Abstract
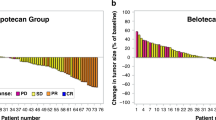
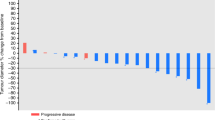
Two open, phase II studies were performed to evaluate the activity and toxicity of infusional topotecan in patients with advanced non-small-cell lung carcinoma (NSCLC) and advanced breast cancer who had not received previous chemotherapy for metastatic disease. Twenty-five patients with an ECOG performance score < 2 were treated with infusional topotecan administered as a daily, continuous intravenous infusion starting at 0.6 mg m(-2) day(-1) (NSCLC) and 0.5 mg m(-2) day(-1) (breast cancer) for 21 days every 4 weeks. Three patients achieved a partial response as defined by WHO criteria: one with NSCLC (8%; 95% CI 0-39%) and two with advanced breast cancer (15%; 95% CI 2-45%). The major toxicities were neutropenia and thrombocytopenia, with one episode of neutropenic sepsis. These data suggest that topotecan delivered as a continuous intravenous infusion over 21 days as single-agent therapy does not appear to offer a clinical advantage over conventional 5-day schedules against advanced NSCLC and advanced breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mainwaring, P., Nicolson, M., Hickish, T. et al. Continuous infusional topotecan in advanced breast and non-small-cell lung cancer: no evidence of increased efficacy. Br J Cancer 76, 1636–1639 (1997). https://doi.org/10.1038/bjc.1997.609
Issue Date:
DOI: https://doi.org/10.1038/bjc.1997.609
This article is cited by
-
A systematic review on topoisomerase 1 inhibition in the treatment of metastatic breast cancer
Breast Cancer Research and Treatment (2013)
-
Activity of topotecan given intravenously for 5 days every three weeks in patients with advanced non-small cell lung cancer pretreated with platinum and taxanes: a phase II study
Investigational New Drugs (2011)