Abstract
Aim:
To develop a stable self-emulsifying formulation for oral delivery of insulin.
Methods:
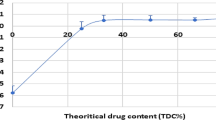
Caco-2 cell line and diabetic beagles were used as in vitro and in vivo models to study the absorption mechanism and the hypoglycemic efficacy of the formulation. In addition, various physicochemical parameters of the formulation such as droplet size, insulin encapsulation efficiency and stability were evaluated.
Results:
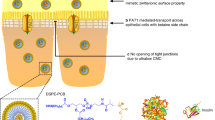
This formulation enabled changes in barrier properties of Caco-2 monolayers, as referred by transepithelial electrical resistance (TEER) and apparent permeability coefficients (Papp) of the paracellular marker ranitidine (20-fold greater than control) but not transcellular marker propranolol, suggesting that the opening of tight junctions was involved. In diabetic beagle dogs, the bioavailability of this formulation was up to 15.2% at a dose of 2.5 IU/kg in comparison with the hypoglycemic effect of native insulin (0.5 IU/kg) delivered by subcutaneous injection.
Conclusion:
This formulation, recently approved by the China State Food and Drug Administration to enter clinical trials, was stable, degradation-protected and absorption-enhanced, and provided a promising formulation for oral insulin delivery.
Similar content being viewed by others
Article PDF
References
Carino GP, Jacob JS, Mathiowitz E . Nanosphere based oral insulin delivery. J Control Release 2000; 65: 261–9.
Whitehead K, Shen Z, Mitragotri S . Oral delivery of macromolecules using intestinal patches: applications for insulin delivery. J Control Release 2004; 98: 37–45.
Pan Y, Li YJ, Zhao HY, Zheng JM, Xu H, Wei G . Bioadhesive polysaccharide in protein delivery system: chitosan nanoparticles improve the intestinal absorption of insulin in vivo. Int J Pharm 2002; 249: 139–47.
Nerurkar MM, Burton PS, Borchardt RT . The use of surfactants to enhance the permeability of peptides through Caco-2 cells by inhibition of an apically polarized efflux system. Pharm Res 1996; 13: 528–34.
Marschutz MK, Bernkop-Schnurch A . Oral peptide drug delivery: polymer-inhibitor conjugates protecting insulin from enzymatic degradation in vitro. Biomaterials 2000; 21: 1499–507.
Kawakami K, Yoshikawa T, Hayashi T, Nishihara Y, Masuda K . Microemulsion formulation for enhanced absorption of poorly soluble drugs II. In vivo study. J Control Release 2002; 81: 75–82.
New RRC, Kirby CJ . Solubilisation of hydrophilic drugs in oily formulations. Adv Drug Deliv Rev 1997; 25: 59–69.
Cilek A, Celebi N, Tirnaksiz F, Tay A . A lecithin-based micro-emulsion of rh-insulin with aprotinin for oral administration: Investigation of hypoglycemic effects in non-diabetic and STZ-induced diabetic rats. Int J Pharm 2005; 298: 176–85.
Gursoy RN, Benita S . Self-emulsifying drug delivery systems (SEDDS) for improved oral delivery of lipophilic drugs. Biomed Pharmacother 2004; 58: 173–82.
Zheng CX, Duan MX, Ma H . Inventors; a method to prepare oily insulin formulation for oral administration. PRC Patent 1 160 122. 2004 Aug 8.
Foss AC, Peppas NA . Investigation of the cytotoxicity and insulin transport of acrylic-based copolymer protein delivery systems in contact with Caco-2 cultures. Eur J Pharm Biopharm 2004; 57: 447–55.
Modamio P, Lastra CF, Montejo O, Marino EL . Development and validation of liquid chromatography methods for the quantitation of propranolol, metoprolol, atenolol and bisoprolol: application in solution stability studies. Int J Pharm 1996; 130: 137–40.
Silva-Cunha A, Grossiord JL, Puisieux F, Seiller M . W/O/W multiple emulsions of insulin containing a protease inhibitor and an absorption enhancer: preparation, characterization and determination of stability towards proteases in vitro. Int J Pharm 1997; 158: 79–89.
Deng B, Liu Z, Luo G, Ma H, Duan M . Rapid quantitative determination and assessment of insulin in oil formulation by micellar electrokinetic capillary chromatography. J Pharm Biomed Anal 2002; 27: 73–80.
