Abstract
Aim:
To investigate the in vitro metabolism of imrecoxib in rat liver microsomes and to identify the cytochrome P450 (CYP) forms involved in its metabolism.
Methods:
Liver microsomes of Wistar rats were prepared using an ultracentrifuge. The in vitro metabolism of imrecoxib was studied by incubation with rat liver microsomes. To characterize the CYP forms involved in the 4′-methyl hydroxylation of imrecoxib, the effects of typical CYP inducers (such as dexamethasone, isoniazid and β-naphthoflavone) and of CYP inhibitors (such as ketoconazole, quinine, α-naphthoflavone, methylpyrazole, and cimetidine) on the formation rate of 4′-hydroxymethyl imrecoxib were investigated.
Results:
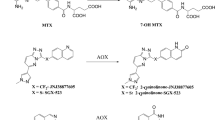
Imrecoxib was metabolized to 3 metabolites by rat liver microsomes: 4′-hydroxymethyl imrecoxib (M4), 4′-hydroxymethyl-5-hydoxyl imrecoxib (M3), and 4′-hydroxymethyl-5-car-bonyl imrecoxib (M5). Over the imrecoxib concentration range studied (5–600 μmol/L), the rate of 4′-methyl hydroxylation conformed to monophasic Michaelis-Menten kinetics. Dexamethasone significantly induced the formation of M4. Ketoconazole markedly lowered the metabolic rate of imrecoxib in a concentration-dependent manner. Moreover, a significant inhibitory effect of quinine on the formation of M4 was observed in microsomes obtained from control rats, isoniazid-induced rats, and β-naphthoflavone-induced rats. In contrast, α-naphthoflavone, cimetidine, and methylpyrazole had no inhibitory effects on this metabolic pathway.
Conclusion:
Imrecoxib is metabolized via 4′-methyl hydroxylation in rat liver microsomes. The reaction is mainly catalyzed by CYP 3 A. CYP 2D also played a role in control rats, in isoniazid-induced rats and in β-naphthoflavone-induced rats.
Similar content being viewed by others
Article PDF
References
Donnelly MT, Hawkey CJ . COX-II inhibitors, a new generation of safer NSAIDS? Aliment Pharmacol Ther 1997; 11: 227–36.
Jouzeau JY, Terlain B, Abid A, Nedelec E, Netter P . Cyclooxygenase isoenzymes: how recent findings affect thinking about nonsteroidal anti-inflammatory drugs. Drugs 1997; 53: 563–82.
Smith JM, Willis AL . Aspirin selectivity inhibits prostaglandin production in human platelets. Nat New Biol 1971; 231: 235–7.
Vane JR . Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat New Biol 1971; 231: 232–5.
Riendeau D, Charleson S, Cromlish W, Mancini JA, Wong E, Guay J . Comparison of the cyclooxygenase-1 inhibitory properties of nonsteroidal anti-inflammatory drugs (NSAIDs) and selective COX-2 inhibitors, using sensitive microsomal and platelet assays. Can J Physiol Pharmacol 1997; 75: 1088–95.
Masferrer JL, Zweifel BS, Manning PT, Hauser DS, Leahy KM, Smith WG, et al. Selective inhibition of inducible cyclooxygenase 2 in vivo is anti-inflammatory and nonulcerogenic. Proc Natl Acad Sci USA 1994; 91: 3228–32.
Needleman P, Isakson PC . The discovery and function of COX-2. J Rheumatol 1997; 24: 9–14.
Smith WL, Dewitt DL . Prostaglandin endoperoxide H synthases-1 and -2. Adv Immunol 1996; 62: 167–215.
Lane NE . Pain management in osteoarthritis: the role of COX-2 inhibitors. J Rheumatol 1997; 24: 20–4.
Simon LS, Lanza FL, Lipsky PE, Hubbard RC, Talwalker S, Schwartz BD, et al. Preliminary study of the safety and efficacy of SC-58635, a novel cyclooxygenase 2 inhibitor: Efficacy and safety in two placebo-controlled trials in osteoarthritis and rheumatoid arthritis, and studies of gastrointestinal and platelet effects. Arthritis Rheum 1998; 41: 1591–602.
Davies NM, Jamali F . COX-2 selective inhibitors cardiac toxicity: getting to the heart of the matter. J Pharm Pharm Sci 2004; 7: 332–6.
