Abstract
OBJECTIVE: First, to determine the sensitivity, specificity, and positive predictive value (PPV) of the direct antiglobulin test (DAT) for significant hemolysis in the neonate, as referenced to end-tidal carbon monoxide, the criterion standard for estimating the rate of hemolysis; and second, to evaluate the predictive value of the two procedures for significant jaundice.
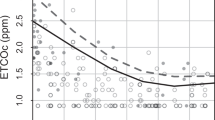
DESIGN: Consecutive term newborns admitted to the nursery of an inner-city university hospital over a 15-week period. DAT screening by the Blood Bank was performed on all. End-tidal carbon monoxide levels were obtained at 12±6 and at 24±6 hours of age. Infants of nonsmoking mothers whose 12-hour exhaled carbon monoxide level was ≥95th percentile were defined as having significant hemolysis.
RESULTS: n=660; DAT was positive in 23 (3.5%). Using the 12-hour end-tidal carbon monoxide ≥3.2 μl/l (≥95th percentile) as reference (n=499 nonsmokers), the sensitivity of the DAT was 38.5% (10 of 26) and specificity 98.5% (466 of 473) for the detection of significant hemolysis. The PPV of the DAT for significant hemolysis at 12 hours was 58.8% (10 of 17). For significant jaundice the PPV of end-tidal carbon monoxide was greater than that for DAT (65.4% vs 52.9%), although not statistically so (p=0.25). The negative predictive values were similar.
CONCLUSION: DAT fails to identify over half of the cases of significant hemolysis that are diagnosed by end-tidal carbon monoxide. A neonate with a positive DAT has about a 59% chance of having significant hemolysis. End-tidal carbon monoxide may also provide a more sensitive index for predicting significant jaundice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Valaes T, Koliopoulos C, Koltsidopoulos A . The impact of phototherapy in the management of neonatal hyperbilirubinemia: comparison of historical cohorts Acta Paediatr 1996; 85: 273–6
Newman TB, Easterling MJ . Yield of reticulocyte counts and blood smears in term infants Clin Pediatr 1994; 33: 2 71–6
Ramasethu J, Luban NLC . Alloimmune hemolytic disease of the newborn In: Beutler E, Coller BS, Lichtman MA, Kipps TJ, Seligsohn U, editors Williams Hematology 6th ed New York: McGraw-Hill 2001; 33: 2 p 665–75
Stevenson DK, Vreman HJ . Carbon monoxide and bilirubin production in neonates Pediatrics 1997; 100: 252–9
Rubaltelli FF, Gourley GR, Loskamp N et al. Transcutaneous bilirubin measurement: a multicenter evaluation of a new device Pediatrics 2001; 107: 1264–71
Hayde M, Bernaschek G, Stevenson DK, Knight GJ, Haddow JE, Widness JA . Antepartum fetal and maternal carboxyhemoglobin and cotinine levels among cigarette smokers Acta Paediatr 1999; 88: 3 327–31
Bhutani VK, Johnson L, Sivieri EM . Predictive ability of a predischarge hour-specific serum bilirubin for subsequent significant hyperbilirubinemia in healthy term and near-term newborns Pediatrics 1999; 103: 6–14
Bhutani VK, Gourley GR, Adler S, Kreamer B, Dalin C, Johnson LH . Noninvasive measurement of total serum bilirubin in a multiracial predischarge newborn population to assess the risk of severe hyperbilirubinemia Pediatrics 2000; 106: e17
American Academy of Pediatrics. Practice parameter: management of hyperbilirubinemia in the healthy term newborn Pediatrics 1994; 94: 558–65
Gordis L . Assessing the validity and reliability of diagnostic and screening tests In Epidemiology 2nd ed Philadelphia: WB Saunders 1996; p 58–76
Leisenring W, Pepe MS, Longton G . A marginal regression modelling framework for evaluating medical diagnostic tests Stat Med 1997; 16: 1263–81
Bennett BM . On comparisons of sensitivity, specificity and predictive value of a number of diagnostic procedures Biometrics 1972; 28: 793–800
Vreman HJ, Mahoney JJ, Stevenson DK . Electrochemical measurement of carbon monoxide in breath: interference by hydrogen Atmos Environ 1993; 27: 14 2193–8
Joint Commission on Accreditation of Healthcare Organizations. Sentinel event alert: kernicterus threatens healthy newborns Issue 18 April 2001
Centers for Disease Control. Kernicterus in full-term infants — United States, 1994–1998 MMWR 2001; 50: 23 491–4
Coombs RAA, Mourant AE, Race RR . A new test for detection of weak and “incomplete” Rh agglutinins Br J Exp Pathol 1945; 26: 255–66
Simpson PP, Hall PE . The antiglobulin test In: Harmening DM, editor Modern Blood Banking and Transfusion Practices 4th ed Philadelphia: FA Davis 1999; p 71–87
