Abstract
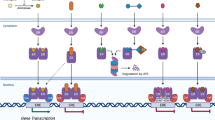
We have assessed the pharmacokinetics, pharmacological and anti-tumour effects of the specific steroidal anti-oestrogen ICI 182780 in 19 patients with advanced breast cancer resistant to tamoxifen. The agent was administered as a monthly depot intramuscular injection. Peak levels of ICI 182780 occurred a median of 8-9 days after dosing and then declined but were above the projected therapeutic threshold at day 28. Cmax during the first month was 10.5 ng/ml-1 and during the sixth month was 12.6 ng ml-1. The AUCs were 140.5 and 206.8 ng day ml-1 on the first and sixth month of dosing respectively, suggesting some drug accumulation. Luteinising hormone (LH) and follicle-stimulating hormone (FSH) levels rose after withdrawal of tamoxifen and then plateaued, suggesting no effect of ICI 182780 on the pituitary-hypothalamic axis. There were no significant changes in serum levels of prolactin, sex hormone-binding globulin (SHBG) or lipids. Side-effects were infrequent. Hot-flushes and sweats were not induced and there was no apparent effect of treatment upon the endometrium or vagina. Thirteen (69%) patients responded (seven had partial responses and six showed "no change' responses) to ICI 182780, after progression on tamoxifen, for a median duration of 25 months. Thus ICI 182780, given by monthly depot injection, and at the drug levels described, is an active second-line anti-oestrogen without apparent negative effects on the liver, brain or genital tract and warrants further evaluation in patients with advanced breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Howell, A., DeFriend, D., Robertson, J. et al. Pharmacokinetics, pharmacological and anti-tumour effects of the specific anti-oestrogen ICI 182780 in women with advanced breast cancer. Br J Cancer 74, 300–308 (1996). https://doi.org/10.1038/bjc.1996.357
Issue Date:
DOI: https://doi.org/10.1038/bjc.1996.357
This article is cited by
-
GLL398, an oral selective estrogen receptor degrader (SERD), blocks tumor growth in xenograft breast cancer models
Breast Cancer Research and Treatment (2020)
-
Fulvestrant: A Review in Advanced Breast Cancer Not Previously Treated with Endocrine Therapy
Drugs (2018)
-
Determination of Fulvestrant in Rat Plasma by LC–MS–MS: Application to a Pharmacokinetic Study
Chromatographia (2011)
-
Benefits of early and prolonged fulvestrant treatment in 848 postmenopausal advanced breast cancer patients
Breast Cancer Research and Treatment (2011)
-
Oestrogen-receptor-mediated transcription and the influence of co-factors and chromatin state
Nature Reviews Cancer (2007)