Abstract
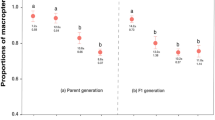
Genetic variability and phenotypic plasticity have been suggested as mutually exclusive ways by which adaptation to environmental heterogeneity can be accomplished. The genetic and environmental components of variation in the darkness of brown pupae of the swallowtail butterfly Papilio polyxenes were investigated. Variation in darkness was found to be both genetic and environmental, with the darkness of the pupation site having a significant influence on the darkness of pupae, thus allowing for the production of cryptic brown pupae in nature. These results suggest that the evolution of phenotypic plasticity in a trait need not be to the exclusion of the maintenance of genetic variation in that trait. The results of field experiments show that cryptic pupae have a higher probability of escaping predation relative to noncryptic pupae.
Similar content being viewed by others
Article PDF
References
Baker, H G. 1965. Characteristics and modes of origin of weeds. In Baker, H. G. and Stebbins, G. L. (eds.) The Genetics of Colonizing Species, Academic Press, N.Y. pp. 147–172.
Bradshaw, A D. 1965. Evolutionary significance of phenotypic plasticity in plants. Adv Genet, 13, 115–155.
Clarke, C A, and Sheppard, P M. 1962. Offspring from double matings in swallowtail butterflies. The Entomologist, August.
Clarke, C A, and Sheppard, P M. 1972. Genetic and environmental factors influencing pupal colour in the swallowtail butterflies Battus philenor (L.) and Papilio polytes L. J Entomol, (A) 46, 123–133.
Falconer, D S. 1952. The problem of environment and selection. Am Natur, 86, 293–298.
Falconer, D S. 1981. Introduction to Quantitative Genetics 2nd ed. Longman, New York.
Gause, G F. 1947. Problems of evolution. Trans Conn Acad Sci, 37, 17–68.
Gottlieb, L D. 1977. Phenotypic variation in Stephanomeria exigua ssp. coronaria (Compositae) and its recent derivative species “Malheurensis”. Amer J Bot, 64, 873–880.
Hazel, W N. 1977. The genetic basis of pupal colour dimorphism and its maintenance by natural selection in Papilio polyxenes (Papilionidae: Lepidoptera). Heredity, 38, 227–236.
Hazel, W N, and West, D A. 1979. Environmental control of pupal colour in swallowtail butterflies (Lepidoptera: Papilioninae): Battus philenor (L.) and Papilio polyxenes Fabr. Ecological Entomology, 4, 393–400.
Hazel, W N, and West, D A. 1982. Pupal colour dimorphism in swallowtail butterflies as a threshold trait: selection in Eurytides marcellus (Cramer). Heredity, 49, 295–301.
Hazel, W N, and West, D A. 1983. The effect of larval photoperiod on pupal colour and diapause in swallowtail butterflies. Ecological Entomology, 8, 37–42.
Jain, S K. 1978. Inheritance of phenotypic plasticity in soft chess, Bromus mollis L. (Gramineae). Experimentia, 34, 835–836.
Jain, S K. 1979. Adaptive strategies: polymorphism, plasticity, and homeostasis. In Solbrig, O. T., Jain, S. K., Johnson. G. B. and Raven, P. H. (eds.) Topics in Plant Population Biology, Columbia University Press, New York, pp. 160–187.
Lande, R. 1976. The maintenance of genetic variability by mutation in a polygenic character with linked loci. Genet Res, 26, 221–235.
Lande, R. 1980. The genetic covariance between characters maintained by pleiotropic mutations. Genetics, 54, 203–215.
Levis, R. 1963. Theory of fitness in a heterogeneous environment. II. Developmental flexibility and niche selection. Amer Natur, 97, 75–90.
Marshall, D M, and Jain, S K. 1968. Phenotypic plasticity of Avenafatua and A. barbata. Am Natur, 102, 457–467.
Scharloo, W. 1964. The effect of disruptive and stabilizing selection on the expression of cubitus interruptus in Drosophila. Genetics, 50, 553–562.
Scheiner, S M, and Goodnight, C J. 1984. The comparison of phenotypic plasticity and genetic variation in populations of the grass Danthonia spicata. Evolution, 38, 845–855.
Schlichting, C D, and Levin, D A. 1984. Phenotypic plasticity of annual Phlox: Tests of some hypothesis. Amer J Bot, 71, 252–260.
Simpson, G G, Roe, A, and Lewontin, R C. 1960. Quantitative Zoology, Harcourt, Brace and Co., Inc. New York.
Sims, S R. 1983. The genetic and environmental basis of pupal colour dimorphism in Papilio zelicaon (Lepidoptera: Papilionidae). Heredity, 50, 159–168.
Smith, A G. 1978. Environmental factors influencing pupal colour in Lepidoptera. I. Experiments with Papilio polytes, Papilio demoleus, and Papilio polyxenes. Proceedings of the Royal Society of London, B, 200, 295–329.
Thoday, J M. 1953. Components of fitness. Symp Soc Exptl Biol, 7, 96–113.
Turelli, M. 1984. Heritable genetic variation via mutation-selection balance: Lerch's zeta meets the abdominal bristle. Theoret Popul Biol, 25, 138–193.
Via, S, and Lande, R. 1985. Genotype-environment interaction and the evolution of phenotypic plasticity. Evolution, 39, 505–522.
West, D A, Snellings, W M, and Herbek, T A. 1972. Pupal colour dimorphism and its environmental control in Papilio polyxenes asterius Stoll. (Lepidoptera: Papilionidae). J N Y Entomol Soc, 80, 205–211.
West, D A, and Hazel, W N. 1979. Natural pupation sites of swallowtail butterflies (Lepidoptera: Papilioninae): Papilio polyxenes Fabr., P. glaucus L. and Battus philenor (L). Ecological Entomology, 4, 387–392.
West, D A, and Hazel, W N. 1985. Pupal colour dimorphism in swallowtail butterflies: timing of the sensitive period and environmental control. Physiological Entomology, 10, 113–119.
Wiklund, C. 1972. Pupal coloration in Papilio machaon in response to wavelength of light. Naturwissenschaften, 59, 219.
Wiklund, C. 1975. Pupal colour polymorphism and the survival in the field of cryptic and non-cryptic pupae in Papilio machaon L. J Roy Ent Soc Lond, 127, 73–84.
Wilken, D H. 1977. Local differentiation for phenotypic plasticity in the wild annual Collomia linearis (Polemoniaceae). Syst Bot, 2, 99–108.
Wright, S. 1969. Evolution and the Genetics of Natural Populations, Vol 2 The Theory of Gene Frequencies Univ. of Chicago Press, Chicago, IL.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hazel, W., Brandt, R. & Grantham, T. Genetic variability and phenotypic plasticity in pupal colour and its adaptive significance in the swallowtail butterfly Papilio polyxenes. Heredity 59, 449–455 (1987). https://doi.org/10.1038/hdy.1987.155
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1987.155
This article is cited by
-
Predator-induced plasticity does not alter the pathway from evolution to ecology among locally adapted populations of Daphnia
Evolutionary Ecology (2017)
-
Colony size, but not density, affects survival and mating success of alternative male reproductive tactics in a polyphenic mite, Rhizoglyphus echinopus
Behavioral Ecology and Sociobiology (2014)
-
Modelling phenotypic plasticity. I. Linear and higher-order effects of dominance, drift, environmental frequency and selection on a one-locus, two-allele model
Journal of Genetics (1992)