Abstract
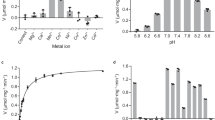
The ornithine aminotransferase (OAT) activity of mouse was found to be highest in the small intestine. The mitochondrial OAT from mouse small intestine was purified to homogeneity by the procedures including heart treatment, ammonium sulfate fractionation, octyl-Sepharose chromatography, and Sephadex G-150 gel filtration. Comparing to the amino acid sequence of mouse hepatic OAT, six N-terminal amino acid residues have been deleted in intestinal OAT. However, the subsequent sequence was identical with that of hepatic OAT. The molecular weights of both intestinal and hepatic OAT were estimated as 46 kDa by SDS-gel electrophoresis and as 92 kDa by gel filtration, indicating that both native OATs are dimeric. Biochemical properties of intestinal OAT, such as molecular weight, pH optimum and K(m) values for L-ornithine and α-ketoglutarate, were similar to those of hepatic OAT. However, intestinal OAT was more labile than hepatic OAT to tryptic digestion.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Lim, SN., Rho, HW., Park, JW. et al. A variant of ornithine aminotransferase from mouse small intestine. Exp Mol Med 30, 131–135 (1998). https://doi.org/10.1038/emm.1998.19
Published:
Issue Date:
DOI: https://doi.org/10.1038/emm.1998.19