Merry AH, Thomson EE, Rawlinson VI, Stratton F . Quantitation of IgG on erythrocytes: correlation of numbers of IgG molecules per cell with the strength of the direct and indirect antiglobulin tests Vox Sang 1984; 47: 73–81
Lapierre Y, Rigal D, Adam J et al. The gel test: a new way to detect red cell antigen–antibody reactions Transfusion 1990; 30: 109–13
Alvarez A, Rives S, Montoto S, Sanz C, Pereira A . Relative sensitivity of direct antiglobulin test, antibody's elution and flow cytometry in the serologic diagnosis of immune hemolytic transfusion reactions Haematologica 2000; 85: 186–8
Hachimura K, Uchiyama Y, Ohtani H . A sensitive enzyme immunoassay for the measurement of small quantities of erythrocyte-associated IgG in patients with systemic lupus erythematosus who had negative direct antiglobulin test Nihon Rinsho Meneki Gakkai Kaishi 1999; 22: 63–71
Barker RN, Gruffydd-Jones TJ, Stokes CR, Elson CJ . Autoimmune haemolysis in the dog: relationship between anaemia and the levels of red blood cell bound immunoglobulins and complement measured by an enzyme-linked antiglobulin test Vet Immunol Immunopathol 1992; 34: 1–20
Lau P, Haesler WE, Wurzel HA . Positive direct antiglobulin reaction in a patient population Am J Clin Pathol 1976; 65: 368–75
Yust I, Frisch B, Goldsher N . Antibody-dependent cell-mediated cytotoxicity and phagocytosis of autologous red blood cells in alphamethyldopa-induced haemolysis Scand J Haematol 1986; 36: 211–6
Doyle JJ, Schmidt B, Blanchette V, Zipursky A . Hematology In: Avery GB, Fletcher MA, MacDonald MG, editors Neonatology: Pathophysiology and Management of the Newborn Philadelphia: Lippincott Williams & Wilkins 1999; p 1045–91
Calhoun DA, Christiansen RD, Edstrom CS et al. Consistent approaches to procedures and practices in neonatal hematology Clin Perinatol Sept 2000; 27: 733–53
Beutler E . G-6-PD deficiency and other red cell enzyme abnormalities In: Beutler E, Coller BS, Lichtman MA, Kipps TJ, Seligsohn U, editors Williams Hematology 6th ed New York: McGraw-Hill 2001; p 527–45
Kaplan M, Hammerman C, Vreman HJ, Stevenson DK, Beutler E . Acute hemolysis and severe neonatal hyperbilirubinemia in glucose-6-phosphate dehydrogenase deficient heterozygotes J Pediatr 2001; 139: 137–40
Valaes T . Commentary: problems with prediction of neonatal hyperbilirubinemia Pediatrics 2001; 108: 175–7
Stevenson DK, Fanaroff AA, Maisels MJ et al. Prediction of hyperbilirubinemia in near-term and term infants Pediatrics 2001; 108: 31–9
Caraballo RS, Giovino GA, Pechacek TF, Mowery PD . Factors associated with discrepancies between self-reports on cigarette smoking and measured serum cotinine levels among persons aged 17 years or older Am J Epidemiol 2001; 153: 807–14
Ebrahim SH, Floyd RL, Merritt RK II, Decoufle P, Holtzman D . Trends in pregnancy-related smoking rates in the United States, 1987–1996 JAMA 2000; 283: 361–6
Acknowledgements
This study was supported by a grant and equipment from Natus Medical. The authors thank Linda Mack, RN, for her invaluable help with the data collection. We also thank Hector L. Hernandez for his conscientious assistance with the case report forms; and Chris Chung, MD, for his thoughtful contributions. We are grateful to Herbert T. Abelson, MD, for his helpful review of the manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Herschel, M., Karrison, T., Wen, M. et al. Evaluation of the Direct Antiglobulin (Coombs') Test for Identifying Newborns at Risk for Hemolysis as Determined by End-Tidal Carbon Monoxide Concentration (ETCOc); and Comparison of the Coombs' Test With ETCOc for Detecting Significant Jaundice. J Perinatol 22, 341–347 (2002). https://doi.org/10.1038/sj.jp.7210702
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7210702
This article is cited by
-
Comparison of end-tidal carbon monoxide measurements with direct antiglobulin tests in the management of neonatal hyperbilirubinemia
Journal of Perinatology (2020)
-
Effect of genetic variants of bilirubin metabolism on the degree of hyperbilirubinemia in African-American newborns
Journal of Perinatology (2017)
-
Carboxyhemoglobin levels as a predictor of risk for significant hyperbilirubinemia in African-American DAT+ infants
Journal of Perinatology (2016)
-
Post-test probability for neonatal hyperbilirubinemia based on umbilical cord blood bilirubin, direct antiglobulin test, and ABO compatibility results
European Journal of Pediatrics (2016)
-
End-tidal carbon monoxide and hemolysis
Journal of Perinatology (2014)