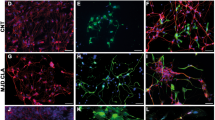
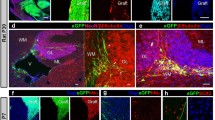
Abstract
Experiments in rodents have shown a potential role for glial cell transplantation as a means of influencing repair in the central nervous system of man. A crucial step in developing human therapy is to establish whether knowledge gained from studies in rodents is applicable to larger mammalian species. In order to explore this issue we examined the ability of cat glial cell cultures to remyel-inate areas of ethidium-bromide-induced demyelination in the spinal cord of immunosuppressed rats and cats. Transplantation of density-gradient-isolated glial cells obtained from the forebrain of 7-day-old kittens resulted in enhanced oligodendrocyte remyelination in the rat but failed to enhance oligodendrocyte remyelination in the cat. The feasibility of enhancing oligodendrocyte remyelination in the cat lesion was demonstrated by transplanting a rat culture containing a high proportion of cells of the oligodendrocyte lineage. Tissue culture of the density-gradient-isolated cell preparations suggested that the failure of the kitten glial preparation to enhance oligodendrocyte remyelination in the cat was most probably due to its poor oligodendrocyte-generating capacity. However, our lack of understanding of the biology of feline glial cells precludes a full understanding of these experiments.
Similar content being viewed by others
Article PDF
References
Franklin RJM . Reconstructing myelin-deficient environments in the CNS by glial cell transplantation. Semin Neu- rosci 1993; 5: 443–51.
Blakemore WF . Transplanted cultured type-1 astrocytes can be used to reconstruct the glia limitans of the CNS: the structure which prevents Schwann cells from myelinating CNS axons. Neuropathol Appl Neurobiol 1992; 18: 460–6.
Franklin RJM, Crang AJ, Blakemore WF . The role of astrocytes in the remyelination of glia-free areas of demyelination. In: Seil FJ, editor. Advances in neurology, vol 59. New York: Raven Press, 1993: 125–33.
Groves AK, Barnett SC, Franklin RJM, Crang AJ, Mayer M, Blakemore WF, Noble M . Repair of demyelinated lesions by transplantation of purified 0-2A progenitor cells. Nature 1993; 362: 453–5.
Warrington AE, Barbarese E, Pfeiffer SE . Differential myelinogenic capacity of specific stages of the oligodendrocyte lineage upon transplantation into hypomyelinating hosts. J Neurosci Res 1993; 34: 1–13.
Sommer I, Schachner M . Monoclonal antibodies (O1-O4) to oligodendrocyte surfaces: an immunocytochemical study in the central nervous system. Dev Biol 1981; 83: 311–27.
Gard AL, Pfeiffer SE . Two proliferative stages of the oligodendrocyte lineage (A2B5 + 04 - ve and 04 + GalC - ve) under different mitogenic control. Neuron 1990; 5: 615–25.
Raff MC, Miller RH, Noble M . A glial progenitor cell that develops in vitro into an astrocyte or an oligodendrocyte depending on the culture medium. Nature 1983; 303: 390–6.
Blakemore WF, Crang AJ . Transplantation of glial cells into areas of demyelination in the adult rat spinal cord. In: Dunnets and Bjorklund A, editor. Neural transplantation: a practical approach. Oxford: Oxford University Press, 1992: 105–22.
Crang AJ, Franklin RJM, Blakemore WF, Noble M, Barnett SC, Groves A, et al. The differentiation of glial cell progenitor populations following transplantation into non-repairing CNS lesions in adult animals. J Neuroimmunol 1992; 40: 243–54.
Blakemore WF . Ethidium bromide induced demyelination in the spinal cord of the cat. Neuropathol Appl Neurobiol 1982; 8: 365–75.
Archer DR, Cuddon PA, Duncan ID . Myelination by cryo- preserved xenografts and allografts in the myelin deficient rat. Exp Neurol 1994; 125: 268–77.
Crang AJ, Blakemore WF . Remyelination of demyelinated rat axons by transplanted mouse oligodendrocytes. Glia 1991; 4: 305–13.
Gumpel M, Lachapelle F, Gransmüller A, Baulac M, Baron-Van Evercooren A, Baumann N . Transplantation of human embryonic oligodendrocytes into shiverer brain. Ann NY Acad Sci 1987; 495: 71–85.
Lubetzki C, Gunsmüller A, Lachapelle F, Lombrail P, Gum-pel M . Myelination of oligodendrocytes isolated from 4-6 week-old rat central nervous system and transplantation into newborn shiverer brain. J Neurol Sci 1988; 88: 161–75.
Franklin RJM, Crang AJ, Blakemore WF . The reconstruction of an astrocytic environment in glia-deficient area of white matter. J Neurocytol 1993; 22: 382–96.
Barnett SC, Franklin RJM, Blakemore WF . In vitro and in vivo analysis of a rat 0-2A progenitor cell line containing the temperature sensitive mutant gene of the SV40 large ? antigen. Eur J Neurosci 1993; 5: 1247–60.
Trotter J, Crang AJ, Schachner M, Blakemore WF . Lines of glial precursor cells immortalised with a temperature-sensitive oncogene give rise to astrocytes and oligodendrocytes following transplantation into demyelinated lesions in the central nervous system. Glia 1993; 9: 25–40.
Compston A, Scolding N, Wren D, Noble M . The pathogenesis of demyelinating disease: insights from cell biology. Trends Neurosci 1991; 14: 175–82.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Targett, M., Blakemore, W. The use of xenografting to evaluate the remyelinating potential of glial cell cultures. Eye 8, 238–244 (1994). https://doi.org/10.1038/eye.1994.52
Issue Date:
DOI: https://doi.org/10.1038/eye.1994.52
Keywords
This article is cited by
-
Reconstruction of the glial environment of a photochemically induced lesion in the rat spinal cord by transplantation of mixed glial cells
Journal of Neurocytology (1996)
-
Conduction properties of central demyelinated and remyelinated axons, and their relation to symptom production in demyelinating disorders
Eye (1994)
-
Repair of demyelinated lesions by glial cell transplantation
Journal of Neurology (1994)