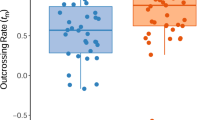
Abstract
Early inbreeding depression, i.e. embryonic recessive lethals, eliminates a large proportion of selfed progeny during embryo development. A model of early inbreeding depression suggested that in most conifers the variation between genotypes in the number of lethals rather than the variation in the actual rate of self-fertilization accounts for the variation between selfing rates at the seed stage. Polyembryony, the formation of several embryos per ovule in conifers, diminished the fitness cost of embryonic lethals and allowed embryo competition. We studied variation in the outcrossing rate at the seed stage in an experimental population of Scots pine. Despite extensive variation, pollen production of the trees, which is expected to predict the probability of self-fertilization, did not account for the low selfing rate variation at the seed stage. The genotypes having lowest numbers of embryonic lethals had the highest selfing rates at the seed stage. Early inbreeding depression maintains a very low selfing rate at the seed stage and masks the correlation between the rate of self-fertilization and the selfing rate at the seed stage. This is typical of most conifers and can also be common among perennial angiosperm species.
Similar content being viewed by others
Article PDF
References
Barrett, S H, and Eckert, C G. 1990. Variation and evolution of mating systems in seed plants. In: Kawano, S. (ed.) Biological Approaches and Evolutionary Trends in Plants, Academic Press, London, pp. 230–254.
Bishir, J, and Namkoong, G. 1987. Unsound seeds in conifers: estimation of lethal alleles and of magnitudes of effects associated with the maternal parent. Silvae Genetica, 36, 180–185.
Burbidge, A H, and James, S H. 1991. Postyzygotic Seed abortion in the genetic system of Stylidium (Angiospermae: Stylidaceae). J Hered, 82, 319–328.
Charlesworth, D. 1989. Why do plants produce so many more ovules than seeds? Nature, 338, 21–22.
Charlesworth, D, and Charlesworth, B. 1987. Inbreeding depression and its evolutionary consequences. Ann Rev Ecol Syst, 18, 237–268.
Charlesworth, D, Morgan, M T, and Charlesworth, B. 1990. Inbreeding depression, genetic load and the evolution of outcrossing rates in a multilocus system with no linkage. Evolution, 44, 1469–1489.
Chung, M-S. 1981. Flowering characteristics of Pinus sylvestris L. with special emphasis on the reproductive adaptation to local temperature factor. Acta Forestalia Fennica, 169, 1–68.
Denti, D, and Schoen, D J. 1988. Self-fertilization rates in white spruce: effect of pollen and seed production. J Hered, 79, 284–288.
El-Kassaby, Y A, and Ritland, K. 1992. Frequency-dependent male reproductive success in a polycross of Douglas fir. Theor Appl Genet, 83, 752–758.
Ellstrand, N C, Torres, A M, and Levin, D A. 1978. Density and the rate of apparent outcrossing in Helianthus (Asteraceae). Syst Bot, 3, 403–407.
Epperson, B K, and Clegg, M T. 1987. Frequency-dependent variation for outcrossing rate among flower-color morphs of Ipomoea purpurea. Evolution, 41, 1302–1311.
Erickson, V J, and Adams, W T. 1990. Mating system variation among individual ramets in a Douglas-fir seed orchard. Can J For Res, 20, 1672–1675.
Farris, M A, and Mitton, J B. 1984. Population density, outcrossing rate and heterozygote superiority in ponderosa pine. Evolution, 38, 1151–1154.
Furnier, G R, and Adams, W T. 1986. Mating system in natural populations of Jeffrey pine. Am J Bot, 73, 1002–1008.
Harju, A, and Muona, O. 1989. Background pollination in Pinus sylvestris seed orchards. Scand J For Res, 4, 513–520.
Holtsford, T P, and Ellstrand, N C. 1992. Genetic and environmental variation in floral traits affecting outcrossing rate in Clarkia tembloriensis (Onagraceae). Evolution, 46, 216–225.
Kärkkäinen, K, and Muona, O. 1991. Reproduction of Pinus sylvestris at the northern timber line. In: Fineschi, S., Malvolti, M. E., Cannata, F. and Hattemer, H. H. (eds), Biochemical Markers in the Population Genetics of Forest Trees, SPB Academic Publishers, The Hague, p. 247.
Knowles, P, Furnier, G R, Aleksiuk, M A, and Perry, D J. 1987. Significant levels of self-fertilization in natural populations of tamarack. Can J Bot, 65, 1087–1091.
Koski, V. 1971. Embryonic lethals of Picea abies and Pinus sylvestris. Commun Inst For Fenn, 75, 3, 1–30.
Koski, V. 1973. On self-pollination, genetic load and subsequent inbreeding in some conifers. Commun Inst For Fenn, 78, 10, 1–42.
Koski, V, and Muona, O. 1986. Probability of inbreeding in relation to clonal differences in male flowering and embryonic lethals. Proceedings IUFRO Conference on Breeding Theory, Progeny Testing and Seed Orchards, Williamsburgh, Virginia, 13–17 October, 1986, pp. 391–400.