Dogne JM, Supuran CT, Pratico D . Adverse cardiovascular effects of the coxibs. J Med Chem 2005; 48: 2251–7.
Thomas LG . Increased risk of cardiovascular events with coxibs and NSAIDs. Lancet 2005; 365: 1538–9.
Bai AP, Guo ZR, Hu WH, Shen F, Cheng GF . Design, synthesis and in vitro evaluation of a new class of novel cyclooxygenase-2 inhibitors: 3,4-diaryl-3-pyrrolin-2-ones. Chin Chem Lett 2001; 12: 775–8.
Shen F, Bai AP, Guo ZR, Cheng GF . Inhibitory effect of 3,4-diaryl-3-pyrrolin-2-one derivatives on cyclooxygenase 1 and 2 in murine peritoneal macrophages. Acta Pharmacol Sin 2002; 23: 762–8.
Chen XH, Bai JY, Shen F, Bai AP, Guo ZR, Cheng GF . Imrecoxib: a novel and selective cyclooxygenase 2 inhibitor with anti-inflammatory effect. Acta Pharmacol Sin 2004; 25: 927–31.
Zhong DF, Zhang SQ, Sun L, Zhao XY . Metabolism of roxithromycin in phenobarbital-treated rat liver microsomes. Acta Pharmacol Sin 2002; 23: 455–60.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ . Protein measurement with the Folin phenol reagent. J Biol Chem 1951; 193: 265–75.
Omura T, Sato R . The carbon monoxide-binding pigment of liver microsomes. I. Evidence for its hemoprotein nature. J Biol Chem 1964; 239: 2370–8.
Yanaoka K, Tanigawara Y, Nakagawa T, Iga T . A pharmacokinetic analysis program (MULTI) for microcomputer. J Pharmacobiodyn 1981; 4: 879–85.
Boobis AR, Sesardic D, Murray BP, Edwards RJ, Singleton AM, Rich KJ, et al. Species variation in the response of the cytochrome P450-dependent monooxygenase system to inducers and inhibitors. Xenobiotica 1990; 20: 1139–61.
Paul LD, Springer D, Staack RF, Kraemer T, Maurer HH . Cytoxhrome P450 isoenzymes involved in rat liver microsomal metabolism of californine and protopine. Eur J Pharmcol 2004; 485: 69–79.
Bjornsson TD, Callaghan JT, Einolf HJ, Fischer V, Gan L, Grimm S, et al. The conduct of in vitro and in vivo drug-drug interaction studies: a pharmaceutical research and manufacturers of America (PhRMA) perspective. Drug Metab Dispos 2003; 31: 815–32.
Sandberg M, Yasar Ü, Strömberg P, Höög JO, Eliasson E . Oxidation of celecoxib by polymorphic cytochrome P450 2C9 and alcohol dehydrogenase. Br J Clin Pharmacol 2002; 54: 423–9.
Yamazaki H . Rat cytochrome P450 1A and 3A enzymes involved in bioactivation of tegafur to 5-fluorouracil and auto-induced by tegafur in liver microsomes. Drug Metab Dispos 2001; 29: 794–7.
Szotáková B, Skálová L, Baliharová V, Dvorščáková M, Štorkánová L, Šišpera L, et al. Characterization of enzymes responsible for biotransformation of the new antileukotrienic drug quinlukast in rat liver microsomes and in primary cultures of rat hepatocytes. J Pharm Pharmacol 2004; 56: 205–12.
Shimizu M, Matsushita R, Matsumoto Y, Fukuoka M . 4′-Hydroxylation of flurbiprofen by rat liver microsomes in fasting and feeding conditions. Biol Pharm Bull 2003; 26: 1448–54.
Szakács T, Veres Z, Vereczkey L . Effect of phenobarbital and spironolactone treatment on the oxidative metabolism of anti-pyrine by rat liver microsomes. Pol J Pharmacol 2001; 53: 11–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by a grant from the “863” program of China (No 2003AA2Z347C).
Rights and permissions
About this article
Cite this article
Xu, Hy., Xie, Zy., Zhang, P. et al. Role of rat liver cytochrome P450 3A and 2D in metabolism of imrecoxib. Acta Pharmacol Sin 27, 372–380 (2006). https://doi.org/10.1111/j.1745-7254.2006.00265.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00265.x