Abstract
Aim:
The present experiments were designed to study the changes in endothelium-dependent contractions with aging.
Methods:
The rat femoral arteries of 20-week and 1-year-old rats with and without endothelium were suspended in organ chambers to record isometric tension. The production of oxygen-derived free radicals in the endothelium was measured with 2′,7′-dichlorodihydrofluorescein diacetate (DCF) using confocal microscopy. Protein presences were determined by Western blotting.
Results:
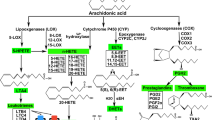
In the arteries from the 1-year-old rats, endothelium-dependent relaxations to A23187 were reduced, but the endothelium-dependent contractions to A23187 (in the presence of Nω-nitro-L-arginine methyl ester hydrochloride [L-NAME; an inhibitor of nitric oxide synthase]) were augmented, demonstrating endothelial dysfunction with aging. Indomethacin normalized the responses, suggesting that a cyclooxygenase (COX)-dependent contraction is prominent in aging. The endothelium-dependent contractions were also prevented by terutroban (a blocker of thromboxane-prostanoid receptors), confirming the activation of thromboxane-prostanoid receptors on vascular smooth muscle. Valeryl salicylate and NS-398 (preferential inhibitors of COX-1 and COX-2, respectively) partially reduced the response, indicating that both COX-1 and COX-2 are involved. Western blotting confirmed the upregulation of both isoforms in the arteries of the 1-year-old rats. In the presence of L-NAME, A23187 increased the DCF fluorescence in the endothelium, demonstrating that the production of oxygen-derived free radicals contributes to endothelium-dependent contractions. The activity of catalase was reduced in the arteries with endothelium of 1-year-old rats, indicating that hydrogen peroxide is the likely mediator of increased oxidative stress in the aging endothelium.
Conclusion:
Endothelium-dependent contractions are augmented with aging. Oxidative stress potentiates the response, and both COX-1 and COX-2 are involved.
Similar content being viewed by others
Article PDF
References
Lüscher TF, Vanhoutte PM . Endothelium-dependent contractions to acetylcholine in the aorta of the spontaneously hypertensive rat. Hypertension 1986; 8: 344–8.
Auch-Schwelk W, Katusic ZS, Vanhoutte PM . Thromboxane A2 receptor antagonists inhibit endothelium-dependent contractions. Hypertension 1990; 15: 699–703.
Yang D, Feletou M, Boulanger CM, Wu HF, Levens N, Zhang JN, et al. Oxygen-derived free radicals mediate endothelium-dependent contractions to acetylcholine in aortas from spontaneously hypertensive rats. Br J Pharmacol. 2002; 136: 104–10.
Tang EH, Feletou M, Huang Y, Man RY, Vanhoutte PM . Acetyl-choline and sodium nitroprusside cause long-term inhibition of EDCF-mediated contractions. Am J Physiol Heart Circ Physiol 2005; 289: H2434–40.
Vanhoutte PM, Feletou M, Taddei S . Endothelium-dependent contractions in hypertension. Br J Pharmacol 2005; 144: 449–58.
Gluais P, Lonchampt M, Morrow JD, Vanhoutte PM, Feletou M . Acetylcholine-induced endothelium-dependent contractions in the SHR aorta: the Janus face of prostacyclin. Br J Pharmacol 2005; 146: 834–45.
Gluais P, Paysant J, Badier-Commander C, Verbeuren T, Vanhoutte PM, Feletou M . In SHR aorta, calcium ionophore A-23187 releases prostacyclin and thromboxane A2 as endothelium-derived contracting factors. Am J Physiol Heart Circ Physiol 2006; 291: H2255–64.
Shi Y, Feletou M, Ku DD, Man RY, Vanhoutte PM . The calcium ionophore A23187 induces endothelium-dependent contractions in femoral arteries from rats with streptozotocin-induced diabetes. Br J Pharmacol 2007; 150: 624–32.
Koga T, Takata Y, Kobayashi K, Takishita S, Yamashita Y, Fujishima M . Age and hypertension promote endothelium-dependent contractions to acetylcholine in the aorta of the rat. Hypertension 1989; 14: 542–8.
Mombouli JV, Vanhoutte PM . Purinergic endothelium-dependent and -independent contractions in rat aorta. Hypertension 1993; 22: 577–83.
Auch-Schwelk W, Katusic ZS, Vanhoutte PM . Contractions to oxygen-derived free radicals are augmented in aorta of the spontaneously hypertensive rat. Hypertension 1989; 13: 859–64.
