Abstract
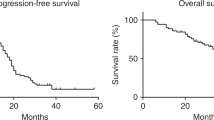
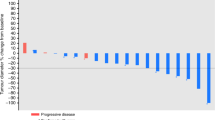
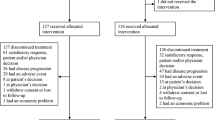
Cisplatin-based combination chemotherapy is frequently used to treat patients with carcinoma of unknown primary site (CUPS). Response rates in the literature range from 12% to 26% and median survival from 5 to 7 months. The goal of this study was to evaluate the combination of carboplatin and prolonged oral etoposide in patients with CUPS, with the hope of minimizing toxicity but improving efficacy and convenience. Treatment consisted of carboplatin, 300 mg m(-2) on day 1, and oral etoposide 50 mg on days 1-20, every 4 weeks for up to nine cycles. A total of 33 patients were treated and all were evaluable for toxicity. Non-haematological toxicity was mild to moderate, with the exception of one case of grade 4 stomatitis. Grade 4 leucopenia was observed in eight (24%) patients and sepsis in four (12%), with two and possibly three treatment-related deaths. For the 26 patients evaluable for response, the response rate was 23% with responses lasting a median of 11 months (range 7-13 months), with one patient still responding at 12 months. An additional nine patients (35%) had stable disease. Median survival for all patients was 5.6 months (range 2 weeks to 33 months). The combination of carboplatin with prolonged oral etoposide has moderate activity similar to that of other platinum-based regimens and is a well tolerated, convenient, outpatient regimen. Dosing according to estimated creatinine clearance to achieve a carboplatin AUC of 6.0 mg ml(-1) min might have decreased the incidence of severe myelotoxicity without compromising the regimen's efficacy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Warner, E., Goel, R., Chang, J. et al. A multicentre phase II study of carboplatin and prolonged oral etoposide in the treatment of cancer of unknown primary site (CUPS). Br J Cancer 77, 2376–2380 (1998). https://doi.org/10.1038/bjc.1998.395
Issue Date:
DOI: https://doi.org/10.1038/bjc.1998.395
This article is cited by
-
Phase II trials in patients with carcinoma of unknown primary: a pooled data analysis
Investigational New Drugs (2010)
-
Cancer of unknown primary site: review of consecutive cases at the National Cancer Center Hospital of Japan
International Journal of Clinical Oncology (2006)
-
Carboplatin, doxorubicin and etoposide in the treatment of tumours of unknown primary site
British Journal of Cancer (2004)
-
A phase II study of mitomycin C, cisplatin and continuous infusion 5-fluorouracil (MCF) in the treatment of patients with carcinoma of unknown primary site
British Journal of Cancer (2002)