Abstract
Aim:
To investigate whether a similar process mediates cytotoxicity of 1-O-octadecyl-2-O-methyl-rac-glycero-3-phosphocholine (ET-18-OCH3, edelfosine) in both yeasts and human tumor cells.
Methods:
A modified version of a previously described assay for the intracellular conversion of nitro blue tetrazolium to formazan by superoxide anion was used to measure the generation of reactive oxygen species (ROS). Apoptotic yeast cells were detected using terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay. DNA fragmentation and the generation of ROS were measured by cytofluorimetric analysis in Jurkat cells.
Results:
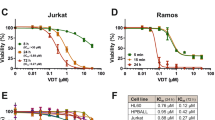
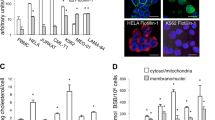
Edelfosine induced apoptosis in Saccharomyces cerevisiae, as assessed by TUNEL assay. Meanwhile, edelfosine induced a time- and concentration-dependent generation of ROS in yeasts. Rotenone, an inhibitor of the mitochondrial electron transport chain, prevented ROS generation and apoptosis in response to edelfosine in S cerevisiae. α-Tocopherol abrogated the edelfosine-induced generation of intracellular ROS and apoptosis. Edelfosine also induced an increase of ROS in human leukemic cells that preceded apoptosis. The overexpression of Bcl-2 by gene transfer abrogated both ROS generation and apoptosis induced by edelfosine in leukemic cells. Changes in the relative mitochondrial membrane potential were detected in both yeasts and Jurkat cells.
Conclusion:
These results indicate that edelfosine induces apoptosis in yeasts in addition to human tumor cells, and this apoptotic process involves mitochondria, likely through mitochondrial-derived ROS. These data also suggest that yeasts can be used as a suitable cell model in elucidating the antitumor mechanism of action of edelfosine.
Similar content being viewed by others
Article PDF
References
Mollinedo F, Martinez-Dalmau R, Modolell M . Early and selective induction of apoptosis in human leukemic cells by the alkyl-lysophospholipid ET–18-OCH3. Biochem Biophys Res Commun, 1993; 192: 603–9.
Mollinedo F, Fernandez-Luna JL, Gajate C, Martin-Martin B, Benito A, Martinez-Dalmau R, et al. Selective induction of apoptosis in cancer cells by the ether lipid ET-18-OCH3 (Edelfo-sine): molecular structure requirements, cellular uptake, and protection by Bcl-2 and Bcl-XL. Cancer Res, 1997; 57: 1320–8.
Gajate C, Mollinedo F . Biological activities, mechanisms of action and biomedical prospect of the antitumor ether phospho-lipid ET-18-OCH3 (edelfosine), a proapoptotic agent in tumor cells. Curr Drug Metab, 2002; 3: 491–525.
Mollinedo F, Gajate C, Martin-Santamaria S, Gago F . ET-18-OCH3 (edelfosine): a selective antitumour lipid targeting apoptosis through intracellular activation of Fas/CD95 death receptor. Curr Med Chem, 2004; 11: 3163–84.
Hanson PK, Malone L, Birchmore JL, Nichols JW . Lem3p is essential for the uptake and potency of alkylphosphocholine drugs, edelfosine and miltefosine. J Biol Chem, 2003; 278: 36041–50.
Zaremberg V, Gajate C, Cacharro LM, Mollinedo F, McMaster CR . Cytotoxicity of an anti-cancer lysophospholipid through selective modification of lipid raft composition. J Biol Chem, 2005; 280: 38047–58.
Gajate C, Mollinedo F . The antitumor ether lipid ET-18-OCH3 induces apoptosis through translocation and capping of Fas/CD95 into membrane rafts in human leukemic cells. Blood, 2001; 98: 3860–3.
Gajate C, Del Canto-Janez E, Acuna AU, Amat-Guerri F, Geijo E, Santos-Beneit AM, et al. Intracellular triggering of Fas aggregation and recruitment of apoptotic molecules into Fas-enriched rafts in selective tumor cell apoptosis. J Exp Med, 2004; 200: 353–65.
Gajate C, Santos-Beneit AM, Macho A, Lazaro M, Hernandez-De Rojas A, Modolell M, et al. Involvement of mitochondria and caspase-3 in ET-18-OCH3-induced apoptosis of human leukemic cells. Int J Cancer, 2000; 86: 208–18.
Vrablic AS, Albright CD, Craciunescu CN, Salganik RI, Zeisel SH . Altered mitochondrial function and overgeneration of reactive oxygen species precede the induction of apoptosis by 1-O-octadecyl-2-methyl-rac-glycero-3-phosphocholine in p53-defective hepatocytes. FASEB J, 2001; 15: 1739–44.
Nieto-Miguel T, Gajate C, Mollinedo F . Differential targets and subcellular localization of antitumor alkyl-lysophospholipid in leukemic versus solid tumor cells. J Biol Chem, 2006; 281: 14833–40.
Green DR, Reed JC . Mitochondria and apoptosis. Science, 1998; 281: 1309–12.
Richter C, Gogvadze V, Laffranchi R, Schlapbach R, Schweizer M, Suter M, et al. Oxidants in mitochondria: from physiology to diseases. Biochim Biophys Acta, 1995; 1271: 67–74.
Castedo M, Macho A, Zamzami N, Hirsch T, Marchetti P, Uriel J, et al. Mitochondrial perturbations define lymphocytes undergoing apoptotic depletion in vivo. Eur J Immunol 1995; 25: 3277–84.
Hug H, Strand S, Grambihler A, Galle J, Hack V, Stremmel W, et al. Reactive oxygen intermediates are involved in the induction of CD95 ligand mRNA expression by cytostatic drugs in hepatoma cells. J Biol Chem, 1997; 272: 28191–3.