Dhein S, Salameh A, Klaus W . A new endothelium-dependent vasoconstricting factor (EDCF) in pig coronary artery. Eur Heart J 1989; 10: 82–5.
Tesfamariam B, Brown ML, Deykin D, Cohen RA . Elevated glucose promotes generation of endothelium-derived vasoconstrictor prostanoids in rabbit aorta. J Clin Invest 1990; 85: 929–32.
Shi Y, So KF, Man RY, Vanhoutte PM . Oxygen-derived free radicals mediate endothelium-dependent contractions in femoral arteries of rats with streptozotocin-induced diabetes. Br J Pharmacol 2007; 152: 1033–41.
Van der Loo B, Labugger R, Skepper JN, Bachschmid M, Kilo J, Powell JM, et al. Enhanced peroxynitrite formation is associated with vascular aging. J Exp Med 2000; 192: 1731–44.
Taddei S, Virdis A, Mattei P, Ghiadoni L, Fasolo CB, Sudano I, et al. Hypertension causes premature aging of endothelial function in humans. Hypertension 1997; 29: 736–43.
Heymes C, Habib A, Yang D, Mathieu E, Marotte F, Samuel J, et al. Cyclo-oxygenase-1 and -2 contribution to endothelial dysfunction in ageing. Br J Pharmacol 2000; 131: 804–10.
Matz RL, Schott C, Stoclet JC, Andriantsitohaina R . Age-related endothelial dysfunction with respect to nitric oxide, endothelium-derived hyperpolarizing factor and cyclooxygenase products. Physiol Res 2000; 49: 11–8.
Finkel T, Holbrook NJ . Oxidants, oxidative stress and the biology of ageing. Nature 2000; 408: 239–47.
Brandes R P, Kreuzer J . Vascular NADPH oxidases: molecular mechanisms of activation. Cardiovasc Res 2005; 65: 16–27.
Rubanyi GM, Vanhoutte PM . Superoxide anions and hyperoxia inactivate endothelium-derived relaxing factor. Am J Physiol 1986; 250: H822–827.
Katusic ZS, Vanhoutte PM . Superoxide anion is an endothelium-derived contracting factor. Am J Physiol 1989; 257: H33–37.
Tang EH, Leung FP, Huang Y, Feletou M, So KF, Man RYK, et al. Calcium and reactive oxygen species increase in endothelial cells in response to releaser of endothelium-derived contracting factor. Br J Pharmacol 2007; 151: 15–23.
Tesfamariam B, Cohen RA . Free radicals mediate endothelial cell dysfunction caused by elevated glucose. Am J Physiol 1992; 263: H321–6.
Wolin MS . Subcellular localization of Nox-containing oxidases provides unique insight into their role in vascular oxidant signaling. Arterioscler Thromb Vasc Biol 2004; 24: 625–7.
Doroudi R, Gan LM, Selin Sjogren L, Jern S . Effects of shear stress on eicosanoid gene expression and metabolite production in vascular endothelium as studied in a novel biomechanical perfusion model. Biochem Biophys Res Commun 2000; 269: 257–64.
Ge T, Hughes H, Junquero DC, Wu KK, Vanhoutte PM, Boulanger CM . Endothelium-dependent contractions are associated with both augmented expression of prostaglandin H synthase-1 and hypersensitivity to prostaglandin H2 in the SHR aorta. Circ Res 1995; 76: 1003–10.
de Sotomayor MA, Pérez-Guerrero C, Herrrera MD, Jimenez L, Marín R, Marhuenda E, et al. Improvement of age-related endothelial dysfunction by simvastatin: effect on NO and COX pathways. Br J Pharmacol 2005; 146: 1130–8.
Kang KB, Rajanayagam MA, van der Zypp A, Majewski H . A role for cyclooxygenase in aging-related changes of beta-adrenoceptor-mediated relaxation in rat aortas. Naunyn Schmiedebergs. Arch Pharmacol 2007; 375: 273–81.
Gendron ME, Thorin-Trescases N, Villeneuve L, Thorin E . Aging associated with mild dyslipidemia reveals that COX-2 preserves dilation despite endothelial dysfunction. Am J Physiol Heart Circ Physiol 2007; 292: H451–8.
Samata K, Kimura T, Satoh S, Watanabe H . Chemical removal of the endothelium by saponin in the isolated dog femoral artery. Eur J Pharmacol 1986; 128: 85–91.
