Abstract
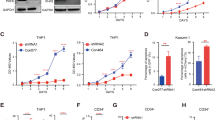
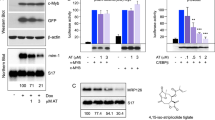
Phorbol-12-myristate-13-acetate (PMA) induces p21WAF-1 expression in human myeloid leukaemic HL-60 cells. We show that this induction is specifically mediated by protein kinase C (PKC). In addition, the PKC inhibitor Ro 31-8220 with predominant PKC-alpha isoform specificity almost completely inhibited PMA-induced up-regulation of p21WAF1 in HL-60 cells as well as in the myelomonocytic leukaemic U937 cells. Pretreatment of HL-60 cells with Ro 31-8220 also inhibited PMA-induced activation of c-raf-1, a known PKC alpha target. In the phorbol ester-tolerant HL-60 subline (PET) with PKC-beta isoform deficiency PMA or bryostatin-1 induced p21WAF1 expression, but to a lesser extent than in wild-type HL-60 cells. In PET cells, Ro 31-8220 also inhibited PMA and bryostatin-1-induced up-regulation of p21WAF1 expression. Our findings indicate that at least in HL-60 cells up-regulation of p21WAF-1 is specifically activated by PKC. We suggest that PKC isoforms other than beta, presumably the PKC-alpha isoform, are involved in this process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schwaller, J., Peters, U., Pabst, T. et al. Up-regulation of p21WAF1 expression in myeloid cells is activated by the protein kinase C pathway. Br J Cancer 76, 1554–1557 (1997). https://doi.org/10.1038/bjc.1997.595
Issue Date:
DOI: https://doi.org/10.1038/bjc.1997.595