Quillet-Mary A, Jaffrezou J P, Mansat V, Bordier C, Naval J, Laurent G . Implication of mitochondrial hydrogen peroxide generation in ceramide-induced apoptosis. J Biol Chem, 1997; 272: 21388–95.
Shaha C . Apoptosis in Leishmania species and its relevance to disease pathogenesis. Indian J Med Res, 2006; 123: 233–44.
Madeo F, Engelhardt S, Herker E, Lehmann N, Maldener C, Proksch A, et al. Apoptosis in yeast: a new model system with applications in cell biology and medicine. Curr Genet, 2002; 41: 208–16.
Phillips AJ, Sudbery I, Ramsdale M . Apoptosis induced by environmental stresses and amphotericin B in Candida albicans. Proc Natl Acad Sci USA, 2003; 100: 14327–32.
Geley S, Hartmann BL, Kofler R . Ceramides induce a form of apoptosis in human acute lymphoblastic leukemia cells that is inhibited by Bcl-2, but not by CrmA. FEBS Lett, 1997; 400: 15–8.
Rook GA, Steele J, Umar S, Dockrell HM . A simple method for the solubilisation of reduced NBT, and its use as a colorimetric assay for activation of human macrophages by gamma-interferon. J Immunol Methods, 1985; 82: 161–7.
Lopez-Barneo J, Ortega-Saenz P, Piruat JI, Garcia-Fernandez M . Oxygen-sensing by ion channels and mitochondrial function in carotid body glomus cells. Novartis Found Symp, 2006; 272: 54–64.
Gajate C, Barasoain I, Andreu JM, Mollinedo F . Induction of apoptosis in leukemic cells by the reversible microtubule-disrupt-ing agent 2-methoxy-5-(2′,3′,4′-trimethoxyphenyl)-2,4,6-cycloheptatrien-1-one: protection by Bcl-2 and Bcl-XL and cell cycle arrest. Cancer Res, 2000; 60: 2651–9.
Madeo F, Frohlich E, Ligr M, Grey M, Sigrist SJ, Wolf DH, et al. Oxygen stress: a regulator of apoptosis in yeast. J Cell Biol, 1999; 145: 757–67.
Ribeiro GF, Corte-Real M, Johansson B . Characterization of DNA damage in yeast apoptosis induced by hydrogen peroxide, acetic acid, and hyperosmotic shock. Mol Biol Cell, 2006; 17: 4584–91.
Herker E, Jungwirth H, Lehmann KA, Maldener C, Frohlich KU, Wissing S, et al. Chronological aging leads to apoptosis in yeast. J Cell Biol, 2004; 164: 501–7.
Fabrizio P, Battistella L, Vardavas R, Gattazzo C, Liou LL, Diaspro A, et al. Superoxide is a mediator of an altruistic aging program in Saccharomyces cerevisiae. J Cell Biol 2004; 166: 1055–67.
Mollinedo F, Gajate C . Microtubules, microtubule-interfering agents and apoptosis. Apoptosis, 2003; 8: 413–50.
Gavrieli Y, Sherman Y, Ben-Sasson SA . Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol, 1992; 119: 493–501.
Gottlieb RA . Mitochondrial signaling in apoptosis: mitochondrial daggers to the breaking heart. Basic Res Cardiol, 2003; 98: 242–9.
Gottlieb RA . Role of mitochondria in apoptosis. Crit Rev Eukaryot Gene Exp, 2000; 10: 231–9.
Decaudin D, Marzo I, Brenner C, Kroemer G . Mitochondria in chemotherapy-induced apoptosis: a prospective novel target of cancer therapy. Int J Oncol, 1998; 12: 141–52.
Pinkoski MJ, Waterhouse NJ, Green DR . Mitochondria, apoptosis and autoimmunity. Curr Dir Autoimmun, 2006; 9: 55–73.
Okun JG, Zickermann V, Zwicker K, Schagger H, Brandt U . Binding of detergents and inhibitors to bovine complex I - a novel purification procedure for bovine complex I retaining full inhibitor sensitivity. Biochim Biophys Acta, 2000; 1459: 77–87.
Schuler F, Yano T, Di Bernardo S, Yagi T, Yankovskaya V, Singer TP, et al. NADH-quinone oxidoreductase: PSST subunit couples electron transfer from iron-sulfur cluster N2 to quinone. Proc Natl Acad Sci USA, 1999; 96: 4149–53.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by grants from the Health Research Fund and European Commission (FIS-FEDER 04/0843 and 06/0813), Department of Education and Science of Spain (SAF2005-04293), “Mutua Madrileña” Medical Research Foundation (FMM), and “la Caixa” Foundation (BM05-30-0). Consuelo Gajate was supported by the “Ramón y Cajal” Program from the Department of Education and Science of Spain.
Rights and permissions
About this article
Cite this article
Zhang, H., Gajate, C., Yu, Lp. et al. Mitochondrial-derived ROS in edelfosine-induced apoptosis in yeasts and tumor cells. Acta Pharmacol Sin 28, 888–894 (2007). https://doi.org/10.1111/j.1745-7254.2007.00568.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00568.x
Keywords
This article is cited by
-
Nerol-induced apoptosis associated with the generation of ROS and Ca2+ overload in saprotrophic fungus Aspergillus flavus
Applied Microbiology and Biotechnology (2018)
-
Yeast caspase-dependent apoptosis in Saccharomyces cerevisiae BY4742 induced by antifungal and potential antitumor agent clotrimazole
Archives of Microbiology (2018)
-
Edelfosine-induced metabolic changes in cancer cells that precede the overproduction of reactive oxygen species and apoptosis
BMC Systems Biology (2010)