Abstract
Aim:
To investigate the effect of antisense compounds (AS) targeting human p53 mRNA on radiosensitivity of MCF-7 cells.
Methods:
Western blotting and RT-PCR were used to analyze the protein content and mRNA level. Additionally, cell proliferation, cell cycle and cell apoptosis were all analyzed in irradiated or sham-irradiated cells.
Results:
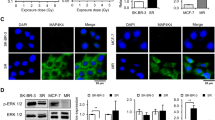
Among the five antisense compounds (AS), AS3 was identified to efficiently inhibit p53 mRNA level and protein content. Interestingly, AS3 transfer has little effect on cell proliferation in DU-145 cells (mutant p53) after ionizing radiation (IR). In contrast, a marked increase of cell apoptosis and growth inhibition were observed in MCF-7 cells (wild-type p53), suggesting that AS3 can increase radiosensitivity of MCF-7 cells. Additionally, it was also observed that the transfection of AS3 decreased the fraction of G1 phase cells, and increased the proportion of S phase cells compared to untreated cells 24 h after IR in MCF-7 cell lines.
Conclusion:
AS3 transfection increases MCF-7 cell apoptosis induced by 5 Gy-radiation, and this mechanism maybe closely associated with abrogation of G1 phase arrest.
Similar content being viewed by others
Article PDF
References
Jung M, Dritschilo A . Signal transduction and cellular responses to ionizing radiation. Semin Radiat Oncol 1996; 6: 268–72.
Gudkov AV, Komarova EA . The role of p53 in determining sensitivity to radiotherapy. Nat Rev (cancer) 2003; 3: 117–28.
Fei P, Bernhard EJ, EI-Deiry WS . Tissue-specific induction of p53 targets in vivo. Cancer Res 2002; 62: 7316–27.
Yonish-Rouach E, Resnitzky D, Lotem J, Sachs L, Kimchi A, Oren M . Wild-type p53 induces apoptosis of myeloid leukaemic cells that is inhibited interleukin-6. Nature 1991; 352: 345–7.
Yonish-Rouach E, Grunwald D, Wilder S, Kimchi A, May E, Lawrence JJ . p53-mediated cell death: relationship to cell cycle control. Mol Cell Biol 1993; 13: 1415–23.
El-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM . WAF1, a potential mediator of p53 tumour suppression. Cell 1993; 75: 817–25.
Reimer CL, Borras AM, Kurdistani SK, Garreau JR, Chung M, Aaronson SA . Altered regulation of cyclin G in human breast cancer and its specific localization at replication foci in response to DNA damage in p53+/+ cells. J Biol Chem 1999; 274: 11022–9.
Chin PL, Momand J, Pfeifer GP . In vivo evidence for binding of p53 to consensus binding sites in the p21 and GADD45 genes in response to ionizing radiation. Oncogene 1997; 15: 87–99.
Bow JJ, Howdgson B . Replication licesing-defining the proliferative state? Trends cell Biol 2002; 12: 72–8.
Short SC, Woodcock M, Marples B, Joiner MC . Effects of cell cycle phase on low-dose hyper-radiosensitivity. Int J Radiat Biol 2003; 79: 99–105.
Pawlik TM, Keyomarsi K . Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol Biol Phys 2004; 59: 928–42.
Ribeiro JCC, Barnetson AR, Fisher RJ, Mameghan H, Russell PJ . Relationship between radiation response and p53 status in human bladder cancer cells. Int J Radiat Biol 1997; 72: 11–20.
Kawashima K, Mihara K, Usuki H, Shimizu N, Namba M . Transfected mutant p53 gene increases X-ray-induced cell killing and mutation in human fibroblasts immortalized with 4-nitroquinoline 1-oxide but does not induce neoplastic transformation of the cells. Int J Cancer 1995; 61: 76–9.
Biard DSF, Martin M, Rhun YL, Duthu A, Lefaix JL, May E . Concomitant p53 gene mutation and increased radiosensitivity in rat lung embryo epithelial cells during neoplastic development. Cancer Res 1994; 54: 3361–4.
McIlwrath AJ, Vasey PA, Ross GM, Brown R . Cell cycle arrests and radiosensitivity of human tumor cell lines: dependence on wild-type p53 for radiosensitivity. Cancer Res 1994; 54: 3718–22.
