Abstract
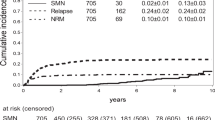
We examined the role of total body magnetic resonance imaging (TB-MRI)-governed involved compartment irradiation (ICI) and high-dose chemotherapy (HDC), followed by stem cell rescue (SCR) in patients with high-risk Ewing tumors (ETs) with multiple primary bone metastases (high-risk ET-MBM). Eleven patients with high-risk ET-MBM receiving initial assessment of involved bones by TB-MRI were registered from 1995 to 2000 (group A). In all, 6 patients out of 11 had additional lung disease at initial diagnosis; all had multifocal bone disease with more than three bones involved. After systemic induction with etoposide, vincristine, adriamycin (doxorubicin), ifosfamide, and actinomycin D (EVAIA) or VAIA chemotherapy, ICI of all sites positive by TB-MRI was administered, followed by HDC and SCR. A second group matched for observation period and consisting of 26 patients with more than three involved bones at diagnosis was treated with the European Intergroup Cooperative Ewing Sarcoma Study-92 (EICESS-92) protocol (group B). These patients did not receive TB-MRI and consequently did not receive TB-MRI-governed ICI, or HDC and SCR. Survival in group A vs group B was 45 vs 8% at 5 years and 27 vs 8% at 10 years after diagnosis (log rank and Breslow: P<0.005). We conclude that TB-MRI-governed ICI followed by HDC and SCR in ET-MBM is feasible and warrants further evaluation in prospective studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Staege MS, Hutter C, Neumann I, Foja S, Hattenhorst UE, Hansen G et al. DNA microarrays reveal relationship of Ewing family tumors to both endothelial and fetal neural crest-derived cells and define novel targets. Cancer Res 2004; 64: 8213–8221.
Schmidt D, Harms D, Burdach S . Malignant peripheral neuroectodermal tumours of childhood and adolescence. Virchows Arch A Pathol Anat Histopathol 1985; 406: 351–365.
Richter GH, Plehm S, Fasan A, Rossler S, Unland R, Bennani-Baiti IM et al. EZH2 is a mediator of EWS/FLI1 driven tumor growth and metastasis blocking endothelial and neuro-ectodermal differentiation. Proc Natl Acad Sci USA 2009; 106: 5324–5329.
Burdach S, Plehm S, Unland R, Dirksen U, Borkhardt A, Staege MS et al. Epigenetic maintenance of stemness and malignancy in peripheral neuroectodermal tumors by EZH2. Cell Cycle 2009; 8: 1991–1996.
Schmidt D, Herrmann C, Jurgens H, Harms D . Malignant peripheral neuroectodermal tumor and its necessary distinction from Ewing's sarcoma. A report from the Kiel Pediatric Tumor Registry. Cancer 1991; 68: 2251–2259.
Delattre O, Zucman J, Plougastel B, Desmaze C, Melot T, Peter M et al. Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours. Nature 1992; 359: 162–165.
Dockhorn-Dworniczak B, Schafer KL, Dantcheva R, Blasius S, van Valen F, Burdach S et al. Molecular genetic detection of t(11;22)(q24;12) translocation in Ewing sarcoma and malignant peripheral neuroectodermal tumors. Pathologe 1994; 15: 103–112.
Paulussen M, Braun-Munzinger G, Burdach S, Deneke S, Dunst J, Fellinger E et al. Results of treatment of primary exclusively pulmonary metastatic Ewing sarcoma. A retrospective analysis of 41 patients]. Klin Padiatr 1993; 205: 210–216.
Burdach S . Treatment of advanced Ewing tumors by combined radiochemotherapy and engineered cellular transplants. Pediatr Transplant 2004; 8 (Suppl 5): 67–82.
Paulussen M, Ahrens S, Burdach S, Craft A, Dockhorn-Dworniczak B, Dunst J et al. Primary metastatic (stage IV) Ewing tumor: survival analysis of 171 patients from the EICESS studies. European Intergroup Cooperative Ewing Sarcoma Studies. Ann Oncol 1998; 9: 275–281.