Auch-Schwelk W, Katusic ZS, Vanhoutte PM . Nitric oxide inactivates endothelium-derived contracting factor in the rat aorta. Hypertension 1992; 19: 442–5.
Yang D, Gluais P, Zhang JN, Vanhoutte PM, Feletou M . Nitric oxide and inactivation of the endothelium-dependent contracting factor released by acetylcholine in spontaneously hypertensive rat. J Cardiovasc Pharmacol 2004; 43: 815–20.
Feletou M, Vanhoutte PM . Endothelial dysfunction: a multifac-eted disorder (The Wiggers Award Lecture). Am J Physiol Heart Circ Physiol 2006; 291: H985–1002.
Singer HA, Peach MJ . Calcium- and endothelial-mediated vascular smooth muscle relaxation in rabbit aorta. Hypertension 1982; 4: 19–25.
Simonet S, Descombes JJ, Vallez MO, Dubuffet T, Lavielle G, Verbeuren TJ . S 18886, a new thromboxane (TP)-receptor antagonist is the active isomer of S 18204 in all species, except in the guinea-pig. Adv Exp Med Biol 1997; 433: 173–6.
Yang D, Félétou M, Levens N, Zhang JN, Vanhoutte PM . A diffusible substance(s) mediates endothelium-dependent contractions in the aorta of SHR. Hypertension 2003; 41: 143–8.
Bhattacharyya DK, Lecomte M, Dunn J, Morgans DJ, Smith WL . Selective inhibition of prostaglandin endoperoxide synthase-1 (cyclooxygenase-1) by valerylsalicylic acid. Arch Biochem Biophys 1995; 317: 19–24.
Futaki N, Takahashi S, Yokoyama M, Arai I, Higuchi S, Otomo S . NS-398, a new anti-inflammatory agent, selectively inhibits pros-taglandin G/H synthase/cyclooxygenase (COX-2) activity in vitro. Prostaglandins 1994; 47: 55–9.
Warner TD, Mitchell JA . Cyclooxygenases: new forms, new inhibitors, and lessons from the clinic. FASEB J 2004; 18: 790–804.
Baker CS, Hall RJ, Evans TJ, Pomerance A, Maclouf J, Creminon C, et al. Cyclooxygenase-2 is widely expressed in atherosclerotic lesions affecting native and transplanted human coronary arteries and colocalizes with inducible nitric oxide synthase and nitrotyrosine particularly in macrophages. Arterioscler Thromb Vasc Biol 1999; 19: 646–55.
Schonbeck U, Sukhova GK, Graber P, Coulter S, Libby P . Augmented expression of cyclooxygenase-2 in human atherosclerotic lesions. Am J Pathol 1999; 155: 1281–91.
Belton O, Byrne D, Kearney D, Leahy A, Fitzgerald DJ . Cyclooxygenase-1 and -2-dependent prostacyclin formation in patients with atherosclerosis. Circulation 2000; 102: 840–5.
Armstead WM . Cyclooxygenase-2-dependent superoxide generation contributes to age-dependent impairment of G protein-mediated cerebrovasodilation. Anesthesiology 2003; 98: 1378–83.
Author information
Authors and Affiliations
Corresponding author
Additional information
The present study was supported in part by grants from the Research Grants Council (HKU 7524) and the Research Centre of the Heart, Brain, Hormonal and Healthy aging (HBHA).
Rights and permissions
About this article
Cite this article
Shi, Y., Man, R. & Vanhoutte, P. Two isoforms of cyclooxygenase contribute to augmented endothelium-dependent contractions in femoral arteries of 1-year-old rats. Acta Pharmacol Sin 29, 185–192 (2008). https://doi.org/10.1111/j.1745-7254.2008.00749.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00749.x
Keywords
This article is cited by
-
The glycolytic process in endothelial cells and its implications
Acta Pharmacologica Sinica (2022)
-
Deletion of T-type calcium channels Cav3.1 or Cav3.2 attenuates endothelial dysfunction in aging mice
Pflügers Archiv - European Journal of Physiology (2018)
-
Prostanoid-mediated contractions of the carotid artery become Nox2-independent with aging
AGE (2015)
-
Characteristics and formation mode of salt-bearing series in typical rift valley basin, Eastern China
Geosciences Journal (2015)
-
Non-steroidal anti-inflammatory drugs attenuate the vascular responses in aging metabolic syndrome rats
Acta Pharmacologica Sinica (2014)