Krebs, S L, and Hancock, J F. 1991. Embryonic genetic load in the highbush blueberry, Vaccinium corymbosum (Ericaceae). Am J Bot, 78, 1427–1437.
Kuittinen, H, and Savolainen, O. 1992. Picea omorika is a fully self-fertile but outcrossing conifer. Heredity, 68, 183–187.
Lande, R, and Schemske, D W. 1985. The evolution of self-fertilization and inbreeding depression in plants. I. Genetic models. Evolution, 39, 24–40.
Lindgren, D. 1975. The relationship between self-fertilization, empty seeds and seeds originating from selfing as a consequence of polyembryony. Studia Forestalia Suecica, 126, 1–24.
Mitchell-Olds, T, and Guries, R P. 1986. Genetic load and heterozygosity in the Pinaceae. Can J Genet Cytol, 28, 942–946.
Moran, G F, and Griffin, A R. 1985. Non-random contribution of pollen in polycrosses of Pinus radiata D. Don. Silvae Genet, 34, 4–5.
Muona, O. 1989. Population genetics in tree improvement. In: Brown, A. H. D., Clegg, M. T. Kahler, A. L. and Weir, B. S. (eds), Plant Population Genetics, Breeding and Genetic Resources, Sinauer Sunderland, MA, pp. 282–298.
Muona, O, and Harju, A. 1989. Effective population sizes, genetic variability and mating system in natural stands and seed orchards of Pinus sylvestris. Silvae Genetica, 38, 221–228.
Muona, O, Harju, A, and Kärkkäinen, K. 1988. Genetic comparison of natural and nursery grown seedlings of Pinus sylvestris. Scand J For Res, 4, 37–46.
Nakamura, R R, and Wheeler, N C. 1992. Pollen competition and paternal success in Douglas-fir. Evolution, 46, 846–851.
Namkoong, G, and Bishir, J. 1987. The frequency of lethal alleles in forest tree populations. Evolution, 41, 1123–1127.
Neale, D B, and Adams, W T. 1985. The mating system in natural and shelterwood stands of Douglas-fir. Theor Appl Genet, 71, 201–207.
Omi, S K, and Adams, W T. 1986. Variation in seed set and proportions of outcrossed progeny with clones, crown position and top pruning in a Douglas-fir seed orchard. Can J For Res, 16, 502–507.
Ritland, K, and El-Kassaby, Y A. 1985. The nature of inbreeding in a seed orchard of Douglas fir as shown by an efficient multilocus model. Theor Appl Genet, 71, 375–384.
Sarvas, R. 1962. Investigations on the flowering and seed crop of Pinus sylvestris. Commun Inst For Fenn, 53, 4, 1–198.
Savolainen, O, Kärkkäinen, K, and Kuittinen, H. 1992. Estimating numbers of embryonic lethals in conifers. Heredity, 69, 308–314.
Schoen, D J, and Cheliak, W M. 1987. Genetics of the poly-cross. 2. Male fertility variation in Norway spruce, Picea abies (L.). Karst Theor Appl Genet, 74, 554–559.
Sedgley, M, and Griffin, A R. 1989. Sexual Reproduction of Tree Crops, Academic Press, London.
Shea, K L. 1987. Effects of population structure and cone production on outcrossing rates in Engelmann spruce and subalpine fir. Evolution, 41, 124–136.
Snow, A A, and Spira, T P. 1991. Differential pollen tube growth rates and non-random fertilization in Hibiscus moscheutos (Malvaceae). Am J Bot, 78, 1419–1426.
Sorensen, F. 1969. Embryonic genetic load in coastal Douglas fir, Pseudotsuga menziesii var. menziesii. Am Nat, 103, 389–398.
Sorensen, F C. 1982. The roles of polyembryony and embryo viability in the genetic system of conifers. Evolution, 36, 725–733.
Wiens, D, Calvin, C L, Wilson, C A, Davern, C I, Frank, D, and Seavey, S R. 1987. Reproductive success, spontaneous embryo abortion, and genetic load in flowering plants. Oecologia, 71, 501–509.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kärkkäinen, K., Savolainen, O. The degree of early inbreeding depression determines the selfing rate at the seed stage: model and results from Pinus sylvestris (Scots pine). Heredity 71, 160–166 (1993). https://doi.org/10.1038/hdy.1993.120
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1993.120
Keywords
This article is cited by
-
How does population outcrossing rate influence seed quality? A case study from a seed tree stand of Pinus massoniana
New Forests (2023)
-
Genetic diversity and fine-scale spatial genetic structure of unmanaged old-growth versus managed second-growth populations of Scots pine (Pinus sylvestris L.) in Lithuania
European Journal of Forest Research (2023)
-
Conservation genomics of an Australian cycad Cycas calcicola, and the Absence of Key Genotypes in Botanic Gardens
Conservation Genetics (2022)
-
Life stage-specific inbreeding depression in long-lived Pinaceae species depends on population connectivity
Scientific Reports (2021)
-
Inbreeding depression from selfing and mating between relatives in the Neotropical tree Cariniana legalis Mart. Kuntze
Conservation Genetics (2017)