Link MP, Goorin AM, Miser AW, Green AA, Pratt CB, Belasco JB et al. The effect of adjuvant chemotherapy on relapse-free survival in patients with osteosarcoma of the extremity. N Engl J Med 1986; 314: 1600–1606.
Weiss RB, DeVita Jr VT . Multimodal primary cancer treatment (adjuvant chemotherapy): current results and future prospects. Ann Intern Med 1979; 91: 251–260.
Burdach S, Jurgens H, Peters C, Nurnberger W, Mauz-Korholz C, Korholz D et al. Myeloablative radiochemotherapy and hematopoietic stem-cell rescue in poor-prognosis Ewing's sarcoma. J Clin Oncol 1993; 11: 1482–1488.
Burdach S, Peters C, Paulussen M, Nurnberger W, Wurm R, Wernet P et al. Improved relapse free survival in patients with poor prognosis Ewing's sarcoma after consolidation with hyperfractionated total body irradiation and fractionated high dose melphalan followed by high dose etoposide and hematopoietic rescue. Bone Marrow Transplant 1991; 7 (Suppl 2): 95.
Frohlich B, Ahrens S, Burdach S, Klingebiel T, Ladenstein R, Paulussen M et al. High-dosage chemotherapy in primary metastasized and relapsed Ewing's sarcoma. (EI)CESS. Klin Padiatr 1999; 211: 284–290.
Engel BC, Laws HJ, Dirksen U, Kramm CM, Gobel U, Burdach SE . Circulating CD34+ cell counts as predictive parameter for the efficacy of peripheral stem cell apheresis in Ewing tumor patients. Klin Padiatr 1997; 209: 186–190.
Meyers PA, Krailo MD, Ladanyi M, Chan KW, Sailer SL, Dickman PS et al. High-dose melphalan, etoposide, total-body irradiation, and autologous stem-cell reconstitution as consolidation therapy for high-risk Ewing's sarcoma does not improve prognosis. J Clin Oncol 2001; 19: 2812–2820.
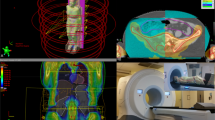
Daldrup-Link HE, Franzius C, Link TM, Laukamp D, Sciuk J, Jurgens H et al. Whole-body MR imaging for detection of bone metastases in children and young adults: comparison with skeletal scintigraphy and FDG PET. Am J Roentgenol 2001; 177: 229–236.
Burdach S, Meyer-Bahlburg A, Laws HJ, Haase R, van Kaik B, Metzner B et al. High-dose therapy for patients with primary multifocal and early relapsed Ewing's tumors: results of two consecutive regimens assessing the role of total-body irradiation. J Clin Oncol 2003; 21: 3072–3078.
Burdach S, van Kaick B, Laws HJ, Ahrens S, Haase R, Korholz D et al. Allogeneic and autologous stem-cell transplantation in advanced Ewing tumors. An update after long-term follow-up from two centers of the European Intergroup Study EICESS. Stem-Cell Transplant Programs at Dusseldorf University Medical Center, Germany and St. Anna Kinderspital, Vienna, Austria. Ann Oncol 2000; 11: 1451–1462.
Paulussen M, Craft AW, Lewis I, Hackshaw A, Douglas C, Dunst J et al. Results of the EICESS-92 Study: two randomized trials of Ewing's sarcoma treatment—cyclophosphamide compared with ifosfamide in standard-risk patients and assessment of benefit of etoposide added to standard treatment in high-risk patients. J Clin Oncol 2008; 26: 4385–4393.
Cotterill SJ, Ahrens S, Paulussen M, Jurgens HF, Voute PA, Gadner H et al. Prognostic factors in Ewing's tumor of bone: analysis of 975 patients from the European Intergroup Cooperative Ewing's Sarcoma Study Group. J Clin Oncol 2000; 18: 3108–3114.
Burdach S, Jurgens H . High-dose chemoradiotherapy (HDC) in the Ewing family of tumors (EFT). Crit Rev Oncol Hematol 2002; 41: 169–189.