Cournarie F, Savelli MP, Rosilio V, Bretez F, Vauthier C, Grossiord JL . Insulin-loaded W/O/W multiple emulsions: comparison of the performance of systems prepared with medium-chain-trig-lycerides and fish oil. Eur J Pharm Biopharm 2004; 58: 477–82.
Garti N, Aserin A, Cohen Y . Mechanistic considerations on the release of electrolytes from multiple emulsions stabilized by BSA and nonionic surfactants. J Control Release 1994; 29: 41–51.
Hu M, Borchardt RT . Transport of a large neutral amino acid in a human intestinal epithelial cell line (Caco-2): uptake and efflux of phenylalanine. Biochim Biophys Acta 1992; 1135: 233–44.
Artursson P, Karlsson J . Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human intestinal epithelial (Caco-2) cells. Biochem Biophys Res Commun 1991; 175: 880–5.
Tanaka Y, Taki Y, Sakane T, Nadai T, Sezaki H, Yamashita S . Characterization of drug transport through tight junction path-way in Caco-2 monolayer: comparison with isolated rat jejunum and colon. Pharm Res 1995; 12: 523–8.
Gan LSL, Thakker DR . Applications of the Caco-2 model in the design and development of orally active drugs: elucidation of biochemical and physical barriers posed by the intestinal epithelium. Adv Drug Deliv Rev 1997; 23: 77–98.
Lee K, Ng C, Brouwer KL, Thakker DR . Secretory transport of ranitidine and famotidine across Caco-2 cell monolayers. J Pharmacol Exp Ther 2002; 303: 574–80.
Markowska M, Oberle R, Juzwin S, Hsu CP, Gryszkiewicz M, Streeter AJ . Optimizing Caco-2 cell monolayers to increase throughput in drug intestinal absorption analysis. J Pharmacol Toxicol Methods 2001; 46: 51–5.
Hayashi M, Tomita M, Awazu S . Transcellular and paracellular contribution to transport processes in the colorectal route. Adv Drug Deliv Rev 1997; 28: 191–204.
Sha X, Yan G, Wu Y, Li J, Fang X . Effect of self-microemulsifying drug delivery systems containing Labrasol on tight junctions in Caco-2 cells. Eur J Pharm Sci 2005; 24: 477–86.
Eaimtrakarn S, Rama Prasad YV, Ohno T, Konishi T, Yoshikawa Y, Shibata N . Absorption enhancing effect of labrasol on the intestinal absorption of insulin in rats. J Drug Target 2002; 10: 255–60.
Mori S, Matsuura A, Rama Prasad YV, Takada K . Studies on the intestinal absorption of low molecular weight heparin using saturated fatty acids and their derivatives as an absorption enhancer in rats. Biol Pharm Bull 2004; 27: 418–21.
Fasano A, Uzzau S . Moldulation of intestinal tight junctions by Zonula occludens toxin permits enteral administration of insulin and other macromolecules in an animal model. J Clin Invest 1997; 99: 1158–64.
Cernea S, Kidron M, Wohlgelernter J, Modi P, Raz I . Comparison of pharmacokinetic and pharmacodynamic properties of single-dose oral insulin spray and subcutaneous insulin injection in healthy subjects using the euglycemic clamp technique. Clin Ther 2004; 26: 2084–91.
Katsuma M, Watanabe S, Kawai H, Takemura S, Sako K . Effects of absorption promoters on insulin absorption through colon-targeted delivery. Int J Pharm 2006; 307: 156–62.
Morishita M, Goto T, Nakamura K, Lowman AM, Takayama K, Peppas NA . Novel oral insulin delivery systems based on complexation polymer hydrogels: single and multiple administration studies in type 1 and 2 diabetic rats. J Control Release 2006; 110: 587–94.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Fosse Bio-Engineering Development Ltd and China National Key Foundation of Science (No 2004AA2Z3410).
Rights and permissions
About this article
Cite this article
Ma, El., Ma, H., Liu, Z. et al. In vitro and in vivo evaluation of a novel oral insulin formulation. Acta Pharmacol Sin 27, 1382–1388 (2006). https://doi.org/10.1111/j.1745-7254.2006.00424.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00424.x
Keywords
This article is cited by
-
Recent progress in hydrophobic ion-pairing and lipid-based drug delivery systems for enhanced oral delivery of biopharmaceuticals
Journal of Pharmaceutical Investigation (2022)
-
Encapsulation of Resveratrol Using Water-in-Oil-in-Water Double Emulsions
Food Biophysics (2010)