Abstract
Aim:
To investigate the ultrastructural localization of atrial natriuretic peptide (ANP)-synthesizing cells and the relationship between ANP-synthesizing cells and microvessels in rat gastric mucosa.
Methods:
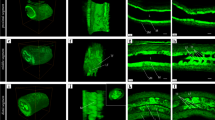
Immunohistochemistry techniques and postembedding immunoelectron microscopy techniques were used to validate the findings regarding the expression of ANP-synthesizing cells and the ultrastructural localization of ANP-synthesizing cells in the gastric mucosa. Histochemistry techniques and the tannic acid-ferric chloride method (TA-Fe staining method) were used to reveal microvessel density and the distribution of ANP-synthesizing cells in different regions of the stomach.
Results:
Cells expressing ANP were localized and ANP-synthesizing cells were identified as enterochromaffin (EC) cells in the gastric mucosa. ANP-synthesizing cells existed in different regions of the stomach. The percentage ANP-synthesizing cells in the mucosa was greatest in the fundus (46.7%±5.3%), intermediate in the antrum (40.1%±4.5%), and least in the body (21.6%±3.6%). There was a positive relationship between the percentage of ANP-synthesizing cells and the density of microvessels in the antral mucosa, but not in the fundus or body mucosa.
Conclusion:
ANP is synthesized by EC cells in rat gastric mucosa, and ANP-synthesizing cells are most dense in the gastric fundus. ANP may act not only as a regional autocrine and/or paracrine regulator, but also as an endocrine regulatory peptide in the gastrointestinal tract.
Similar content being viewed by others
Article PDF
References
De Bold AJ, Borenstein HB, Veress AT, Sonnenbery H . A rapid and potent natriuretic response to intravenous injection of atrial myocardial extracts in rats. J Am Soc Nephrol 2001; 12: 403–9.
Lai FJ, Hsieh MC, Hsin SC, Lin SR, Guh JY, Chen HC, et al. The cellular localization of increased atrial natriuretic peptide mRNA and immunoreactivity in diabetic rat kidneys. J Histochem Cytochem 2002; 50: 1501–8.
Cayli S, Ustunel I, Celik-Ozenci C, Korgun ET, Demir R . Distribution patterns of PCNA and ANP in perinatal stages of the developing rat heart. Acta Histochem 2002; 104: 271–7.
Zhao L, Mason NA, Strange JW, Walker H, Wilkins MR . Beneficial effects of phosphodiesterase 5 inhibition in pulmonary hypertension are influenced by natriuretic peptide activity. Circulation 2003; 107: 234–7.
Zhao L, Mason NA, Strange JW, Walker H, Wilkins MR . Beneficial effects of phosphodiesterase 5 inhibition in pulmonary hypertension are influenced by natriuretic peptide activity. Circulation 2003; 107: 234–7.
Peng N, Chambless BD, Oparil S, Wyss JM . Alpha2A-adrenergic receptors mediate sympathoinhibitory responses to atrial natriuretic peptide in the mouse anterior hypothalamic nucleus. Hypertension 2003; 41: 571–5.
Gower WR Jr, Salhab KF, Foulis WL, Pillai N, Bundy JR, Vesely DL, et al. Regulation of atrial natriuretic peptide gene expression in gastric antrum by fasting. Am J Physiol Regul Integr Comp Physiol 2000; 278: R770–80.
Gower WR, McCuen RW, Arimura A, Coy DA, Dietz JR, Landon CS, et al. Reciprocal paracrine pathways link atrial natriuretic peptide and somatostatin secretion in the antrum of the stomach. Regul Pept 2003; 110: 101–6.
Gerbes AL, Nathrat W, Cantin M, Denecke H . Presence of atrial natriuretic factor prohormone in enterochromaffin cells of the human large intestine. Gastroenterology 1991; 101: 424–9.
Guo HS, Xun C, Cui YG, Kim SZ, Cho KW, Li ZL, et al. Inhibitory effect of C-type natriuretic peptide on spontaneous contraction in antral circular smooth muscle of rat. Acta Pharmacol Sin 2003; 24: 1021–6.
