, a new class of microtubule-stabilizing agents with a taxol-like mechanism of action. Cancer Res 55: 2325–2333 ( 1995)
Harris C, Danishefsky S . Complex target-oriented synthesis in the drug discovery process: a case history in the dEpoB series. J Org Chem 64: 8434–8456 ( 1999)
Julien B, Shah S . Heterologous expression of epothilone biosynthetic genes in Myxococcus xanthus. Antimicrob Agents Chemother 46: 2772–2778 ( 2002)
Lau J, Frykman S, Regentin R, Ou S, Tsuruta H, Licari P . Biotechnol Bioeng 78: 280–288 ( 2002)
McDaniel R, Thamchaipenet A, Gustafsson C, Fu H, Betlach M, Ashley G . Multiple genetic modifications of the erythromycin gene cluster to produce a library of novel “unnatural” natural products. Proc Natl Acad Sci USA 96: 1846–1851 ( 1999)
Weissman KJ, Staunton J . Polyketide synthases: analysis and use in synthesis. In Enzyme Technologies for Pharmaceutical and Biotechnological Applications. Ed., Kirst HA et al., pp. 427–470, Marcel Dekker, Inc., New York, 2001
Arslanian RL, Tang L, Blough S, Ma W, Qiu R-G, Katz L, Carney JR . A new cytotoxic epothilone from modified polyketide synthases heterologously expressed in Myxococcus xanthus. J Nat Prod 65: 1061–1064 ( 2002)
Tang L, Ward S, Chung L, Carney JR, Li Y, Reid R, Katz L . Elucidating the mechanism of cis double bond formation in epothilone biosynthesis. J Am Chem Soc 126: 47–48 ( 2004)
Reid R, Piagentini M, Rodriguez E, Ashley G, Viswanathan N, Carney JR, Santi DV, Hutchinson CR, McDaniel R . A model of structure and catalysis for ketoreductase domains in modular polyketide synthases. Biochemistry 42: 72–79 ( 2003)
Starks C, Zhou Y, Liu F, Licari P . Isolation and characterization of new epothilone analogues from recombinant Myxococcus xanthus fermentations. J Nat Prod 66: 1313–1317 ( 2003)
Kashefi K, Hartzell P . Genetic suppression and phenotypic masking of a Myxococcus xanthus frzF− defect. Mol Microbiol 15: 483–494 ( 1995)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tang, L., Chung, L., Carney, J. et al. Generation of New Epothilones by Genetic Engineering of a Polyketide Synthase in Myxococcus xanthus. J Antibiot 58, 178–184 (2005). https://doi.org/10.1038/ja.2005.20
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2005.20
Keywords
This article is cited by
-
Engineering the acyltransferase domain of epothilone polyketide synthase to alter the substrate specificity
Microbial Cell Factories (2021)
-
Natural product discovery: past, present, and future
Journal of Industrial Microbiology and Biotechnology (2016)
-
Allopatric integrations selectively change host transcriptomes, leading to varied expression efficiencies of exotic genes in Myxococcus xanthus
Microbial Cell Factories (2015)
-
Microbial natural products: molecular blueprints for antitumor drugs
Journal of Industrial Microbiology and Biotechnology (2013)
-
Nature: a vital source of leads for anticancer drug development
Phytochemistry Reviews (2009)