Brachman DG, Beckett M, Graves D, Haraf D, Vokes E, Weichselbaum RR . p53 mutation does not correlate with radiosensitivity in 24 head and neck cancer cell lines. Cancer Res 1993; 53: 3667–9.
Böhnke A, Westphal F, Schmidt A, El-Awady A, Dahm-Daphi J . Role of p53 mutations, protein function and DNA damage for the radiosensitivity of human tumour cells. Int J Radiat Biol 2004; 1: 53–63.
Dai JM, Zhang SQ, Zhang W, Lin RX, Ji ZZ, Wang SQ . Antisense oligodeoxynucleotides targeting the serine/threonine kinase Pim-2 inhibited proliferation of DU-145 cells. Acta Pharmacol Sin 2005; 26: 364–8.
Yang SP, Song ST, Tang ZM, Song HF . Optimization of antisense drug design against conservative local motif in stimulant secondary structures of HER-2 mRNA and QSAR analysis. Acta Pharmacol Sin 2003; 24: 897–902.
Chalk AM, Sonnhammer EL . Computational antisense oligo prediction with a neural network model. Bioinformatics 2002; 18: 1567–75.
Zuker M . Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res 2003; 31: 3406–15.
Shao RG, Cao CX, Shimizu T, O'Connor PM, Kohn KW, Pommier Y . Abrogation of an S-phase checkpoint and potentiation of camptothecin cytotoxicity by 7-hydroxystaurosporine (UCN-01) in human cancer cell lines, possibly influenced by p53 function. Cancer Res 1997; 57: 4029–35.
Matsuzoe D, Hideshima T, Kimura A, Inada K, Watanabe K, Akita Y, et al. p53 mutations predict non-small cell lung carcinoma response to radiotherapy. Cancer Lett 1999; 135: 189–94.
Nicole C, Christa Z, Margit S, Ingrid S, Dan T, Uwe W, et al. p53-dependent radioresistance in ovarian carcinoma cell lines. Cancer Lett 2000; 150: 191–9.
Slichenmer WJ, Nelson WG, Slebos RJ, Kastan MB . Loss of a p53-assocaited G1 checkpoint does not decrease cell survival following DNA damage. Cancer Res 1993; 53: 4164–8.
Brachman DG, Beckett M, Graves D, Haraf D, Vokes E, Weichselbaum RR . p53 mutation does not correlate with radiosensitivity in 24 head and neck cancer cell lines. Cancer Res 1993; 53: 3667–9.
Liu XF, Xia YF, Li MZ, Wang HM, He YX, Zheng ML, et al. The effect of p21 antisense oligodeoxynucleotides on the radiosensitivity of nasopharyngeal carcinoma cells with normal p53 function. Cell Biol Int 2006; 30: 283–7.
Deng C, Zhang P, Harper JW, Elledge SJ, Leder P . Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell 1995; 82: 675–84.
Kokunai T, Urui S, Tomita H, Tamaki N . Overcoming of radioresistance in human gliomas by p21WAF1/CIP1 antisense oligonucleotide. J Neurooncol 2001; 51: 111–9.
Sak A, Wurm R, Elo B, Grehl S, Christoph P, Stuben G, et al. Increased radiation-induced apoptosis and altered cell cycle progression of human lung cancer cell lines by antisense oligodeoxynucleotides targeting p53 and p21WAF1/CIP1. Cancer Gene Ther 2003; 10: 926–34.
El-Deiry WS . The role of p53 in chemosensitivity and radiosensitivity. Oncogene 2003; 22: 7486–95.
Li G, Ho VC . p53-dependent DNA repair and apoptosis respond differently to high-and low-dose ultraviolet radiation. Br J Dermatol 1998; 139: 3–10.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by Zhejiang Province Medicine and Sanitation Research Foundation (No 2003A077).
Rights and permissions
About this article
Cite this article
Dai, Lc., Wang, X., Yao, X. et al. Antisense oligonucleotide targeting p53 increased apoptosis of MCF-7 cells induced by ionizing radiation. Acta Pharmacol Sin 27, 1453–1458 (2006). https://doi.org/10.1111/j.1745-7254.2006.00405.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00405.x
Keywords
This article is cited by
-
Genome-wide profiling induced by ionizing radiation (IR) in non-small cell lung cancer (NSCLC) grown as three-dimensional spheroid
Molecular & Cellular Toxicology (2010)