Guo HS, Jin Z, Jin ZY, Li ZH, Cui YF, Wang ZY, et al. Comparative study in the effect of C-type natriuretic peptide on gastric motility in various animals. World J Gastroenterol 2003; 9: 547–52.
Grube D . The endocrine cells of the digestive system: amine, peptides, and modes of action. Anat Embryol 1986; 175: 151–62.
Kong XY, Zhao SM, Yang FG, Ma Q, Zhao LX, Liu S, et al. Quantitative investigations of microvessels by TA-Fe method in different regions of crerebrum. Prog Anat Sci 2004; 10: 100–2. Chinese.
Ma Q, Zhao SM, Kong XY, Rong Y, Liu S . Observation of pulmonary microvasculature and cells with tannic acid-ferric chloride (TA-Fe) staining under the light microscope. Prog Anat Sci 2003; 9: 230–2. Chinese.
Osman AH, Yuge S, Hyodo S, Sato S, Maeda S, Marie H, et al. Molecular identification and immunohistochemical localization of atrial natriuretic peptide in the heart of the dromedary camel (Camelus dromedaries). Comp Biochem Physiol A Mol Integr Physiol 2004; 139: 471–4.
Gower WR Jr, Dietz, JR, Vesely DL, Finley CL, Skolnick KA, Fabri PJ, et al. Atrial natriuretic peptide gene expression in the rat gastrointestinal tract. Biochem Biophys Res Commun 1994; 202: 562–70.
Vuolteenaho O, Arjama O, Vakkuri O, Maksniemi T, Nikkila L, Kangas J, et al. Atrial natriuretic peptide (ANP) in the rat gastrointestinal tract. FEBS Lett 1988; 233: 79–82.
Sundler F, Bottcher G, Ekblad E, Hakanson R . The neuroendocrine system of the gut. Acta Oncol 1989; 28: 303–14.
Gower WR Jr, Skvorak JP . The gastrointestinal natriuretic peptide system: a regional system with tissue-specific and vascular components. In: Vesely DL, editor. Atrial natriuretic peptides. Trivandrum, India: Research Signpost; 1997. p 139–50.
Ehrenreich H, Sinowatz F, Schulz R, Arendt RM, Goebel FD . Immunoreactive atrial natriuretic peptide (ANP) in endoscopic biopsies of human gastrointestinal tract. Res Exp Med 1989; 189: 421–5.
Li Z, Goy MF . Peptide-regulated guanylate cyclase pathways in rat colon: in situ localization of GCA, GCC, and guanylin mRNA. Am J Physiol Gastrointest Liver Physiol 1993; 265: G394–402.
Rambotti MG, Giambanco I, Spreca A . Detection of guanylate cyclases A and B stimulated by natriuretic peptides in gastrointestinal tract of rat. Histochem J 1996; 29: 117–26.
Cherner JA, Singh G, Naik L . Atrial natriuretic factor activates membrane-bound guanylate cyclase of chief cells. Life Sci 1990; 47: 669–77.
Chijiiwa Y, Kabemura T, Misawa T, Kawakami O, Nawata H . Direct inhibitory effect of calcitonin gene-related peptide and atrial natriuretic peptide on gastric smooth muscle cells via different mechanisms. Life Sci 1992; 50: 1615–23.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 30360031).
Rights and permissions
About this article
Cite this article
Li, Ch., Yang, Zw., Yin, Zr. et al. Relationship between atrial natriuretic peptide-immunoreactive cells and microvessels in rat gastric mucosa. Acta Pharmacol Sin 27, 205–211 (2006). https://doi.org/10.1111/j.1745-7254.2006.00226.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00226.x
Keywords
This article is cited by
-
Carcinoid of the esophagus concomitant with adenocarcinoma of the esophagus: One case report and literature review
Clinical Oncology and Cancer Research (2010)
-
Natriuretic Peptide-Dependent cGMP Signal Pathway Potentiated the Relaxation of Gastric Smooth Muscle in Streptozotocin-Induced Diabetic Rats
Digestive Diseases and Sciences (2010)
-
A modified ferric tannate method for visualizing a blood vessel and its usage in the study of spinal cord injury
Spinal Cord (2009)