Abstract
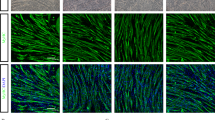
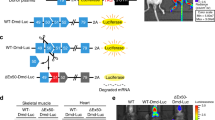
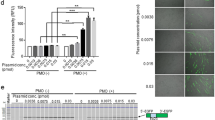
Transplantation of muscle precursor cells (MPCs) is a promising approach for the treatment of muscular dystrophies. However, preclinical and clinical results have shown that the technology is not yet efficient enough for most therapeutic applications. Among the problems that remain unsolved are low cellular survival, poor proliferation and lack of migration of the transplanted cells. One major technical hurdle for the optimization of transplantation protocols is how to follow precisely the fate of the cells after transplantation. In this study, we examined the use of a secreted form of the mouse alkaline phosphatase (mSeAP) enzyme as the reporter system transduced into MPCs using a retroviral vector. We show that circulating mSeAP could be detected in the serum of the transplanted mice at different time points after MPC transplantation. We also found that the level of circulating mSeAP is highly correlated with the number of transplanted cells and that mSeAP is an excellent histological marker. Further, studying the levels of circulating mSeAP compared with the number of muscle fibers positive to mSeAP and to dystrophin, enabled detailed analyses of bottleneck steps for successful transplantation. Taken together, our results show that mSeAP is an excellent quantitative ‘real-time’ reporter gene for cell therapy preclinical studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Charge SB, Rudnicki MA . Cellular and molecular regulation of muscle regeneration. Physiol Rev 2004; 84: 209–238.
Skuk D, Tremblay JP . Myoblast transplantation: the current status of a potential therapeutic tool for myopathies. J Muscle Res Cell Motil 2003; 24: 285–300.
Smythe GM, Hodgetts SI, Grounds MD . Problems and solutions in myoblast transfer therapy. J Cell Mol Med 2001; 5: 33–47.
Partridge TA, Grounds M, Sloper JC . Evidence of fusion between host and donor myoblasts in skeletal muscle grafts. Nature 1978; 273: 306–308.
Partridge TA, Morgan JE, Coulton GR, Hoffman EP, Kunkel LM . Conversion of mdx myofibres from dystrophin-negative to -positive by injection of normal myoblasts. Nature 1989; 337: 176–179.
Gussoni E, Soneoka Y, Strickland CD, Buzney EA, Khan MK, Flint AF et al. Dystrophin expression in the mdx mouse restored by stem cell transplantation. Nature 1999; 401: 390–394.
Cullen BR, Malim MH . Secreted placental alkaline phosphatase as a eukaryotic reporter gene. Methods Enzymol 1992; 216: 362–368.
Bettan M, Darteil R, Scherman D . Secreted human placental alkaline phosphatase as a reporter gene for in vivo gene transfer. Anal Biochem 1999; 271: 187–189.
Wang M, Orsini C, Casanova D, Millan JL, Mahfoudi A, Thuillier V . MUSEAP, a novel reporter gene for the study of long-term gene expression in immunocompetent mice. Gene 2001; 279: 99–108.
Bartoli M, Poupiot J, Goyenvalle A, Perez N, Garcia L, Danos O et al. Noninvasive monitoring of therapeutic gene transfer in animal models of muscular dystrophies. Gene Therapy 2006; 13: 20–28.
Bosio E, Lee-Pullen TF, Fragall CT, Beilharz MW, Bennett AL, Grounds MD et al. A comparison between real-time quantitative PCR and DNA hybridization for quantitation of male DNA following myoblast transplantation. Cell Transplant 2004; 13: 817–821.
Boulanger A, Asselin I, Roy R, Tremblay JP . Role of non-major histocompatibility complex antigens in the rejection of transplanted myoblasts. Transplantation 1997; 63: 893–899.
Fukada S, Higuchi S, Segawa M, Koda K, Yamamoto Y, Tsujikawa K et al. Purification and cell-surface marker characterization of quiescent satellite cells from murine skeletal muscle by a novel monoclonal antibody. Exp Cell Res 2004; 296: 245–255.
Israeli D, Ziaei S, Gjata B, Benchaouir R, Rameau P, Marais T et al. Expression of mdr1 is required for efficient long term regeneration of dystrophic muscle. Exp Cell Res 2007; 313: 2438–2450.
Benchaouir R, Rameau P, Decraene C, Dreyfus P, Israeli D, Pietu G et al. Evidence for a resident subset of cells with SP phenotype in the C2C12 myogenic line: a tool to explore muscle stem cell biology. Exp Cell Res 2004; 294: 254–268.
Fan Y, Maley M, Beilharz M, Grounds M . Rapid death of injected myoblasts in myoblast transfer therapy. Muscle Nerve 1996; 19: 853–860.
Lu QL, Morris GE, Wilton SD, Ly T, Artem′yeva OV, Strong P et al. Massive idiosyncratic exon skipping corrects the nonsense mutation in dystrophic mouse muscle and produces functional revertant fibers by clonal expansion. J Cell Biol 2000; 148: 985–996.
Acknowledgements
The authors wish to acknowledge the excellent technical help of Simon Jimenez, Christophe Georger and Bernard Gjata from the histology service of Genethon, and to Christel Rivière from the AAV team of Genethon for the gift of recombinant mSeAP.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gerard, X., Vignaud, L., Charles, S. et al. Real-time monitoring of cell transplantation in mouse dystrophic muscles by a secreted alkaline phosphatase reporter gene. Gene Ther 16, 815–819 (2009). https://doi.org/10.1038/gt.2009.28
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2009.28