Abstract
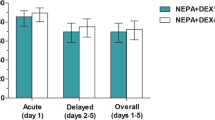
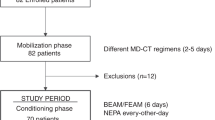
Despite recent advances in control of acute emesis following cisplatin-based chemotherapy regimens, delayed emesis remains a significant cause of treatment-related morbidity and factors associated with delayed emesis have not yet been evaluated. A prospective randomised trial was conducted to compare the efficacy and toxicity of granisetron, dexamethasone plus prochlorperazine with granisetron alone in controlling cisplatin-induced delayed emesis and to identify the important factors that influence its occurrence and severity. Seventy cisplatin-naive patients with inoperable solid tumors participated in the trial. Patients who received 80 mg m-2 or 100 mg m-2 of cisplatin were randomly assigned to receive either granisetron 40 micrograms kg-1 intravenously (i.v.) on day 1, dexamethasone 20 mg i.v. on days 2 and 3 and prochlorperazine 5 mg orally thrice daily on days 1-5 or granisetron 40 micrograms kg-1 i.v. on day 1 alone. There was no difference in their acute antiemetic efficacy. A combination regimen was more effective than granisetron alone in preventing delayed symptoms, with superior rates of complete plus major responses of 77% vs 51% (P = 0.0460). Treatment arm was the only determinant factor for the occurrence of delayed emesis (P = 0.0101).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Matsui, K., Fukuoka, M., Takada, M. et al. Randomised trial for the prevention of delayed emesis in patients receiving high-dose cisplatin. Br J Cancer 73, 217–221 (1996). https://doi.org/10.1038/bjc.1996.38
Issue Date:
DOI: https://doi.org/10.1038/bjc.1996.38
This article is cited by
-
A double-blind, crossover, randomized dose-comparison trial of granisetron for the prevention of acute and delayed nausea and emesis in children receiving moderately emetogenic carboplatin-based chemotherapy
Supportive Care in Cancer (2007)
-
Consensus proposals for the prevention of acute and delayed vomiting and nausea following high-emetic-risk chemotherapy
Supportive Care in Cancer (2005)
-
Granisetron compared with prednisolone plus metopimazine as anti-emetic prophylaxis during multiple cycles of moderately emetogenic chemotherapy
British Journal of Cancer (1999)
-
Management der Chemotherapie-induzierten Emesis: Was ist Standard nach 20 Jahren klinischer Forschung?
Medizinische Klinik (1998)