Abstract
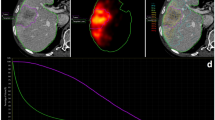
Insufficient blood flow within colo-rectal hepatic metastases is a factor which may limit drug delivery to, and thus the response of, these tumours to regional chemotherapy. Loco-regional flow may be manipulated pharmacologically to enhance the tumour blood flow relative to that of the normal liver. However, as yet, only transient effects have been studied. Patients receiving regional chemotherapy for unresectable hepatic disease were given a 45 min regional infusion of the vasoconstrictor Angiotensin II. Intrahepatic blood flow distribution was assessed serially by Positron Emission Tomography (PET) imaging together with the trapping tracer copper(II) pyruvaldehyde bis(N-4-methylthiosemicarbazone) (Cu-PTSM) labelled using copper-62. Eleven lesions in nine patients were studied, with no adverse effects. Prior to Angiotensin II administration tumour blood flow was generally found to be greater than that of liver (10/11 lesions; 8/9 patients; median TNR 1.3, iqr 0.9–2.5). A significant increase in relative flow to tumour was seen in response to 10 min Angiotensin II infusion in most cases (7/11 lesions; 7/9 patients; median TNR 2.1, iqr 1.4–4.1; P = 0.008), which appeared to be sustained throughout the 45 min infusion period (median TNR 1.85, iqr 1.3–3.8; P = 0.03). These effects were accompanied by transient elevation of mean arterial pressure, but no change in pulse rate. These observations suggest that prolonged regional vasoconstrictor administration could prove useful in the management of unresectable colo-rectal hepatic metastases, and that further development of vascular manipulation to enhance tumour targeting and drug delivery is warranted. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ackerman NB (1974) The blood supply of experimental liver metastases. IV. Changes in vascularity with increasing tumour growth. Surgery 75: 589–596
Allen-Mersh TG, Earlam S, Fordy C, Abrams K and Houghton J (1994) Quality of life and survival in patients with colorectal liver metastases treated with continuous hepatic artery floxuridine by an implanted pump. Lancet 344: 1255–1260
Ashraf S, Loizidou M, Crowe R, Turmaine M, Taylor I and Burnstock G (1997) Blood vessels in liver metastases from both sarcoma and carcinoma lack perivascular innervation and smooth muscle cells. Clin Exp Metastasis 15: 484–498
Daly JM, Butler J, Kemeny N, Yeh SDJ, Ridge JA, Biotet J, Bading JR, Decosse JJ and Benua RS (1985) Predicting tumour response in patients with colorectal hepatic metastases. Ann Surg 202: 384–393
Dimitrakopoulou-Strauss A, Strauss LG, Schlag P, Hohenberger P, Irngartinger G, Oberdorfer F, Doll J and van Kaick G (1998) Intravenous and intra-arterial oxygen-15-labeled water and fluorine-18-labeled fluorouracil in patients with liver metastases from colorectal carcinoma. J Nucl Med 39: 465–473
Dworkin MJ, Carnochan P and Allen-Mersh TG (1995) Nitric oxide inhibition sustains vasopressin-induced vasoconstriction. Br J Cancer 71: 942–944
Dworkin MJ, Carnochan P and Allen-Mersh TG (1997) Effect of continuous regional vasoactive agent infusion on liver metastasis blood flow. Br J Cancer 76: 1205–1210
Flower MA, Young HE, Wells K, Zweit J, Mundy J, Hall A and Ott RJ (1996) Optimisation of acquisition parameters for 62Cu imaging with MUP-PET. Nucl Med Commun 17: 266
Flower MA, Zweit J, Hall AD, Burke D, Davies MM, Dworkin MJ, Young H, Mundy J, Ott RJ, McCready VR, Carnochan P and Allen-Mersh TG (2001) 62Cu-PTSM and PET for the assessment of angiotensin II induced blood flow changes in patients with colorectal liver metastases. Eur J Nucl Med 28: 99–103