Kushner BH, Meyers PA . How effective is dose-intensive/myeloablative therapy against Ewing's sarcoma/primitive neuroectodermal tumor metastatic to bone or bone marrow? The Memorial Sloan-Kettering experience and a literature review. J Clin Oncol 2001; 19: 870–880.
Steinborn MM, Heuck AF, Tiling R, Bruegel M, Gauger L, Reiser MF . Whole-body bone marrow MRI in patients with metastatic disease to the skeletal system. J Comput Assist Tomogr 1999; 23: 123–129.
Bloem JL, Taminiau AH, Eulderink F, Hermans J, Pauwels EK . Radiologic staging of primary bone sarcoma: MR imaging, scintigraphy, angiography, and CT correlated with pathologic examination. Radiology 1988; 169: 805–810.
Lang P, Grampp S, Vahlensieck M, Johnston JO, Honda G, Rosenau W et al. Primary bone tumors: value of MR angiography for preoperative planning and monitoring response to chemotherapy. Am J Roentgenol 1995; 165: 135–142.
Henk CB, Grampp S, Wiesbauer P, Zoubek A, Kainberger F, Breitenseher M et al. Ewing sarcoma. Diagnostic imaging. Radiologe 1998; 38: 509–522.
Algra PR, Bloem JL, Tissing H, Falke TH, Arndt JW, Verboom LJ . Detection of vertebral metastases: comparison between MR imaging and bone scintigraphy. Radiographics 1991; 11: 219–232.
Mehta RC, Wilson MA, Perlman SB . False-negative bone scan in extensive metastatic disease: CT and MR findings. J Comput Assist Tomogr 1989; 13: 717–719.
Frank JA, Ling A, Patronas NJ, Carrasquillo JA, Horvath K, Hickey AM et al. Detection of malignant bone tumors: MR imaging vs scintigraphy. Am J Roentgenol 1990; 155: 1043–1048.
Kattapuram SV, Khurana JS, Scott JA, el-Khoury GY . Negative scintigraphy with positive magnetic resonance imaging in bone metastases. Skeletal Radiol 1990; 19: 113–116.
Mazumdar A, Siegel MJ, Narra V, Luchtman-Jones L . Whole-body fast inversion recovery MR imaging of small cell neoplasms in pediatric patients: a pilot study. Am J Roentgenol 2002; 179: 1261–1266.
Eustace S, Tello R, DeCarvalho V, Carey J, Wroblicka JT, Melhem ER et al. A comparison of whole-body turboSTIR MR imaging and planar 99mTc-methylene diphosphonate scintigraphy in the examination of patients with suspected skeletal metastases. Am J Roentgenol 1997; 169: 1655–1661.
Acknowledgements
We thank B Metzner, Klinikum Oldenburg, Department of Hematology and Oncology, Oldenburg, and B Kremens, Department of Pediatric Hematology-Oncology, University of Essen, Essen; and the respective institutions for their participation in the study. This study was supported by unrestricted grants to SB from the Wilhelm Sander-Stiftung (2006.109.1), Else Kroener–Fresenius–Stiftung (P31/08//A123/07), BMBF (TranSarNet FK:01GM0870), AmGen and Chugai.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Burdach, S., Thiel, U., Schöniger, M. et al. Total body MRI-governed involved compartment irradiation combined with high-dose chemotherapy and stem cell rescue improves long-term survival in Ewing tumor patients with multiple primary bone metastases. Bone Marrow Transplant 45, 483–489 (2010). https://doi.org/10.1038/bmt.2009.184
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2009.184
Keywords
This article is cited by
-
Whole-body magnetic resonance imaging in children – how and why? A systematic review
Pediatric Radiology (2021)
-
Whole-body magnetic resonance imaging in pediatric oncology — recommendations by the Oncology Task Force of the ESPR
Pediatric Radiology (2020)
-
Non-odontogenic tumors of the facial bones in children and adolescents: role of multiparametric imaging
Neuroradiology (2017)
-
Optimal Management of Ewing Sarcoma Family of Tumors: Recent Developments in Systemic Therapy
Pediatric Drugs (2013)
-
Consolidation of first-line therapy with busulphan and melphalan, and autologous stem cell rescue in children with Ewing’s sarcoma
Bone Marrow Transplantation (2012)