Goldberg JA, Thompson JAK, Bradnam MS, Fenner J, Bessent RG, McKillop JH, Kerr DJ and McArdle CS (1991) Angiotensin II as a potential method of targeting cytotoxic-loaded microspheres in patients with colorectal liver metastases. Br J Cancer 64: 114–119
Hafström L, Nobin A, Persson B and Sundkvist K (1980) Effects of catecholamines on cardiovascular response and blood flow distribution to normal tissue and liver tumours in rats. Cancer Res 40: 481–485
Hemingway DM, Angerson WJ, Anderson JH, Goldberg JA, McArdle CS and Cooke TG (1992) Monitoring blood flow to colorectal liver metastases using laser Doppler flowmetry: the effect of angiotensin II. Br J Cancer 66: 958–960
Hennigan T, Earlam S and Allen-Mersh TG (1992) Is liver to lung shunting in colorectal liver metastasis the cause of toxicity following treatment with cytotoxic microsphere aggregates?. Br J Cancer 66: 1169–1171
Marsden PK, Ott RJ, Bateman JE, Cherry SR, Flower MA and Webb S (1989) The performance of a multiwire chamber positron camera for clinical use. Phys Med Biol 34: 1043–1062
Mathias CJ, Welch MJ, Raichle ME, Mintun MA, Lich LL, McGuire AH, Zinn KR, John EK and Green MA (1990) Evaluation of a potential generator-produced PET tracer for cerebral perfusion imaging: single-pass cerebral extraction measurements and imaging with radiolabelled Cu-PTSM. J Nucl Med 31: 351–359
Mathie RT, Ralevic V, Alexander B and Burnstock G (1991) Nitric oxide is the mediator of ATP-induced dilatation of the rabbit hepatic arterial vascular bed. Br J Pharmacol 103: 1602–1606
Nettie PA, Laurence TB, Boucher Y, Skalak R and Jain RK (1995) Time-dependent behavior of interstitial fluid pressure in solid tumors: implications for drug delivery. Can Res 55: 5451–5458
Nettie PA, Hamberg LM, Babich JW, Kierstead D, Graham W, Hunter GJ, Wolf GL, Fishman A, Boucher Y and Jain RK (1999) Enhancement of fluid filtration across tumor vessels: implication for delivery of macromolecules. Proc Natl Acad Sci USA 96: 3137–3142
Robb RA and Barillot C (1998) Interactive display and analysis of 3-D medical images. IEEE Trans Med Imag 18: 217–266
Rougier P (1998) Are there indications for intraarterial hepatic chemotherapy or isolated liver perfusion? The case of liver metastases from colorectal cancer. Recent Results Cancer Res 147: 3–12
Sasaki Y, Imaoka S, Hasegawa Y, Nakano S, Ishikawa O, Ohigashi H, Taniguchi K, Koyama H, Iwanaga T and Terasawa T (1985) Changes in distribution of hepatic blood flow induced by intra-arterial infusion of angiotensin II in human hepatic cancer. Cancer 55: 311–316
Wallhaus TR, Lacy J, Whang J, Green MA, Nickles RJ and Stone CK (1998) Human biodistribution and dosimetry of the PET perfusion agent copper-62-PTSM. J Nucl Med 39: 1958–1964
Zweit J, Goodall R, Cox M, Babich JW, Potter GA, Sharma HL and Ott RJ (1992) Development of a high performance zinc-62/copper-62 radionuclide generator for positron emission tomography. Eur J Nud Med 19: 418–425
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Burke, D., Davies, M., Zweit, J. et al. Continuous angiotensin II infusion increases tumour: normal blood flow ratio in colo-rectal liver metastases. Br J Cancer 85, 1640–1645 (2001). https://doi.org/10.1054/bjoc.2001.2152
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.2152
Keywords
This article is cited by
-
Angiotensin II increases gene expression after selective intra-arterial adenovirus delivery in a rabbit model assessed using in vivo SSTR2-based reporter imaging
EJNMMI Research (2016)
-
Recommendations for measurement of tumour vascularity with positron emission tomography in early phase clinical trials
European Radiology (2012)
-
The potential of positron-emission tomography to study anticancer-drug resistance
Nature Reviews Cancer (2004)